Link Between Microbes and Obesity: Difference between revisions
No edit summary |
No edit summary |
||
| Line 29: | Line 29: | ||
<br>The link between body weight and the gut microbiota was discovered in 2006 by researchers at the Washington University School of Medicine St. Louis [18]. Researches looked at the link between the body weight and the proportion of bacteria that are dominant in the gut. Bacteroides and Firmicutes are the two most dominant groups of bacteria present in the gut. Obese people were more likely to have a smaller proportion of Bacteroides than Firmicutes within the gut microbiota while this ratio of Bacteroides to Firmicutes increases as fat mass decreases, which was confirmed by two groups of people who were placed on two different types of low-calorie diets [18]. Obese people had higher levels of <i>Firmicutes</i> bacteria present in their gut microbiota but they also had more genes present in the gut that were able to breakdown indigestible foods which suggests that these bacteria can absorb more calories from their food. Therefore, a change in the bacterial composition within the gut microbiota will affect weight. This was the first clear correlation between how the gut microbiota determines body weight [10]. | <br>The link between body weight and the gut microbiota was discovered in 2006 by researchers at the Washington University School of Medicine St. Louis [18]. Researches looked at the link between the body weight and the proportion of bacteria that are dominant in the gut. Bacteroides and Firmicutes are the two most dominant groups of bacteria present in the gut. Obese people were more likely to have a smaller proportion of Bacteroides than Firmicutes within the gut microbiota while this ratio of Bacteroides to Firmicutes increases as fat mass decreases, which was confirmed by two groups of people who were placed on two different types of low-calorie diets [18]. Obese people had higher levels of <i>Firmicutes</i> bacteria present in their gut microbiota but they also had more genes present in the gut that were able to breakdown indigestible foods which suggests that these bacteria can absorb more calories from their food. Therefore, a change in the bacterial composition within the gut microbiota will affect weight. This was the first clear correlation between how the gut microbiota determines body weight [10]. | ||
[[Image: Results.jpeg |thumb|2000px| Figure 4. This data shows the impact of removing the protein (TLR₅), which is a protein that affects percentage of gut bugs in a mouse's system, on the body mass of the mice. Source: http://people.bu.edu/sobieraj/articles/MetabolicSyndrome_AlteredGutFlora.9Apr10.pdf]] | [[Image: Results.jpeg |thumb|2000px| Figure 4. This data shows the impact of removing the protein (TLR₅), which is a protein that affects percentage of gut bugs in a mouse's system, on the body mass of the mice. Source: http://people.bu.edu/sobieraj/articles/MetabolicSyndrome_AlteredGutFlora.9Apr10.pdf]] | ||
Current research led by Andrew Gewirtz at Emory University indicate that the gut microbiota plays an important role in how your body stores the food you consume. They designed an experiment using lab mice to look at the relation between weight and gut bugs [17]. Lab mice lacking protein, toll-like receptor 5, had a much higher percentage of the gut bugs in their system and were about 20% heavier [Figure 4] [20]. Results from magnetic resonance imaging indicated that the mice lacking the protein had a increase in fat mass throughout the body, and especially in visceral fat. Mice lacking the toll-like receptor 5 had epididymal fat pads that were double the size of those found in the mice with the protein present [20]. Data also indicated that the increase in fat mass found in mice without TLR₅ led to higher serum levels of triglycerides and cholesterol and also led to an increase in blood pressure [Figure 4, E to G]. Mice lacking the toll-like receptor 5 protein had higher levels of inflammation, which was one of the main explanations for the extra weight. High levels of inflammation causes insulin resistance which makes the mice eat more food. Lab mice were bred to lack a protein known as TLR₅, toll-like receptor 5. This protein sprouts on the surface of most intestinal cells. TLR₅ recognizes and binds to the whiplike flagella that bacteria use to move around; therefore, it controls the mass of pathogens living in the intestine [17]. This protein sprouts on the surface of most intestinal cells. Without this protein, the normally harmless bacteria are able to multipy and expand in number. An increase in the number of these bacteria leads to an inflammatory state; the body tries to respond this increased number of bacteria while also making cells less sensitive to insulin. Intestinal cells need to balance between responding to inflammatory factors and insulin. Therefore, if a cell is busy responding to inflammatory factors triggered by the increase