Sulfolobus solfataricus: Difference between revisions
| Line 54: | Line 54: | ||
[[Image:Vocano_solfatara2_sm.jpg|frame|right|The red of these rocks found near Naples, Italy is created by ''S. solfataricus'' colonization. Image courtesy of Science@NASA (1998).]] | [[Image:Vocano_solfatara2_sm.jpg|frame|right|The red of these rocks found near Naples, Italy is created by ''S. solfataricus'' colonization. Image courtesy of Science@NASA (1998).]] | ||
''Sulfolobus solfataricus'' is a thermoacidophile, and was first characterized in sulfur-rich volcanic springs in Yellowstone National Park. Strains of ''S. solfataricus'' have also been found in hot mud pools in the Solfatara crater north of Naples, Italy. It has also been discovered in El Salvador and the Dominica. While the optimum temperature for growth is ≈75<font size="-1"><sup>o</sup></font>C, ''S. solfataricus'' can live in a range of 55-90<font size="-1"><sup>o</sup></font>C. It can live in a pH range of 0.9-5.8, with its optimum being 2-3. However, ''S. solfataricus'' maintains its cytoplasmic pH at 6.5. It has also been shown that ''S. solfataricus'' requires an aerobic environment, in which it exists as a free-living sulfur chemolithotroph. | ''Sulfolobus solfataricus'' is a thermoacidophile, and was first characterized in sulfur-rich volcanic springs in Yellowstone National Park. Strains of ''S. solfataricus'' have also been found in hot mud pools in the Solfatara crater north of Naples, Italy. It has also been discovered in El Salvador and the Dominica. While the optimum temperature for growth is ≈75<font size="-1"><sup>o</sup></font>C, ''S. solfataricus'' can live in a range of 55-90<font size="-1"><sup>o</sup></font>C. It can live in a pH range of 0.9-5.8, with its optimum being 2-3. However, ''S. solfataricus'' maintains its cytoplasmic pH at 6.5. It has also been shown that ''S. solfataricus'' requires an aerobic environment, in which it exists as a free-living sulfur chemolithotroph. | ||
[[Image:sulfolobus_6.JPG|frame|left|Fusellovirus SSV1. Courtesy of | [[Image:sulfolobus_6.JPG|frame|left|Fusellovirus SSV1. Courtesy of Prangishvili, Stedman, Zillig (2001). Scale bar = 200nm]] | ||
Some studies have shown that ''S. solfataricus'' auto-aggregate when exposed to UV light, creating multi-cellular structures. Extracellular type-IV pili-like structures have been observed due to the UV-induced increase in transcription of the pili-like forming genes. The formation of ''S. solfataricus'' aggregates has been shown to be independent of pH and temperature. In addition, strains of ''S. solfataricus'' are known to harbor plasmids that encourage bacteria-like conjugation between cells. However, the genes behind these mechanisms are dissimilar between ''S. solfataricus'' and bacteria. | Some studies have shown that ''S. solfataricus'' auto-aggregate when exposed to UV light, creating multi-cellular structures. Extracellular type-IV pili-like structures have been observed due to the UV-induced increase in transcription of the pili-like forming genes. The formation of ''S. solfataricus'' aggregates has been shown to be independent of pH and temperature. In addition, strains of ''S. solfataricus'' are known to harbor plasmids that encourage bacteria-like conjugation between cells. However, the genes behind these mechanisms are dissimilar between ''S. solfataricus'' and bacteria. | ||
Revision as of 22:39, 19 April 2009
Classification
Archaea; Crenarchaeota; Thermoprotei; Sulfolobales; Sulfolobaceae
Species
|
NCBI: Taxonomy |
Sulfolobus solfataricus
Description and Significance
Sulfolobus solfataricus grow in volcanic hot springs where they have ample sulfur and low pH. S. solfataricus is a very widely studied crenarchaeal organism. It is used as a model organism in archaeal research, including DNA replication, the cell cycle, chromosomal integration, and RNA processing.
Proteins found in S. solfataricus are being investigated for use in biotechnology because they remain functional in a wide range of temperatures. S. solfataricus has also been used on the US Space Shuttle for protein crystallography studies.
Genome Structure
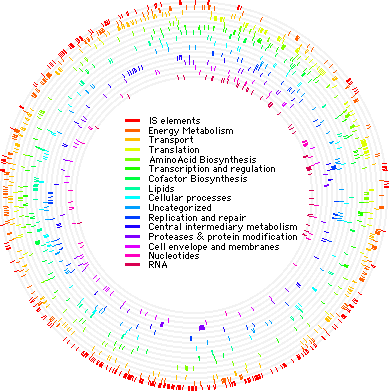
The genome of S. solfataricus P2 was sequenced in 2001. It consists of a single circular chromosome containing 2,992,245 base pairs. There are 3,032 genes which encode 2,977 proteins. It has been shown that approximately one-third of the S. solfataricus P2 genome is unique. However, much of the genome (≈40%) is archaeal, while small portions overlap with Bacteria and Eukarya (12 and 2.3%, respectively). It has its own 743 open reading frames, and about 11% of the genome is made up of mobile elements.
