Chromohalobacter Salexigens
A Microbial Biorealm page on the genus Chromohalobacter Salexigens
Classification
Higher order taxa
Bacteria; Proteobacteria; Gammaproteobacteria; Oceanospirillales; Halomonadaceae; Chromohalobacter;
Species
C. Salexigens
Description and significance
This bacterium is a moderate halophile, yet does not require high concentrations of sodium chloride. The salt requirements of this organism can be met by ions of other salts, such as potassium, rubidium, ammonium, bromide, and others.
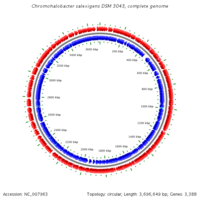
Genome structure
DNA Bases: 3696649
Chromosome Type: Circular
Total Genes: 3403
Protein Coding Genes: 3319 RNA Genes: 84 Pseudo Genes: 21
Cell structure and metabolism
The growth rate of Chromohalobacter salexigens DSM 3043 can be stimulated in media containing 0.3 M NaCl by a 0.7 M concentration of other salts of Na+, K+, Rb+, or NH4+, Cl-, Br-, NO3-, or SO4(2-) ions.
Ecology
Describe any interactions with other organisms (included eukaryotes), contributions to the environment, effect on environment, etc.
Pathology
C. Salexigens is not known to be pathogenic
Application to Biotechnology
The halophilic bacterium Chromohalobacter salexigens synthesizes and accumulates compatible solutes in response to salt and temperature stress. The ectD gene, which is involved in the synthesis of the compatible solute hydroxyectoine, is essential for thermoprotection of the halophilic bacterium Chromohalobacter salexigens.
Current Research
N(gamma)-acetyl-2,4-diaminobutyrate (NADA), the precursor of the compatible solute ectoine, was shown to function as an osmoprotectant for the non-halophilic bacterium Salmonella enterica serovar Typhimurium. The addition of NADA-containing extracts of an ectoine synthase mutant of the broad salt-growing halophile Chromohalobacter salexigens DSM 3043(T) could alleviate the inhibitory effects of high salinity in S. enterica, which lacks the ectoine biosynthetic pathway. NADA, purified from extracts of the mutant, protected S. enterica against salinity stress.
In another study, the long-term response of the broad-salt growing halophile Chromohalobacter salexigens DSM 3043T to salt stress has been investigated with respect to adaptive changes in membrane lipid composition. This study included the wild-type and three salt-sensitive, ectoine-deficient strains: CHR62 (ectA::Tn1732, unable to grow above 0.75 M NaCl), CHR63 (ectC::Tn1732, unable to grow above 1.5 M NaCl), and CHR64, which was able to grow in minimal medium M63 up to 2.5 M NaCl, but its growth was slower than the wild-type strain at salinities above 1.5 M NaCl. This mutant accumulated ectoine and hydroxyectoine as major compatible solutes, but also the ectoine precursor, N-gamma-acetyldiaminobutyric acid, and was found to be affected in the ectoine synthase gene ectC. The main phospholipids of the wild-type strain were phosphatidylethanolamine, phosphatidylglycerol (PG), and cardiolipin (CL). Major fatty acids were detected as 16:0, 18:1, and 16:1, including significant amounts of cyc-19:0, and cyc-17:0. CL and cyclopropane fatty acids (CFA) levels were elevated when the wild-type strain was grown at high salinity (2.5 M NaCl). Membranes of the most salt-sensitive trains CHR62 and CHR63, but not of the less salt-sensitive strain CHR64, contained lower levels of CL. The proportion of cyc-19:0 in CHR64 was three-fold (at 2.0M NaCl) and 2.5-fold (at 2.5 M NaCl) lower than that of the wild type, suggesting that this mutant has a limited capacity to incorporate CFA into phospholipids at high salt. The addition of 1 mM ectoine to cultures of the wild-type strain increased the ratio PG/CL from 1.8 to 3.3 at 0.75 M NaCl, and from 1 to 6.5 at 2.5 M NaCl, and led to a slight decrease in CFA content. Addition of 1 mM ectoine to the mutants restored the steady-state levels of CL and CFA found in the wild-type strain supplemented with ectoine. These findings suggest that exogenous ectoine might attenuate the osmostress response involving changes in membrane lipids.
References
Edited by Chris Wittrock, a student of Rachel Larsen and Kit Pogliano
