Vibrio vulnificus: Difference between revisions
m (kmg) |
|||
| Line 9: | Line 9: | ||
Bacteria; Proteobacteria; Gammaproteobacteria; Vibrionales; Vibrionaceae; Vibrio | Bacteria; Proteobacteria; Gammaproteobacteria; Vibrionales; Vibrionaceae; Vibrio | ||
===Species=== | ===Genus and Species=== | ||
''Vibrio vulnificus'' | ''Vibrio vulnificus'' | ||
==Description and significance== | ==Description and significance== | ||
''V. vulnificus'', a Gram-negative, rod shaped, halophile, is a virulent bacterium associated with about 95 percent of all seafood related deaths.(7) It is a human pathogen known to cause gastroenteritis, which is an infection of the stomach and intestines, wound infections and primary septicemia, which occurs when the bacterium | ''V. vulnificus'', a Gram-negative, rod shaped, halophile, is a virulent bacterium associated with about 95 percent of all seafood related deaths.(7) It is a human pathogen known to cause gastroenteritis, which is an infection of the stomach and intestines, wound infections and primary septicemia, which occurs when the bacterium enters the host's blood and causes infection throughout the body. | ||
This organism can typically be found in salty, coastal waters, thriving especially in molluscan shellfish including oysters and clams, | This organism can typically be found in salty, coastal waters, thriving especially in molluscan shellfish including oysters and clams, thereby causing health risks to those who ingest raw or undercooked seafood. | ||
''V. vulnificus'' has three biotypes, with biotype one, identified as a new species in 1976, | ''V. vulnificus'' has three biotypes, with biotype one, identified as a new species in 1976, causing the greatest public health concern due to its involvement with human disease.(7) Furthermore, biotype two is associated with infected eels, and biotype three is associated with wound infections of humans.(5) | ||
==Genome structure== | ==Genome structure== | ||
The genomes of two strains of ''V. vulnificus'', YJ016 and CMCP6, have been fully sequenced, each with two, circular chromosomes.(6) | The genomes of two strains of ''V. vulnificus'', YJ016 and CMCP6, have been fully sequenced, each with two, circular chromosomes.(6) | ||
Chromosome I of strain YJ016 has a length of 3,354,505 nucleotides, while chromosome II contains 1,857,073 nucleotides, with a size of 5,260,086 base pairs.(4) | Chromosome I of strain YJ016 has a length of 3,354,505 nucleotides, while chromosome II contains 1,857,073 nucleotides, with a total size of 5,260,086 base pairs.(4) | ||
Chromosome I of strain CMCP6 has 3,281,945 nucleotides in length, while chromosome II has 1,844,853 nucleotides, | Chromosome I of strain CMCP6 has 3,281,945 nucleotides in length, while chromosome II has 1,844,853 nucleotides, for a total size of 5,126,798 base pairs.(4) | ||
It is believed that ''Vibrios'' have two chromosomes as an evolutionary advantage. The larger chromosome plays a role in growth, while the smaller one is involved in adaptation and environmental change.(7) Phenotypic variation is also present within ''V. vulnificus''. It is observed that no two strains are genetically identical. | It is believed that ''Vibrios'' have two chromosomes as an evolutionary advantage. The larger chromosome plays a role in growth, while the smaller one is involved in adaptation and environmental change.(7) Phenotypic variation is also present within ''V. vulnificus''. It is observed that no two strains are genetically identical. | ||
| Line 31: | Line 31: | ||
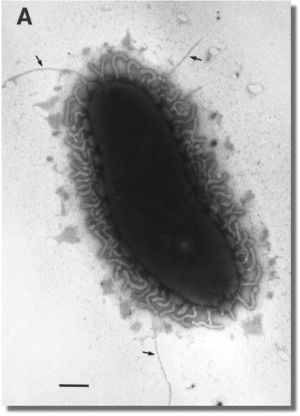
[[Image:Bimm120_vvpili_picture.jpg|thumbnail|right|''Vibrio vulnificus'' pili is used for many reasons such as motility, adherence and secretion. Courtesy of Rohinee Paranjpye and Mark Strom, Northwest Fisheries Science Center, NOAA Fisheries Service, Seattle, WA]] | [[Image:Bimm120_vvpili_picture.jpg|thumbnail|right|''Vibrio vulnificus'' pili is used for many reasons such as motility, adherence and secretion. Courtesy of Rohinee Paranjpye and Mark Strom, Northwest Fisheries Science Center, NOAA Fisheries Service, Seattle, WA]] | ||