in bacterial numbers, these intestinal cells are more likely to forget to take up glucose. This desensitization to insulin and glucose results in the emergence of symptoms of metabolic syndrome, which include weight gain, high cholesterol, and elevated blood pressure [20]. To reiterate, the bacteria present are causing inflammation which can lead to insulin resistance which in turn makes the mice eat more. This study on mice shows the important of applying this concept to humans. Studies on humans can indicate that the tendency to eat is driven by the change in bacteria in intestines; people consume more since their appetite increase to the the low grade inflammation produced. Future studies like this will reveal how pathogens like bacteria can cause disease. <br> | Current research led by Andrew Gewirtz at Emory University indicate that the gut microbiota plays an important role in how your body stores the food you consume. They designed an experiment using lab mice to look at the relation between weight and gut bugs [17]. Lab mice lacking protein, toll-like receptor 5, had a much higher percentage of the gut bugs in their system and were about 20% heavier [Figure 4, A] [20]. Results from magnetic resonance imaging indicated that the mice lacking the protein had a increase in fat mass throughout the body, and especially in visceral fat [Figure 4, C]. Mice lacking the toll-like receptor 5 had epididymal fat pads that were double the size of those found in the mice with the protein present [20]. Data also indicated that the increase in fat mass found in mice without TLR₅ led to higher serum levels of triglycerides and cholesterol and also led to an increase in blood pressure [Figure 4, E to G]. Mice lacking the toll-like receptor 5 protein had higher levels of inflammation, which was one of the main explanations for the extra weight. High levels of inflammation causes insulin resistance which makes the mice eat more food. Lab mice were bred to lack a protein known as TLR₅, toll-like receptor 5. This protein sprouts on the surface of most intestinal cells. TLR₅ recognizes and binds to the whiplike flagella that bacteria use to move around; therefore, it controls the mass of pathogens living in the intestine [17]. This protein sprouts on the surface of most intestinal cells. Without this protein, the normally harmless bacteria are able to multipy and expand in number. An increase in the number of these bacteria leads to an inflammatory state; the body tries to respond this increased number of bacteria while also making cells less sensitive to insulin. Intestinal cells need to balance between responding to inflammatory factors and insulin. Therefore, if a cell is busy responding to inflammatory factors triggered by the increase in bacterial numbers, these intestinal cells are more likely to forget to take up glucose. This desensitization to insulin and glucose results in the emergence of symptoms of metabolic syndrome, which include weight gain, high cholesterol, and elevated blood pressure [20]. To reiterate, the bacteria present are causing inflammation which can lead to insulin resistance which in turn makes the mice eat more. This study on mice shows the important of applying this concept to humans. Studies on humans can indicate that the tendency to eat is driven by the change in bacteria in intestines; people consume more since their appetite increase to the the low grade inflammation produced. Future studies like this will reveal how pathogens like bacteria can cause disease. <br> | ||
===Gases emitted from breath indicate a link between obesity and gut microbiota=== | ===Gases emitted from breath indicate a link between obesity and gut microbiota=== | ||
Revision as of 02:59, 10 May 2013
Introduction
The human body is host to millions of bacterial cells that have both beneficial or negative impacts on health and the immune system. The gut microbiota is home to over 35,000 bacterial species but there is believed to be an estimated 3.3 million genetic strains of gut bacteria. However, many have not been cultured or defined. The human gut microbiota contains mostly strict anaerobes but is also host to archaea, viruses, and eukaryotes. The most predominant phyla of bacteria include Bacteroides and Firmicutes.
Current Research shows a distinct correlation between the composition of the git microbiota, metabolism, and body weight. Some explanations suggest that some bacteria are able to absorb more calories from other foods. This absorption of extra calories is then taken up by the body and results in excess fat. Another possible explanation is that some bacteria are responsible for helping to maintain metabolic homeostasis which in turn can influence weight loss. The main take-away point from current research in this field is that these gut microbiota play an important role in health and immunity and there are many different causes and effects of the bacterial composition within the human gut.