The S. solfataricus P2 genome has already been studied extensively. It has been integral in the study of crenarchaeal biology, and has been compared to many euryarchaea to elucidate differences between the two groups. It has also provided more evidence for the separations of archaea, eukarya, and bacteria. The S. solfataricus P2 genome has been manipulated for studies of transcription, translation, the cell cycle, and other genetic events. Currently, the Sulfolobus solfataricus 98/2 strain is being sequenced for further studies.
Cell Structure, Metabolism and Life Cycle
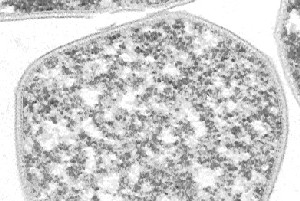
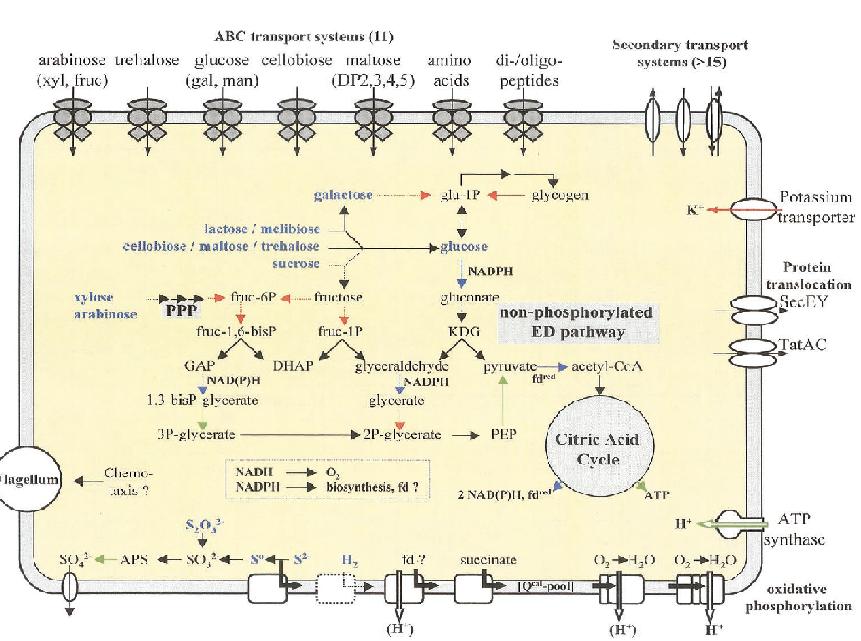
Sulfolobus solfataricus has a spherical shape and a clockwise flagellum. Its outer wall is unusual in that it does not contain any peptidoglycan. The cell wall contains: proteins that mediate the transport of essential carbohydrates into the cell; ion channels that keep potassium levels at vital concentrations; and ATPases that uptake protons from oxidative phosphorylation.
S. solfataricus utilizes the glycolitic pathway to convert glucose to pyruvate, which is used in the citric acid cycle. Glucose is not available in the environment, and so S. solfataricus converts the carbohydrates present into glucose at an expense of energy. Glycolysis yields no net energy; however, the citric acid cycle produces some ATP and reduces NADP+ to NADPH that it can be used to drive oxidative phosphorylation. S. solfataricus also utilizes the pentose phosphate pathway when as a method of obtaining NADPH from carbohydrates that do not go through the glycolitic pathway.
S. solfataricus is able to synthesize all 20 amino acids.
S. solfataricus requires oxygen as its final electron acceptor. When necessary, S. solfataricus can utilize sulfide and hydrogen as electron donors.
Ecology and Pathogenesis
Sulfolobus solfataricus is a thermoacidophile, and was first characterized in sulfur-rich volcanic springs in Yellowstone National Park. Strains of S. solfataricus have also been found in hot mud pools in the Solfatara crater north of Naples, Italy. It has also been discovered in El Salvador and the Dominica. While the optimum temperature for growth is ≈75oC, S. solfataricus can live in a range of 55-90oC. It can live in a pH range of 0.9-5.8, with its optimum being 2-3. However, S. solfataricus maintains its cytoplasmic pH at 6.5. It has also been shown that S. solfataricus requires an aerobic environment, in which it exists as a free-living sulfur chemolithotroph.
Some studies have shown that S. solfataricus auto-aggregate when exposed to UV light, creating multi-cellular structures. Extracellular type-IV pili-like structures have been observed due to the UV-induced increase in transcription of the pili-like forming genes. The formation of S. solfataricus aggregates has been shown to be independent of pH and temperature. In addition, strains of S. solfataricus are known to harbor plasmids that encourage bacteria-like conjugation between cells. However, the genes behind these mechanisms are dissimilar between S. solfataricus and bacteria.
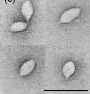
S. solfataricus can be infected by morphologically unusual viruses, such as SSV1 (Sulfolobus spindle-shaped virus). The virions formed by SSV1 often cluster together, forming rosette-like shapes. These viruses have been found in Yellowstone National Park, Iceland, and Japan.
References
Author
Page authored by Alvin Makohon-Moore and Antoni Malachowski, students of Prof. Jay Lennon at Michigan State University.