''V. vulnificus'' is a Gram-negative bacterium, which contains an outer membrane which surrounds a thin layer of petidoglycan, or cell wall, and an inner membrane. It contains pili- | ''V. vulnificus'' is a Gram-negative bacterium, which contains an outer membrane which surrounds a thin layer of petidoglycan, or cell wall, and an inner membrane. It contains pili- hairlike appendages found on the surface of the bacteria- for motility, adherence to epithelial cells and involvement in biofilm formation. Some strains contain capsules which determine virulence and are also associated with biofilm formation.(3) In addition, iron is an important factor for growth and siderophores are produced, which are receptors that bind and deliver the iron to the bacteria.(1) | ||
This organism is also known to be “lactose-positive” with its fermentation of lactose, salicin and cellobiose. But since phenotypic variation is observed, about 15% of strains were found to be lactose negative and another 15% were found to be sucrose positive.(7) | This organism is also known to be “lactose-positive” with its fermentation of lactose, salicin and cellobiose. But since phenotypic variation is observed, about 15% of strains were found to be lactose negative and another 15% were found to be sucrose positive.(7) | ||
| Line 42: | Line 42: | ||
''V. vulnificus'' has a generalized stress response.(5) Depending on its environment, changes induce production of heat shock proteins in response to different temperatures, changes in osmolarity, the presence of pollutants, or its interactions with other organisms. With the production of these proteins, the bacteria may adapt comfortably in its environment. | ''V. vulnificus'' has a generalized stress response.(5) Depending on its environment, changes induce production of heat shock proteins in response to different temperatures, changes in osmolarity, the presence of pollutants, or its interactions with other organisms. With the production of these proteins, the bacteria may adapt comfortably in its environment. | ||
Typically, more of this organism can be found in waters during the summer months, with its optimal temperature being between 20 to 30 degrees Celsius.(7) Furthermore, ''V. vulnificus'', a halophile, | Typically, more of this organism can be found in waters during the summer months, with its optimal temperature being between 20 to 30 degrees Celsius.(7) Furthermore, ''V. vulnificus'', a halophile, thriving in saline environments. | ||
Other marine sources for this bacterium, other than water and shell fish, also include plankton, other types of fish and isolated sediment.(7) | Other marine sources for this bacterium, other than water and shell fish, also include plankton, other types of fish and isolated sediment.(7) | ||
| Line 62: | Line 62: | ||
Since it is known that ''V. vulnificus'' thrives in saline conditions, some researchers have conducted experiments to observe the result of exposing the ''V. vulnificus'' YJ03 strain in low-salinity environments. Results show that a strain adapted to acid, bile and heat stresses, did not survive the exposure to low salinity. On the other hand, strains of ''V. vulnificus'' that were adapted to bile stresses and were in the state of exponential and stationary growth, where cross protected from low-salinity.(8) | Since it is known that ''V. vulnificus'' thrives in saline conditions, some researchers have conducted experiments to observe the result of exposing the ''V. vulnificus'' YJ03 strain in low-salinity environments. Results show that a strain adapted to acid, bile and heat stresses, did not survive the exposure to low salinity. On the other hand, strains of ''V. vulnificus'' that were adapted to bile stresses and were in the state of exponential and stationary growth, where cross protected from low-salinity.(8) | ||
When it comes to food contamination, research has been done to find a way to detect different kinds of pathogens, even if they are present in very low concentrations. An assay found to detect these pathogens, including ''V. vulnificus'', is called buoyant density gradient centrifugation, quickly separating the pathogen from food matrices. After the centrifugation, the next procedure is PCR (polymerase chain reaction) to | When it comes to food contamination, research has been done to find a way to detect different kinds of pathogens, even if they are present in very low concentrations. An assay found to detect these pathogens, including ''V. vulnificus'', is called buoyant density gradient centrifugation, quickly separating the pathogen from food matrices. After the centrifugation, the next procedure is PCR (polymerase chain reaction) to detect bacterial DNA that could be present even in minute concentrations.(10) | ||