Human Gastrointestinal Tract

The human gastrointestinal tract, known as the GI tract, is composed of the stomach and the intestine. The upper gastrointestinal tract is composed of the esophagus, stomach, and duodenum while the lower gastrointestinal tract includes most of the small intestine and all of the large intestine [Figure 1]. The gastrointestinal tract works like a chemostat in the sense that new nutrients are always entering into the system from the throat while equivalent amounts of bacteria are exiting in the form of fecal waste. The GI tract is always releasing hormones to help regulate this digestive process. The human gut is an unusual organ because it is open to the outside environment. The gut flora plays a vital role in helping food pass through the system and for deriving important nutrients from the food [14].
Gut Microbiota
The role of the gut microbiome is the focus of renewed interest in health and disease [19]. There has been an increasing number of scientific articles published on intestinal microbiota each year over the past several decades. Most studies were devoted to understanding the impact in the intestinal environment, however, new studies are looking at the effect of commensal microbes on the mammalian gut. The human gut microbiota is composed of microorganisms that live in the digestive tract and is considered the largest supply of the human flora. The composition of the human gut microbial community is host specific. The microbial community is continually evolving due to exogenous and endogenous modifications. The microbiota can be at the center of causing many diseases which can have serious effects on all organ systems in the body. The compositional make-up of the microorganisms in the human gut is an important determinant of maintaining homeostasis or not. There are many contributing factors to the composition of the gut microbiota. Gut microbial composition can depend on host genetics, mutations, and diet based on studies of obesity [4].
90% of the cells in the body are living, bacteria alive in the human gut, and 10% are of the bodies own cells [16]. The body does not come pre-equipped with these essential gut bacteria but are acquired at birth. At birth, bacteria is transferred from mother to child. During the first year of life, the gut microbiota varies from human to human. This variation in the gut microbial ecosystem can occur due to many various factors such as how an individual is fed, interactions with other humans, and medications given. However, after one year of age, the gut microbiota stabilizes and colonizes[19].

The presence of the gut microbial ecosystem is essential within the human body. The gut flora is responsible for maintaining a healthy digestive system, providing nutrients, protecting the body from invasive pathogens, such as Clostridium difficile, Salmonella, and Shigella, and in maintaining a healthy immune system [Figure 2.] [6].
The human body serves as beneficial host for many microorganisms including: bacteria, archaea, viruses, and unicellular eukaryotes. The microbiota refers to the group of microorganisms living in peaceful coexistence within their host. The human microbiota holds as many as 10¹⁴ bacterial cells. The human gut, a very large organ, has the surface size of approximately 200 m². Therefore, the human gut is an important surface for microbial colonization, along with GIT which is rich in microbial nutrients.
The gut microbiota is mostly composed of strict anaerobes; 99% of the bacteria in the gut are anaerobes. Two bacterial phyla, Bacteroidetes and Firmicutes, are the most predominant in the human gut. The following bacterial phyla: proteobacteria, verrucomicrobia, actinobacteria, fusobacteria, and cyanobacteria also have a smaller presence in the gut microbiota. There are an estimated 3.3 million different genetic strains of gut bacteria, but very few have been cultured and identified. The human gut microbiota contains approximately 500 to 1,000 species and the collective human gut microbiota is composed of over 35,000 bacterial species [1]. The intestinal microbiota is not homogeneous; the number of bacterial cells increases as it moves through the GI tract. 10¹ to 10³ bacteria per gram are present in the stomach and duodenum. The number of bacteria per gram increases to 10¹² in the colon, which is the final location of the GI tract. Different bacterial groups thrive at each different site in the intestine. For example, Bacteroides are found only in feces whereas Clostridium, Lactobacillus, and Enterococcus were found only in the mucus layer of the small intestine [19].
