Yersinia Pestis (Pathogenesis)

Etiology/Bacteriology
Taxonomy
| Domain = Bacteria | Phylum = Proteobacteria | Class = Gammaproteobacteria | Order = Enterobacteriales | Family = Enterobacteriaceae | Genus = Yersinia | species = Yersinia pestis
Description
Yersinia pestis is a zoonotic pathogen that is most commonly transmitted through fleas on rodents. Y. pestis is a Gram-negative rod, facultative anaerobe that is non-motile and non-spore-forming. In the past, this pathogen ravaged through cities located throughout Europe, Asia, and Africa. Sudden outbreaks of infection would happen, and take thousands of innocent lives. Without proper antibiotic treatment, infection by Y. pestis would usually lead to death within a few days. Humans are usually infected through the bite of an infected flea or by inhalation of bacteria-infested droplets. The infection exists in three major plague forms: bubonic, septic, and pneumonic. The bubonic plague is the most common form of infection and targets the victim’s lymphatic system. After being taken up by macrophages, the bacteria proliferate in the affected lymph node, causing it to be inflamed and swollen. For the septic plague, it affects the bloodstream, and could be a consequent of the infected lymph nodes. Victims are usually covered with black patches due to hemorrhages throughout the skin, leading to its “Black Death” nickname. Occasionally, this leads the infection of the lungs, which would result in the pneumonic plague. More than often, victims do not survive infection by the pneumonic plague. In today's society, infection usually occurs in summer when the chances of being bit by a flea are higher in warmer weather.[1]
Pathogenesis
Transmission
Fleas
Y. pestis is most commonly transmitted to humans through the bites of infected fleas, resulting in either primary bubonic plague or septicemic plague [2]. In the midgut of its principal flea vector (Xenopsylla cheopis), Y. pestis survives cytotoxic digestion of blood plasma through the action of Yersinia murine toxin (Ymt) (2). Ymt is a plasmid-encoded phospholipase D (PLD) [3]. Through the action of this PLD, Y. pestis is able to colonize the flea midgut, which may have caused the bacterium to obligately transmit to arthropods [3]. The hemin storage system locus (hms) also contributes to the pathogenicity of Y. pestis in fleas (3). Y. pestis hms encodes for a storage system that synthesizes extracellular saccharides in order to facilitate colonization of the proventriculus in the foregut (3). The formation of biofilms in the proventriculus contributes to the transmission of the plague in fleas [4].
Fluid/Tissue
Y. pestis can be transmitted to humans through the handling of fluids or tissue from infected animals[2]. Once Y. pestis has entered the human host, the bacterium spreads throughout the lymphatic system and enters the bloodstream within 2-6 days [5]. The spread of Y. pestis throughout the lymphatic system triggers a large-scale immune response with the appearance of buboes on the armpits, groin, and neck [6]. Increased numbers of bacteria in the bloodstream promotes the odds of human-human transmission (6). Contact with contaminated fluid or tissue typically results in bubonic plague or septicemic plague [2].
Infectious Droplets
Y. pestis can also be transmitted through the air via infectious droplets from coughing[2]. Transmission of infectious droplets is the only method of spreading the plague from person to person[2]. Transmission of infectious droplets through coughing enables Y. pestis to colonize the lungs[6]. This type of infection is called “pneumonic plague” and has a mortality rate close to 100 percent [2], [6].
Infectious dose, incubation, colonization
In humans, the infectious dose of Y. pestis has been estimated to range from 100 organisms to 20,000 organisms[8]. The incubation period of the bubonic, septicemic, and pneumonic plague types ranges from 2-6 days[5]. Y. pestis colonizes lymph nodes of the host and proliferates[9]. Left untreated, Y. pestis is able to spread to the bloodstream and cause secondary infection as well as septicemic plague, in rare cases[10]. Y. pestis is also able to colonize lung tissue as pneumonic plague and proliferate[2].
Epidemiology
United States
In the United States, natural Y. pestis loci exist in primarily rural and uninhabited areas[11]. From 1994 to 1999, 49 reported cases of plague occurred in Arizona, California, Colorado, New Mexico, and Utah, resulting in three deaths[12]. In 2006, California, Colorado, New Mexico, and Texas reported 13 cases of plague and two deaths[13]. 10-15 cases of plague on average are reported each year in the United States[11].