Other researches have investigated ways to inhibit or prevent ''V. vulnificus'' growth in oysters. They observed the effects of using electrolyzed oxidizing (EO) water, acting as an antibacterial against ''V. vulnificus''. Within seconds of exposing the oysters to the EO water, a decrease in the bacterium population occurred. Further experimentation showed that exposure to EO water, containing high amounts of chlorine, for more than 12 hours, were harmful to the oyster. They observed that the optimal exposure time ranged between 4 to 6 hours, without being hazardous to the oyster. In light of this discovery, they suggested that this treatment be used after harvest to reduce contamination of the seafood.(9) | Other researches have investigated ways to inhibit or prevent ''V. vulnificus'' growth in oysters. They observed the effects of using electrolyzed oxidizing (EO) water, acting as an antibacterial against ''V. vulnificus''. Within seconds of exposing the oysters to the EO water, a decrease in the bacterium population occurred. Further experimentation showed that exposure to EO water, containing high amounts of chlorine, for more than 12 hours, were harmful to the oyster. They observed that the optimal exposure time ranged between 4 to 6 hours, without being hazardous to the oyster. In light of this discovery, they suggested that this treatment be used after harvest to reduce contamination of the seafood.(9) | ||
Revision as of 15:34, 11 July 2007
A Microbial Biorealm page on the genus Vibrio vulnificus
Classification
Higher order taxa
Bacteria; Proteobacteria; Gammaproteobacteria; Vibrionales; Vibrionaceae; Vibrio
Genus and Species
Vibrio vulnificus
Description and significance
V. vulnificus, a Gram-negative, rod shaped, halophile, is a virulent bacterium associated with about 95 percent of all seafood related deaths.(7) It is a human pathogen known to cause gastroenteritis, which is an infection of the stomach and intestines, wound infections and primary septicemia, which occurs when the bacterium enters the host's blood and causes infection throughout the body.
This organism can typically be found in salty, coastal waters, thriving especially in molluscan shellfish including oysters and clams, thereby causing health risks to those who ingest raw or undercooked seafood.
V. vulnificus has three biotypes, with biotype one, identified as a new species in 1976, causing the greatest public health concern due to its involvement with human disease.(7) Furthermore, biotype two is associated with infected eels, and biotype three is associated with wound infections of humans.(5)
Genome structure
The genomes of two strains of V. vulnificus, YJ016 and CMCP6, have been fully sequenced, each with two, circular chromosomes.(6)
Chromosome I of strain YJ016 has a length of 3,354,505 nucleotides, while chromosome II contains 1,857,073 nucleotides, with a total size of 5,260,086 base pairs.(4)
Chromosome I of strain CMCP6 has 3,281,945 nucleotides in length, while chromosome II has 1,844,853 nucleotides, for a total size of 5,126,798 base pairs.(4)
It is believed that Vibrios have two chromosomes as an evolutionary advantage. The larger chromosome plays a role in growth, while the smaller one is involved in adaptation and environmental change.(7) Phenotypic variation is also present within V. vulnificus. It is observed that no two strains are genetically identical.
Cell structure and metabolism
V. vulnificus is a Gram-negative bacterium, which contains an outer membrane which surrounds a thin layer of petidoglycan, or cell wall, and an inner membrane. It contains pili- hairlike appendages found on the surface of the bacteria- for motility, adherence to epithelial cells and involvement in biofilm formation. Some strains contain capsules which determine virulence and are also associated with biofilm formation.(3) In addition, iron is an important factor for growth and siderophores are produced, which are receptors that bind and deliver the iron to the bacteria.(1)
This organism is also known to be “lactose-positive” with its fermentation of lactose, salicin and cellobiose. But since phenotypic variation is observed, about 15% of strains were found to be lactose negative and another 15% were found to be sucrose positive.(7)
In addition, this bacterium is a known halophile, and usually grows in areas with a minimum of 5% salinity.(7)
Lipopolysaccharides can be found on the outer layer of the membrane, and contain endotoxin.(5) This trait is the major cause for the organism’s virulence and is the leading factor which causes shock or death in an infected individual.
