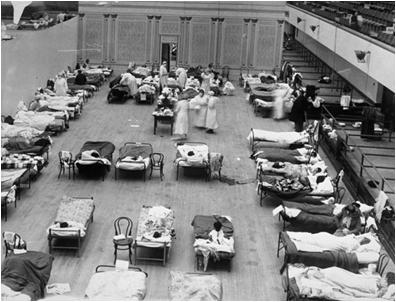
|
|
| Line 10: |
Line 10: |
| | | |
|
| |
|
| <H4>References</H4> | | <H4>Major Topic 3</H4> |
| “Avian Influenza or Bird Flu: Reference Summary.” (2008) “X-Plain Avian Influenza or Bird Flu,” http://www.nlm.nih.gov/medlineplus/tutorials/avianflu/id509103.pdf<BR>
| | Discuss major topic 3.<br><br> |
| Bakalar, N. (2007) “How (and How Not) to Battle Flu: A Tale of 23 Cities” New York Times http://www.nytimes.com/2007/04/17/health/17flu.html<BR>
| |
| Baum, L. (2006) “The Deadliest Fall,” http://ideaexplore.net/news/041116.html<BR>
| |
| Billings, M. (2005) “The Influenza Pandemic of 1918.” http://www.stanford.edu/group/virus/uda/<BR>
| |
| Borenstein, S. (2007) “Research on monkeys finds resurrected 1918 flu killed by turning the body against itself,” http://www.usatoday.com/tech/science/discoveries/flu-research.htm?POE=NEWISVA<BR>
| |
| Center for Infectious Disease Research and Policy, University of Minnesota, http://id_center.apic.org/cidrap/content/influenza/panflu/biofacts/panflu.html<BR>
| |
| Cummings, S. “Spanish Influenza Outbreak, 1918” http://history-world.org/spanish_influenza_of_1918.htm<BR>
| |
| Duffy, M. (2002) “The Influenza Pandemic” First World War: The War to End All Wars. http://www.firstworldwar.com/atoz/influenza.htm<BR>
| |
| "Influenza Epidemic of 1918–19" (2009) Encyclopædia Britannica Online http://www.britannica.com/EBchecked/topic/287805/influenza-epidemic-of-1918-19<BR>
| |
| Kindt, T.J., R.A. Goldsby and B.A. Osborne (2007) Kuby Immunology, 6th Revised Ed, WH Freeman and Co, NY; p 302<BR>
| |
| Lamb, R.A, and D. Jackson (2005) “Extinct 1918 virus comes alive,” <i>Nature Med</i> <b>11</b>1154 – 1156<BR>
| |
| Mamelund, S.-E. (2001) “Effects of the Spanish Influenza pandemic on fertility and nuptiality in Norway” http://www.iussp.org/Brazil2001/s30/S34_P01_Mamelund.pdf<BR>
| |
| Morens, D.M., and A.S. Fauci (2007) “The 1918 Influenza Pandemic: Insights for the 21st Century,” <i>J Infect Dis</i> <b>195</b>:1018-1028<BR>
| |
| Parsons, D. (2006) “The Spanish Lady and the Newfoundland Regiment” WWI: The Medical Front. Dr. Geoffrey Miller” http://www.vlib.us/medical/parsons.htm<BR>
| |
| Osterholm, M.T. (2005) “Preparing for the Next Pandemic,” <i>New Eng J Med</i> <b>352</b>:1839-1842<BR>
| |
| Osterholm; M.T., and A.L. Petrosino (2005) “Cytokine Storm and the Influenza Pandemic,” http://www.cytokinestorm.com/<BR>
| |
| Researchers unlock secrets of 1918 flu pandemic” (2008) http://www.reuters.com/article/newsOne/idUSTRE4BS56420081229?pageNumber=1&virtualBrandChannel=0<BR>
| |
| Schoenstadt, A. (2008) “Spanish Flu” http://flu.emedtv.com/spanish-flu/spanish-flu.html<BR>
| |
| Taubenberger, JK., and D.M. Morens “1918 Influenza: The Mother of All Pandemics.” The Center for Disease Control. 2005. Emerging Infectious Diseases. 8 April 2009. <http://www.cdc.gov/ncidod/eid/vol12no01/05-0979.htm#cit>.<BR>
| |
| “The Great Pandemic: The United States in 1918 – 1919.” http://1918.pandemicflu.gov/the_pandemic/04.htm<BR>
| |
| “The Threat of Pandemic Influenza: Are We Ready?” National Academies of Science Workshop Summary (2005) http://www.nap.edu/openbook.php?record_id=11150&page=1<BR>
| |
| “The Deadly Virus: The Influenza Epidemic of 1918” National Archives and Records Administration http://www.archives.gov/exhibits/influenza-epidemic/<BR>
| |
| “The 1918 Flu Virus is Resurrected” (2005) <i>Nature</i> <b>437</b>:794-795. <BR>
| |
| “1918 Spanish flu treatment may also be effective for current avian influenza patients,” http://www.news-medical.net/?id=19832<BR>
| |
| <H2>Other Influenza Epidemics</h2>
| |
| <H3>The Asian Flu 1957-1958</H3>
| |
| The 1957-1958 Asian flu pandemic was not as virulent as the 1918 pandemic. There were far fewer deaths and cases of the flu, but it was important for two reasons. First, the incidence rate was highest for children; second, it was the first pandemic for which a vaccine was available. The pandemic was responsible for an estimated 2 million deaths worldwide. Those most affected by the pandemic were school children, young adults, pregnant women, and the elderly <BR><BR>