Worldwide
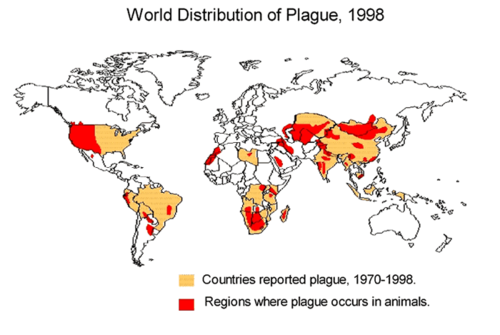
Internationally, cases of plague are typically reported from developing countries in Africa and Asia [11]. From 1990-1995, 12,998 cases worldwide were reported to the World Health Organization (WHO). Countries that had the highest incidences of plague were India, Zaire (now the Democratic Republic of the Congo), Peru, Malawi, and Mozambique [11]. In 2003, 2118 cases were reported with 182 deaths worldwide[11]. 98.7 percent of cases and 98.9 percent of deaths were reported from Africa (10). Australia is the only continent that has never reported a case of plague [11].
Virulence factors
Adhesion/invasion
The plasminogen activator (Pla) protein of Y. pestis facilitates the adhesion and invasion of the bacterium to the extracellular matrix of host tissues [18]. Pla induces the activation of plasminogen into plasmin, which causes proteolysis and damage to host tissues (18). In addition, Pla contributes to Y. pestis’s ability to invade epithelial cells [19].
Nutrient acquisition
Yersiniabactin (Ybt) is a siderophore that acquires iron produced by the host[20]. Ybt is secreted by Y. pestis to scavenge iron in the host, and once it binds iron, it returns to the bacterial cell[20]. Iron acquisition is a crucial component of initial colonization via the tissues, as Ybt-defective strains cannot spread from the initial site of infection[20].
Type III secretion system
Y. pestis utilizes a type III secretion system (T3SS) in order to evade host responses. The plasmid of Yersinia virulence (pYV) encodes the following components of the T3SS [14]. Yersinia secretion (Ysc) proteins construct the secretion organelles [15]. Effector Yersinia outer proteins (Yops) interfere with host cell signaling[15]. Translocators deliver the effectors across the host cell membrane [15]. Chaperones assist with transportation of the effectors and translocators, and regulatory components regulate the system [15].
Yop effectors
The Yop effectors YopH, YopE, YpkA, and YopM contribute to the virulence of Y. pestis while YopJ’s role is debated[16]. YopH disrupts focal complexes and and inhibits pro-inflammatory signaling to help Y. pestis avoid host phagocytosis and inflammation [17]. YopE and YpkA disrupt actin microfilaments so that Y. pestis may avoid phagocytosis [17]. YopM migrates to the nucleus and interferes with the eukaryotic cell cycle [17]. YopJ inhibits MAPK and NF-κB signaling pathways, inducing apoptosis of macrophages and contributing to anti-inflammation[17].
Anti-phagocytic antigens
The anti-phagocytic antigens Factor 1 (F1) and V-antigen (LcrV) also contribute to the virulence of Y. pestis. The bacterium exports F1, and it is assembled into a capsule-like structure[21]. This structure increases Y. pestis resistance to phagocytosis by macrophages[21]. LcrV also increases resistance to phagocytosis as well as downregulation of the inflammatory response [22]. LcrV works with the adhesin YadA and the Yop effectors to facilitate this response[21].
Clinical features
Symptoms
The symptoms of Yersinia Pestis presents in different ways, but the three most common are bubonic, septicemic, and pneumonic plague. [23]
Bubonic Plague
This type of plague usually results from the bite of an infected flea. Once infection sets, the patient has sudden onset of fever, headache, chills, weakness, and the development of swollen nodes known as buboes, where isolated bacteria multiply and grow. If not treated the bacteria can spread to other areas [23] .