Human gut-microbe populations fall into three distinct categories [9]. The three categories are named after the dominant genus: Bacteroides, Prevotella, and Ruminococcus. Bacteroides are known for being able to break down carbohydrates. Bacteroides bacteria are an important species in the human gut because they provide nutrients and inhibit pathogens. Bacteroides bacteria stain gram negative, obligate anaerobes, rod shaped, and genetically distant from proteobacteria. The Bacteroides bacteria can have positive impacts on human health because they break down potential toxins found in plant food, ferment products account for 15% of the caloric value obtained from food, and their metabolism aids human digestion. Negative impacts that Bacteroides have on humans include abscesses forming due to these Bacteroides bacteria escaping from the colon and invading surrounding tissues and organs [21]. Prebotella are known for degrading the slimy mucus in the gut and will therefore lead to increased gut pain. Ruminococcus is commonly associated with weight gain because it helps cells absorb sugar. There are many possible explanations for the gut-microbe population only falling into three categories. These possible explanations are that the gut-microbe composition is determined by blood type, by metabolism, or that the first microbes that a baby is exposed to as the immune system develops determines the composition. The revelation of three types of gut-microbe population can help identify the causes of obesity and inflammatory bowel disease. The identification of a person’s gut type makes it possible for a person to determine what they can eat to stay slim and how well they metabolize.
Obesity

Obesity is the leading preventable cause of death worldwide. Obesity is a medical condition that affects 35.9% of the American population 20 years and older [7]. Excess body fat accumulates and leads to adverse effects on health which can lead to a reduced life expectancy and increase health problems. An person is considered obese when their BMI, body mass index, exceeds 30 kg/m²[5]. Body mass index is determined by dividing a person’s weight in kilograms by the person’s height squared [Figure 3]. Obesity increases the likelihood of the following diseases: heart disease, type 2 diabetes, obstructive sleep apnea, and osteoarthritis. Obesity occurs when a person consumes more calories than he or she burns [15]. Therefore, the most common cause is a person eating too much and exercising too little. Other factors that play a role in obesity are: age, gender, genetics, environmental factors, physical activity, psychological factors, illness, and medication. Treatment options for obesity include dieting, physical exercise, and gastric bypass surgery [15]. It is important to understand the mechanisms behind obesity and look to the correlation between the gut microbiota and obesity. Obese individuals tend to have a makeup of pathogens in their intestine that is different from that of people who are of normal weight.
Current Research
Obesity and Gut Microbiota Composition
The link between body weight and the gut microbiota was discovered in 2006 by researchers at the Washington University School of Medicine St. Louis [18]. Researches looked at the link between the body weight and the proportion of bacteria that are dominant in the gut. Bacteroides and Firmicutes are the two most dominant groups of bacteria present in the gut. Obese people were more likely to have a smaller proportion of Bacteroides than Firmicutes within the gut microbiota while this ratio of Bacteroides to Firmicutes increases as fat mass decreases, which was confirmed by two groups of people who were placed on two different types of low-calorie diets [18]. Obese people had higher levels of Firmicutes bacteria present in their gut microbiota but they also had more genes present in the gut that were able to breakdown indigestible foods which suggests that these bacteria can absorb more calories from their food. Therefore, a change in the bacterial composition within the gut microbiota will affect weight. This was the first clear correlation between how the gut microbiota determines body weight [10].