| |
| | |
| The pandemic had two waves. It began in China in February 1957. By June it had spread to Asia, Europe, and America, affecting primarily school age children. A second wave occurred in January and February, 1958 and affected the elderly more than children. The first wave was attributed to children going back to school in the Fall of 1957. The close contact with other children helped spread the disease. The average age of those affected was 6-12. Schoolchildren then helped spread the flu by taking it home to their families. Many schools closed for a time to combat the spread of the pandemic, a measure that was somewhat effective. Although the infection rate in the first wave was highest for children and adolescents, the elderly had the highest death rate. The lower incidence in the elderly may have been due to partial immunity because most had been exposed to the Spanish flu of 1918. They had a higher death rate however because of their age. <BR><BR>
| |
| | |
| Because of medical advances since the 1918 Spanish flu, the virus responsible for this new epidemic was quickly identified as a H2N2 type, and a vaccine was quickly made. This was the first vaccine available for a flu pandemic. It was available by May of 1957, it became generally accessible to the U.S. by August and by October in Britain. Despite availability, in the U.S., less than half of the 60 million doses were used.
| |
| <H4>References:</H4>
| |
| http://cns.miis.edu/flu_watch/history.htm<BR>
| |
| “Influenza: A Short History of the Disease,” http://lhncbc.nlm.nih.gov/apdb/phsHistory/health_news.html<BR>
| |
| “Pandemic Influenza,” http://www.globalsecurity.org/security/ops/hsc-scen-3_pandemic-influenza.htm <BR>
| |
| “1957: British public gets 'Asian Flu' vaccine,” http://news.bbc.co.uk/onthisday/hi/dates/stories/october/1/newsid_3086000/3086843.stm
| |
| <H3>The Hong Kong Flu 1968-1969</H3>
| |
| The Hong Kong virus, an H3N2 strain, evolved from the Asian Flu virus, which circulated from 1957 to 1968. (3, 7) The Hong Kong flu was probably a result of antigenic drift, meaning that the original virus was an Asian Flu virus with an advantageous mutation that kept the human immune system from recognizing it as a pathogen. Because Hong Kong Flu evolved from Asian Flu, many people who had Asian Flu were immune or partially immune, largely due to the overlapping N2 subtype. Mortality worldwide was 500,000 to 750,000 from 1968 to 1969. The United States recorded 34,000 deaths. The Hong Kong Flu reappeared in 1970 and in 1972 but has not been seen since. <BR><BR>
| |
| | |
| Hong Kong Flu appeared in the United States in September of 1968, possibly brought by soldiers returning from Vietnam. The incidence escalated through December and into January, the month with the highest number of fatalities. This is a slightly atypical timeline. Flu season typically begins in early October and goes through March, with the highest number of cases in occurring in February. The Hong Kong Flu peaked several weeks earlier. The height of the infectious period coincided with schools’ winter breaks. Children, who suffered the highest incidence of infection, were at home where they were more isolated than if they had been at school. This may have lowered the number of people exposed to contagious patients and stunted the flu’s spread. Many schools closed which likely also helped keep the pandemic from worsening. The US Department of Health and Human Services called it “the mildest pandemic of the twentieth century.” <BR><BR>
| |
| | |
| Hong Kong Flu was first recognized in July of 1968 and the World Health Organization (WHO) determined its H3N2 type on August 16th. The WHO provided antibiotics, though as is always the case with a viral infection, they were ineffective. Vaccines were manufactured in the United States, but they were not finished in time to prevent the disease from spreading.