Septicemic Plague
This type of plague can either develop primarily or as a result from untreated bubonic plague. Symptoms from this include bleeding into the skin and other organs ranging to tissue blackening and death, especially in the fingers, toes, and the nose. [23]
Pneumonic Plague
This type of plague either develops from inhaling infectious droplets or from untreated bubonic/septicemic plague and bacteria spreading to the lungs. At this point the plague is infectious and can be spread from person to person by infectious droplets. Symptoms from this type of plague include fever, headache, weakness, and a developing pneumonia that heightens symptoms of cough, chest pain and shortness of breath. [23]
Morbidity and Mortality
Worldwide, the number of cases reported to the World Health Organization range between 1000 and 2000 per year. However, according to WHO, the real number is likely much higher. Because of this, it is difficult to assess mortality rate, especially in developing countries with poor diagnostics and under reporting. The mortality rate cited by WHO, is between 8-10%, but again, the predicted percentage is expected to be much higher. [24]
Diagnosis
Diagnoses of the plague are usually reported to a state public health lab. The most common sign of the plague is the development of bubo, swollen lymph nodes, after a fleabite. Usually, blood from the patient and parts of the swollen lymph nodes are submitted to a Level A lab for testing. For a culture ID, blood is checked for a positive blood culture, with BACTEC Media and SEPTI-CHEK BHI. Next, it is cultured on TSA w/5% Sheep Blood/MacConkey II Agar, and incubated for 24 hours at 28°C. The colonies should be gray-white, translucent, little to no hemolysis, and be non-lactose fermentor. Afterwards, it should test positive for catalase, but negative for oxidase and Christensen’s Urea Slant.[25]
Treatment
Antibiotics
Individuals suspected of infection from Yesinia pestis should be immediately admitted to hospitals for isolation and proper management. A number of powerful antibiotics are used to treat the illness, with streptomycin usually prescribed as the primary drug of choice. Other possible antibiotics include gentamicin, chloramphenicol, tetracyclines, and fluoroquinolones. [26] The antibiotic levofloxacin has also been recently approved by the Food and Drug Administration as appropriate treatment.[27] Antibiotic dosages are typically administered for the full period of ten days or for three days after the fever has subsided. However, the selection of antibiotic therapy is crucial, as several classes of antibiotics have proven to be ineffective in treatment for the plague. These include penicillins, cephalosporins, and macrolides.[28]
Prophylaxis
Prophylactic therapy is a common mode of treatment for individuals who have been exposed to potentially infected individuals. Patients with possible exposure to Y. pestis should be administered antibiotics if the exposure occurred within the span of six days as a means of preventative therapy.
The usage of prophylactic therapy may also be used as a preventative measure for individuals who must be, for short periods of time, involved in circumstances where the potential of infection may be unavoidable.[28]
Prevention
Yersinia pestis can be transmitted to humans from the bites of inflected fleas or handling of plague-infected tissues. In the case of plague pneumonia, transmission can occur via inhalation of the cough droplets from another infected individual. While no available vaccine currently exists, several effective means of prevention are to diminish the possibility of rodent infestation around homes by clearing away cluttered debris within the vicinity and to apply flea control products for pets that roam freely in the open. The application of insect repellent for individuals in outdoor areas is an effective measure for protection against flea bites. Any contact with potentially infected animals should be limited, and the usage of gloves as a barrier against possible transmission should be utilized when necessary.[26]
Means of prevention can also be applied in hospital settings where the possibility of transmission can be high. Standardized procedures of handwashing and utilization of gowns, latex gloves, and protective devices should be followed to protect all body orifices from coming into contact with Y. pestis. Restrictions of patients suspected with plague should be enacted to prevent the spread of disease to other individuals. This includes isolated treatment of infected patient as well as the inhibition of movement of the patient outside of the isolation room until the infection ceases to exist.[28]
Host Immune Response
The host innate immune response involves macrophages, inflammation, and the activation of the complement cascade. However, Yersinia Pestis has evolved different mechanisms for evading this immune response, both in the innate and adaptive immune response.[29]
Attack on Innate Immune Response
Most of Y. pestis are killed off by encounter with neutrophils and many that survive manage are a special subtype (facultative Y. pestis). Using the macrophages, they are then able to proliferate and express different virulence factors, before the spread systemically throughout the body. In addition, the LPS (lipopolysaccharide) structure in this organism allows the bacteria to become resistant to serum-mediated lysis during its transition from its flea vector to animal host. The bacteria coming from the macrophages, therefore, are resistant to phagocytosis and can inhibit the production of proinflammatory cytokines, which in turn attenuates the adaptive immune response of the host. Yet another immune response that is affected is the complement pathways. The complement cascade of the innate immune response (which is initiated by macrophages binding to foreign antigen) has three different effector functions: opsonization, which leads to phagocytosis, inflammation, and the formation of a membrane-attack complex, which leads to direct killing. However, Y. pestis has developed a resistance to complement-mediated lysis in an effort to survive transmission between flea and animal. In addition, during replication within a macrophage, the bacteria form a needle-like complex that (once released from the macrophage), they use to inject six different effector proteins into different cells to further inhibit the immune response. Targets for this injection include macrophages, dendritic cells, and neutrophils. Besides paralyzing these phagocytic cells, these proteins also target the proinflammatory recruitment response initiated by infected cells. Finally, these proteins also targeting NK cells, which further inhibit the innate immune response. [29]
Attack on Adaptive Immune Response
Because the innate immune response has been so severely affected, the adaptive immune system cannot be properly initiated. Dendritic cells (which are the cells that link between the innate and adaptive immune response) are targeted early on, and as a result the activation of the adaptive immune response is hindered because dendritic cells cannot mature and start T-cell mediated immune response. Because of this, the humoral response (B-cell response) cannot be properly triggered.[29,[30].