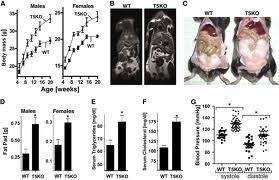
Current research led by Andrew Gewirtz at Emory University indicate that the gut microbiota plays an important role in how your body stores the food you consume. They designed an experiment using lab mice to look at the relation between weight and gut bugs [17]. Lab mice lacking protein, toll-like receptor 5, had a much higher percentage of the gut bugs in their system and were about 20% heavier [Figure 4, A] [20]. Results from magnetic resonance imaging indicated that the mice lacking the protein had a increase in fat mass throughout the body, and especially in visceral fat [Figure 4, C]. Mice lacking the toll-like receptor 5 had epididymal fat pads that were double the size of those found in the mice with the protein present [20]. Data also indicated that the increase in fat mass found in mice without TLR₅ led to higher serum levels of triglycerides and cholesterol and also led to an increase in blood pressure [Figure 4, E to G]. Mice lacking the toll-like receptor 5 protein had higher levels of inflammation, which was one of the main explanations for the extra weight. High levels of inflammation causes insulin resistance which makes the mice eat more food. Lab mice were bred to lack a protein known as TLR₅, toll-like receptor 5. This protein sprouts on the surface of most intestinal cells. TLR₅ recognizes and binds to the whiplike flagella that bacteria use to move around; therefore, it controls the mass of pathogens living in the intestine [17]. This protein sprouts on the surface of most intestinal cells. Without this protein, the normally harmless bacteria are able to multipy and expand in number. An increase in the number of these bacteria leads to an inflammatory state; the body tries to respond this increased number of bacteria while also making cells less sensitive to insulin. Intestinal cells need to balance between responding to inflammatory factors and insulin. Therefore, if a cell is busy responding to inflammatory factors triggered by the increase in bacterial numbers, these intestinal cells are more likely to forget to take up glucose. This desensitization to insulin and glucose results in the emergence of symptoms of metabolic syndrome, which include weight gain, high cholesterol, and elevated blood pressure [20]. To reiterate, the bacteria present are causing inflammation which can lead to insulin resistance which in turn makes the mice eat more. This study on mice shows the important of applying this concept to humans. Studies on humans can indicate that the tendency to eat is driven by the change in bacteria in intestines; people consume more since their appetite increase to the the low grade inflammation produced. Future studies like this will reveal how pathogens like bacteria can cause disease.
Gases emitted from breath indicate a link between obesity and gut microbiota
Another study also found a link between gut bacteria and obesity using breath test of emitted gases from the body. Researchers at Cedars-Sinai Medical Center indicate that people with high levels of both hydrogen and methane in their breath are more likely to have a higher BMI and higher proportion of body fat [13]. This was the first large-scale study to look at the correlation between gas production and body weight. Tests done on 792 patients indicated that there were four main patterns: normal breath, breath containing higher levels of methane, higher levels of hydrogen, or higher levels of both gases. Patients with higher levels of both methane and hydrogen had a greater likelihood of having a higher BMI and higher proportion of body fat. The gut bacterium, Methanobrevibacter smithii, is responsible for most of the methane produced in the human gut. M. smithii produces methane by scavenging hydrogen from other microorganisms. Abnormally high levels of M. smithii can alter the energy balance leading people to put on more weight. This study creates a direct link between gut bacteria and weight gain by looking at gas production created by gut bacteria [13].
Treatment for obesity using gut bacteria
Researchers have started to look out how bacteria can be used to treat obesity without surgery [2]. Studies on mice have shown that gastric bypass surgery can lead to different types of bacteria to colonize in the gut. Transfer of these colonized bacteria present after surgery into healthy mice led to these healthy mice to rapidly lose weight without surgery. This study highlights the fact that bacteria microbiota is much different in the stomach and intestines of obese people compared with those of people of normal weight. If gastric bypass surgery occurs, the gut microbiota will change [12]. A study was designed by looking at three groups of obese mice on high calorie diets: 1) group of mice that underwent gastric bypass surgery, 2) group of mice that underwent a fake surgery and the higher calorie diet continued, and 3) group of mice that was give same fake operation but then fed low calorie diet to promote weight loss. The results indicate that only the group that underwent gastric bypass surgery had any change in the micro-organisms present in the stomach and intestines. These microbiota were then transferred to germ-free mice. The group that received the bacteria from bypass mice had the most significant amount of weight loss while the other two groups had no change in weight [Figure 5].The bacteria that are responsible for the weight loss perform two beneficial services: 1) these bacteria increase metabolism, making it easier for people to burn off calories more easily than people with lower concentrations of these bacteria, and 2) these bacteria absorb fewer calories while the bacteria associated with weight gain absorb as many calories as possible. This study shows how certain gut bacteria populations can influence weight loss [8]. Future studies will begin to look at the mechanisms by which a microbial population changed due to gastric bypass and its effects on environment.