| |
| <h4>References</h4>
| |
| http://medicine.science-tips.org/health/diseases-and-conditions/hong-kong-flu.html
| |
| http://cns.miis.edu/flu_watch/history.htm<BR>
| |
| “Pandemics and Pandemic Scares in the Twentieth Century,” http://www.hhs.gov/nvpo/pandemics/flu3.htm<BR>
| |
| Earn, D.J.D., J. Dushoff and S.A. Levin (2002) “Ecology and evolution of the flu,” <i>Trends Ecol Evol</i> <b>17</b>:334-340. <BR>
| |
| http://www.medterms.com/script/main/art.asp?articlekey=26429<BR>
| |
| Harder, T.C., and W. Ortrud (2006) “Avian Influenza,” in <i>Influenza Report</i>, ed. B.S. Kamps, C. Hoffmann and W.Preiser, Flying Publisher (www.flyingpublisher.com), Ch. 2<BR>
| |
| http://www.cdc.gov/flu/about/season/flu-season.htm<BR>
| |
| http://www.pandemicflu.gov/general/historicaloverview.html<BR>
| |
| http://www.publications.parliament.uk/pa/ld200506/ldselect/ldsctech/88/8805.htm
| |
| <H3>Avian Influenza A, H5N1</H3>
| |
| <H4>Introduction</H4>
| |
| Avian influenza A (H5N1) is an influenza virus subtype that occurs primarily in bird populations. The current form of virus does not typically infect humans unless there is direct, extensive contact with infected poultry or other contaminated surfaces (1). Fewer than 500 human cases of H5N1 influenza have been reported worldwide in the 5 years since the virus first crossed the species barrier from birds to humans. Nonetheless, there is cause for concern because the H5N1 virus has caused the largest number of cases of severe disease and death in humans of any avian flu virus. At present, human-to-human spread of the virus has been “limited, inefficient and unsustained” (2). Even without human-to-human transmission, fatality rates are over 50%, far higher than from any other flu epidemic in history (2,3). By comparison, the fatality rate for recent pandemics has been about 0.1% while the Spanish flu of 1918-1919 had a fatality rate of about 2.5%. This high rate for the H5N1 virus is presumably because there is little preexisting immunity to its (4). Should the virus develop an improved ability to spread from person-to-person, a devastating pandemic could occur.
| |
| It should be noted that the fatality rate data collected by the WHO may be skewed because it is the most severe cases that are reported. Cases that are not as severe go unreported to the WHO, thus making it seem as if there are a disproportionate number of fatalities (5).
| |
| <H4>History</H4>
| |
| The highly pathogenic H5N1 virus was first isolated in 1996 from farmed geese in Guangdong Province, China near Hong Kong. T he following year, the first confirmed cases of human infection with H5N1 were reported in 18 patients in Hong Kong with 6 fatalities (6). By 2004, the virus had spread through the bird populations of East Asia, becoming endemic. Widespread human cases were reported for the first time. <BR><BR>
| |
| | |
| The virus has predominantly affected humans in Vietnam and Indonesia and China with fewer cases in other Southeast Asian countries (see Table). Egypt has also shown a number of cases with a very high fatality rate. Over the last several years the H5N1 virus has become increasingly endemic in bird populations further and further from Hong Kong. Infections in birds have been confirmed throughout most of Asia and the Middle East, with human cases reported as well in countries as far from Hong Kong as Nigeria (2).<BR><BR>
| |
| <b>Human Incidence of H5N1 by Country</b> (2)<br> <table border="1" width="300"> <tr> <td><b>Country</b></td> <td><b>Cases</b></td> <td><b>Deaths</b></td> </tr> <tr> <td>Azerbaijan</td> <td>8</td> <td>5</td> </tr> <tr> <td>Bangladesh</td> <td>1</td> <td>0</td> </tr> <tr> <td>Cambodia</td> <td>8</td> <td>7</td> </tr> <tr> <td>China</td> <td>38</td> <td>25</td> </tr> <tr> <td>Djibouti</td> <td>1</td> <td>0</td> </tr> <tr> <td>Egypt</td> <td>63</td> <td>23</td> </tr> <tr> <td>Indonesia</td> <td>141</td> <td>115</td> </tr> <tr> <td>Iraq</td> <td>3</td> <td>2</td> </tr> <tr> <td>Laos PDR</td> <td>2</td> <td>2</td> </tr> <tr> <td>Myanmar</td> <td>1</td> <td>0</td> </tr> <tr> <td>Nigeria</td> <td>1</td> <td>1</td> </tr> <tr> <td>Pakistan</td> <td>3</td> <td>1</td> </tr> <tr> <td>Thailand</td> <td>25</td> <td>17</td> </tr> <tr> <td>Turkey</td> <td>12</td> <td>4</td> </tr> <tr> <td>Vietnam</td> <td>110</td> <td>55</td> </tr><tr> <td><b>Total</b></td> <td>417</td> <td>257</td> </tr></table>
| |
| <H4>Treatment</H4>
| |
| As for all influenza strains, there are several antiviral drugs available to reduce the duration and severity of flu caused by the H5N1 virus. A popular drug for treating this strain of flu is the neuraminidase inhibitor Oseltamivir (marketed as Tamiflu). This drug is potentially efficacious because it not only targets proteins that are common to all influenza A strains but also targets proteins especially important in the H5N1 genetic structure (7).<BR><BR>
| |
| | |
| However, due to rapid mutation of the H5N1virus (8) and poor agricultural practices, there already are viral strains that are immune to some drugs. Drugs targeting M2 viral coat protein (amantadine or rimantadine) are generally cheaper than neuraminidase inhibitors like Tamiflu, although less effective. They were therefore originally chosen by the World Health Organization (WHO) to combat H5N1. When the drugs were found to be ineffective, it was discovered that poultry farmers in South China commonly administered these drugs to poultry, thus allowing H5N1 to evolve to become immune to this particular class of drugs. As a result in 2005, pharmaceutical company Hoffman-La Roche donated 3 million doses of Tamiflu to the WHO to prevent a pandemic (7,8).