Host Immune Response
In order to combat the Y. pestis infection, the host cell must reactivate its specific humoral and cellular response mechanisms to establish a protective immunity. This involves neutralizing virulence factors and delivering antibody/antigen complexes to B cells, macrophages, and dendritic cells (which promotes T-cell activation).[30] Often times, this activation is a result of the very pathology of the organism. Although the bacterium attenuates the inflammatory response and causes apoptosis of naïve macrophages, activated macrophages are killed by a process known as pyroptosis. Interestingly, this process has the opposite effect and activates the previously attenuated inflammatory response, and benefits the host further by accelerating and amplifying this response to combat the bacterial infection and stimulate proper immune response pathways. Hyper-inflammation, however can result in tissue damage and organ malfunction.[30]
References
1. Gross L. How the plague bacillus and its transmission through fleas were discovered: reminiscences from my years at the Pasteur Institute in Paris. Proc Natl Acad Sci USA 1995 15;92(17):7609-11.
2. Centers for Disease Control and Prevention. Plague: Ecology and Transmission. [<http://www.cdc.gov/plague/transmission/>].
3. Hinnebusch BJ, Rudolph AE, Cherepanov P, Dixon, JE, Dixon, JE, Schwan, TG, Forsberg Å. 2002. Role of Yersinia Murine Toxin in survival of Yersinia pestis in the midgut of the flea vector. Science. 296: 733-735.
4. Hinnebusch BJ, Perry RD, Schwan TG. 1996. Role of the Yersinia pestis hemin storage (hms) locus in the transmission of plague by fleas. Science. 273: 367-370.
5. Brubaker RR. 1991. Factors promoting acute and chronic diseases caused by yersiniae. Clin. Microbiol. Rev. 4: 309-324.
6. Smego RA, Frean, J, and Koornhof, HJ. 1999. Yersiniosis I: microbiological and clinicoepidemiological aspects of plague and non-plague Yersinia infections. Eur. J. Clin. Microbiol. Infect. Dis. 18: 1-15.
7. Lorange EA, Race BL, Sebbane F, Hinnebusch BJ. 2005. Poor vector competence of fleas and the evolution of hypervirulence in Yersinia pestis. J. Infect. Dis. 191: 1907-1912.
8. Rose LJ, Donlan R, Banerjee SN, Arduino, MJ. 2003. Survival of Yersinia pestis on environmental surfaces. Appl. Environ. Microbiol. 69: 2166-2171.
9. Inglesby TV, Dennis DT, Henderson DA, Bartlett JG, Ascher MS, Eitzen E, Fine AD, Friedlander AM, Hauer J, Koerner JF, Layton M, McDade J, Osterholm MT, O'Toole T, Parker G, Perl TM, Russell PK, Schoch-Spana M, Tonat K. 2000. Plague as a biological weapon: medical and public health management. JAMA. 283: 2281-2290.