Conclusion
The human gut is home to a large microbiota of bacteria, viruses, and unicellular eukaryotes. The human gut microbiota is a very complex and important system that affects human health. New studies in microbiology have shown the link between obesity and microorganisms living in the gut. Research has shown how certain bacteria are responsible for an increase in weight. These studies are revolutionary because they show the importance of genetic and biotic factors that might be responsible for changes in weight. This new field can lead to new treatments on how to alter the gut microbiota to manage changes in weight. Although these studies present a clear correlation between the gut microbiota composition and body weight, it is still imperative that each individual continues to maintain their body weight through diet and exercise as a preventative measure for obesity.
References
1.Arumugam, M. et al. (2011)Nature Advance online publication doi:10.1038/nature09944
2.BBC News. Bacteria find 'key to treating obesity without surgery. 28 March 2013 Last updated at 06:49 ET
3.Begley, Sharon. After weight-loss surgery, new gut bacteria keep obesity away. Reuters. New York. Wed Mar 27, 2013 2:01pm EDT
4.Benson A et al. (2010) Individuality in gut microbiota composition is a complex polygenic trait shaped by multiple environmental and host genetic factors. PNAS doi:10.1073/pnas.1007028107
5.B4 Tea. How to Calculate your body mass index (BMI)? Online. Retrieved April 24, 2013.
6.Chierico et al. (2012)Early-life gut microbiota under physiological and pathological conditions: The central role of combined meta-omics-based approaches. Journal of Proteomics Volume 75, Issue 15, 3 August 2012, Pages 4580–4587
7.CDC. Obesity and Overweight. 2013.
8.Grady, Denise. Bacteria in the Intestines May Help Tip the Bathroom Scale, Studies Show. The New York Times, Updated March 27, 2013
9.Jones, Nicola. (2011) Gut study divides people into three types. Published online 20 April 2011 | Nature | doi:10.1038/news.2011.249
10.Krznarić Z, Vranešić Bender D, Kunović A, Kekez D, Stimac D. (2012) Gut Microbiota and Obesity. Dig Dis 30(2):196-200. doi: 10.1159/000336965
11.Ley RE, Turnbaugh PJ, Klein S, Gordon JI. (2006) Microbial Ecology: Human gut microbes associated with obesity. Nature. 2006 Dec 21;444(7122):1022-3
12.Liou A, Paziuk M, Luevano J, Machineni S, Turnbaugh P, Kaplan L. (2013) Conserved Shifts in the Gut Microbiota Due to Gastric Bypass Reduce Host Weight and Adiposity. Sci Transl Med Vol. 5, Issue 178, p. 178ra41
13.Mather R, Amichai M, Chua KS, Mirocha J, Barlow GM, Pimentel M. (2013) Methane and Hydrogen Positivity on Breath Test Is Associated With Greater Body Mass Index and Body Fat. The Journal of Clinical Endocrinology & Metabolism March 26, 2013 jc.2012-3144
14.Mayo Clinic. Gastrointestinal Tract. Updated May 2013
15.Mayo Clinic. Obesity. Updated May 2013
16.Nograndy, Bianca. 2013. Gut bugs - the bacteria you need. ABC Health and Wellness.
17.Park, Alice. A Hidden Trigger of Obesity: Intestinal Bugs. Time: Health and Family. Friday, Mar. 05, 2010
18.Pearson, Helen. 2006. Fat people harbour 'fat' microbes. Nature | doi:10.1038/news061218-6
19.Sekirov I, Russell S, Caetano L, Antunes M, Finlay B. (2010) Gut Microbiota in Health and Disease. doi: 10. 1152/ physrev. 00045. 2009 Physiol Rev July 1, 2010 vol. 90no. 3 859-904
20. Vijay-Kumar M. et al. (2010) Metabolic syndrome and altered gut microbiota in mice lacking toll-like Receptor 5. Science 328, 228
21.Wexler,Hannah. 2007. Bacteroides: the Good, the Bad, and the Nitty-Gritty. Clinical Microbiology Rev. 10.1128/CMR.00008-07
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2013, Kenyon College.