| |
| <H4>Pandemic Prevention</H4>
| |
| Currently, the WHO conducts extensive monitoring for the H5N1 virus. In 2005, the WHO released a document recommending a number of “strategic actions” to prevent a pandemic from H5N1, and should it occur, to contain it as best as possible (9). One of the pre-pandemic objectives is to “reduce the opportunities for human infection” (9) through vaccinating all at-risk workers (9). This would significantly reduce the most likely transmission route by which the H5N1 virus could become a virus capable of human to human transmission (8). Over time, as more people are infected, the chances of a genetic exchange between human flu virus and avian flu virus will increase, thus allowing the avian flu virus more opportunities to create a transmissible human flu virus. Should a pandemic virus emerge, the WHO has plans to take steps to “contain or delay spread at the source. As part of this goal of slowing evolution of the virus, approximately 150 million birds either infected or at extreme risk of infection have been “destroyed” since 2003 (8).<BR><BR>
| |
| | |
| The most pressing concern is that the extremely virulent H5N1 avian virus could acquire one of the hemagglutinins reactive with human cells and thus be carried by humans. The most straightforward way for this to occur would be for a human flu virus and the H5N1 avian flu virus to exist at the same time in a pig. Pigs have sugar chains necessary for both the human virus and the avian virus to flourish and whenever both viruses inhabit a pig at the same time there is a chance that the viruses will trade genes resulting in a human form of the H5N1 avian flu (10).<BR><BR>
| |
| | |
| Several countries are in the process of developing human vaccines against the H5N1 virus, which would significantly hinder disease progress. However, typical seasonal flu can change significantly from year to year rendering previous vaccines ineffective, and the H5N1 virus mutates at an even higher rate. Thus by the time a vaccine is implemented the virus may have changed sufficiently to render the vaccine less than fully effective (8). This makes H5N1 especially dangerous as there is currently no technology capable of keeping up with its natural evolution.<BR><BR>
| |
|
| |
|
| According to the WHO, the risk of pandemic H5N1 is serious (8). Two of the three criteria for a pandemic as described by the WHO have been met: H5N1 is a new virus subtype and it causes serious human illness. The last criterion, that it can spread easily and sustainably from person-to-person, has not yet occurred. If it does, another pandemic, even more deadly than the “Spanish flu” pandemic, could occur.
| |
| <H4>References</H4> | | <H4>References</H4> |
| | List all references here. Here are some example formats.<br><br> |
| 1. “Evolution of H5N1 Avian Influenza Viruses in Asia.” http://www.cdc.gov/ncidod/EID/vol11no10/05-0644.htm<BR> | | 1. “Evolution of H5N1 Avian Influenza Viruses in Asia.” http://www.cdc.gov/ncidod/EID/vol11no10/05-0644.htm<BR> |
| 2. “Cumulative Number of Confirmed Human Cases of Avian Influenza A/(H5N1) Reported to WHO,” http://www.who.int/csr/disease/avian_influenza/country/cases_table_2009_04_08/en/index.html<BR> | | 2. “Cumulative Number of Confirmed Human Cases of Avian Influenza A/(H5N1) Reported to WHO,” http://www.who.int/csr/disease/avian_influenza/country/cases_table_2009_04_08/en/index.html<BR> |
| 3. Taubenberger, JK., and D.M. Morens “1918 Influenza: The Mother of All Pandemics.” The Center for Disease Control. 2005. Emerging Infectious Diseases. 8 April 2009. <http://www.cdc.gov/ncidod/eid/vol12no01/05-0979.htm#cit>.<BR> | | 3. Taubenberger, JK., and D.M. Morens “1918 Influenza: The Mother of All Pandemics.” The Center for Disease Control. 2005. Emerging Infectious Diseases. 8 April 2009. <http://www.cdc.gov/ncidod/eid/vol12no01/05-0979.htm#cit>.<BR> |