Ecology
V. vulnificus has a generalized stress response.(5) Depending on its environment, changes induce production of heat shock proteins in response to different temperatures, changes in osmolarity, the presence of pollutants, or its interactions with other organisms. With the production of these proteins, the bacteria may adapt comfortably in its environment.
Typically, more of this organism can be found in waters during the summer months, with its optimal temperature being between 20 to 30 degrees Celsius.(7) Furthermore, V. vulnificus, a halophile, thriving in saline environments.
Other marine sources for this bacterium, other than water and shell fish, also include plankton, other types of fish and isolated sediment.(7)
Pathology
This bacterium may infect an individual if ingested or can enter the body through an open wound. From there, it invades the GI tract or blood and can lead to septicemia, depending if the person is healthy or has a vulnerable immune system. Symptoms of invasion include blister-like lesions on the skin.(5) Other symptoms also include fever, nausea or gastroenteritis.(1)
V. vulnificus has a significant number of potential determinants of virulence. Some determinants include the presence of a capsule to avoid host phagocytosis, Type IV pili used to attach to host cells or for iron transfer, and biofilm formation.(3) Another major determinant for virulence is the presence of lipopolysaccharides, an endotoxin, which are responsible for septicemia, shock and even death.(1)
On the contrary, biotype two is associated exclusively with eels, causing eel vibriosis or death, regardless of the presence of a capsule on the bacterium. This type differs from biotype one in other ways including the production of exoproteins and the O-antigenic side chain on the lipopolysaccharide molecule.(7)
Application to Biotechnology
This microbe does not produce any antibiotics, but does produce many enzymes and cytoxins. Some of these enzymes and cytotoxins include proteases, endotoxin, lipopolysaccharides, hemolysin, lipases, DNAases and cytolysins.(1) All of these are known virulence factors, contributing to gastroenteritis, septicemia and infection of wounds.
Furthermore, this microbe produces additional sigma factors, or initiation factors for prokaryotic transcription. One in particular, called RpoS, demonstrates a role in regulation to environmental stresses.(7) Its mutant shows a decrease in motility and a decrease in production of a couple of virulence factors. The production of cyclic AMP and a mutant in the cya gene shows similar results in inhibition and decrease production or activity.(7)
Current Research
Since it is known that V. vulnificus thrives in saline conditions, some researchers have conducted experiments to observe the result of exposing the V. vulnificus YJ03 strain in low-salinity environments. Results show that a strain adapted to acid, bile and heat stresses, did not survive the exposure to low salinity. On the other hand, strains of V. vulnificus that were adapted to bile stresses and were in the state of exponential and stationary growth, where cross protected from low-salinity.(8)
When it comes to food contamination, research has been done to find a way to detect different kinds of pathogens, even if they are present in very low concentrations. An assay found to detect these pathogens, including V. vulnificus, is called buoyant density gradient centrifugation, quickly separating the pathogen from food matrices. After the centrifugation, the next procedure is PCR (polymerase chain reaction) to detect bacterial DNA that could be present even in minute concentrations.(10)
Other researches have investigated ways to inhibit or prevent V. vulnificus growth in oysters. They observed the effects of using electrolyzed oxidizing (EO) water, acting as an antibacterial against V. vulnificus. Within seconds of exposing the oysters to the EO water, a decrease in the bacterium population occurred. Further experimentation showed that exposure to EO water, containing high amounts of chlorine, for more than 12 hours, were harmful to the oyster. They observed that the optimal exposure time ranged between 4 to 6 hours, without being hazardous to the oyster. In light of this discovery, they suggested that this treatment be used after harvest to reduce contamination of the seafood.(9)
References
4. "Vibrio". National Microbial Pathogen Data Resource Center.
5. Todar, Kenneth. "Vibrio vulnificus". Todar's Online Textbook of Bacteriology. 2005.
7. [ Thompson, F., Austin, B., and Swings, J. (Eds.). (2006). The Biology of Vibrios. (pp. 349-354, 359-361). Washington, D.C.: ASM Press.]
Edited by Kristine Yambao, student of Rachel Larsen and Kit Pogliano