10. Wayangankar S, Bronze MS, Minnaganti VR, Jackson RL. 2013. Plague: Epidemiology. Medscape. [<http://emedicine.medscape.com/article/235627-overview#a0199>]
11. Centers for Disease Control and Prevention. Plague: Maps and Statistics. [<http://www.cdc.gov/plague/maps/>].
12. Human plague--United States, 1993-1994. 1994. MMWR Morb. Mortal Wkly Rep. 43: 242-246.
13. Ben-Gurion R, Shafferman A. 1981. Essential virulence determinants of different Yersinia species are carried on a common plasmid. Plasmid 5: 183-187.
14. Portnoy DA, Wolf-Watz H, Bolin I, Beeder AB, Falkow, S. 1984. Characterization of common virulence plasmids in Yersinia species and their role in the expression of outer membrane proteins. Infect. Immun. 43: 108-114.
15. Bolin I, Wolf-Watz, H. 1988. The plasmid-encoded Yop2b protein of Yersinia pseudotuberculosis is a virulence determinant regulated by calcium and temperature at the level of transcription. Mol. Microbiol. 2: 237-245.
16. Bliska JB. 2000. Yop effectors of Yersinia spp. and actin rearrangements. Trends Microbiol. 8: 205-208.
17. Cowan C, Jones HA, Kaya YH, Perry RD, Straley, SC. 2000. Invasion of epithelial cells by Yersinia pestis: evidence for a Y. pestis-specific invasin. Infect. Immun. 68: 4523-4530.
18. Lahteenmaki K, Virkola R, Saren A, Emody L, Korhonen TK. 1998. Expression of plasminogen activator pla of Yersinia pestis enhances bacterial attachment to the mammalian extracellular matrix. Infect. Immun. 66: 5755-5762.
19. Lesic B, Carniel E. 2004. The high-pathogenicity island: a broad-host-range pathogenicity island, p 285-306. In. Carniel E, Hinnebusch, BJ (eds), Yersinia: molecular and cellular biology. Wymondham: Horizon Bioscience.
20. Perry RD, Fetherston, JD. 2004. Iron and Heme uptake systems, p 257-283. In. Carniel E, Hinnebusch, BJ (eds), Yersinia: molecular and cellular biology. Wymondham: Horizon Bioscience.
21. Du Y, Rosqvist R, Forsberg A. 2002. Role of fraction 1 antigen of Yersinia pestis in
inhibition of phagocytosis. Infect. Immun. 70: 1453-1460.
22. Cornelis GR, Boland A, Boyd AP, Geuijen C, Iriarte M, Neyt C, Sory MP, Stainier I. 1998. The virulence plasmid of Yersinia, an antihost genome. Microbiol. Mol. Biol. Rev. 62: 1315-1352.
23. "Plague-Symptoms" Centers for Disease Control and Prevention. Centers for Disease Control and Prevention, 25 July 2012. Web. 16 July 2014. [<http://www.cdc.gov/plague/symptoms/index.html>]
24. "Plague-frequently asked questions." Centers for Disease Control and Prevention. Centers for Disease Control and Prevention, 25 July 2012. Web. 16 July 2014. [<http://www.cdc.gov/plague/faq/>.]
25. "Learning Center Yersinia pestis." Becton Dickinson and Company. Web. 16 July 2014. [<http://www.bd.com/ds/learningcenter/biodefense/yersiniapestis.asp>.]
26. Centers for Disease Control and Prevention. Plague: Prevention. [<http://www.cdc.gov/plague/prevention/>].
27. Mayo Clinic. Plague: Prevention. [<http://www.mayoclinic.org/diseases-conditions/plague/basics/prevention/con-20021610>]
28. Poland JD, Dennis, DT. Plague Manual: Epidemiology, Distribution, Surveillance and Control. WHO/CDS/CSR/EDC/99.2
29. D'elios, Mario Milco. "Role of immune response in Yersinia pestis infection." The Journal of Infection in Developing Countries: n. pag. Web. 16 July 2014. [<www.jidc.org/index.php/journal/article/download/21918303/599>]
30. "Innate immune response during Yersinia infection: critical modulation of cell death mechanisms through phagocyte activation." Innate immune response during Yersinia infection: critical modulation of cell death mechanisms through phagocyte activation. N.p., 1 Sept. 2009. Web. 16 July 2014. [<http://www.jleukbio.org/content/86/5/1153.full>]
Created by {Krishna Manohar, Michael Grassi, Christina Cheng, Johnson Ong}, students of Tyrrell Conway at the University of Oklahoma.