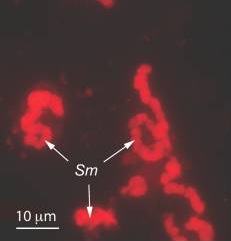
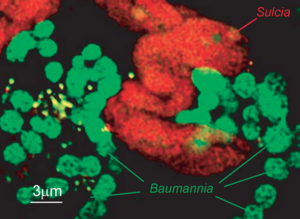
Sulcia muelleri
Classification
Domain: Bacteria; Phylum; Bacteroidetes; Class: Flavobacteria; Order Flavobacteriales
Species
|
NCBI: Taxonomy |
Sulcia muelleri
Description and Significance
Sulcia muelleri is a bacterial obligate intracellular symbiont primarily found in specialized cells in insects of the suborder Auchenorrhyncha (examples of this suborder include Cicadas and Treehoppers). It is gram negative, roughly rod-shaped, with a width ranging from 3-5 microns and a widely variable length, up to 80 microns. They are often observed in a coiled or 'balled up' conformation.[1] As Sulcia is found as a symbiont across many different species in Auchenorrhyncha, it is likely that the symbiosis predates the origin of the suborder approximately 260MYA[1]. Today, it is often a partner in tripartite symbiosises with its sap-feeding insect host and other bacteria, such as Baumannia cicadellinicola (Gammaproteobacteria) or Zinderia Insectola (Betaproteobacteria). It is responsible for amino acid biosynthesis for the insect while the third partner often contributes vitamins and cofactors. These mutualistic interactions have enabled the utilization of otherwise inaccessible niches by Auchenorrhyncha, and thus helped spur their diversification.
Genome Structure
Sulcia has a single circular chromosome around 245 kilobases in length. It is notable for the extreme levels of gene loss that have occurred over the course of its symbiosis with Auchenorrhyncha. It retains only 263 genes, 227 of which are protein-coding. Notably absent are genes for membrane biosynthesis (it depends entirely on host-derived membranes), all but two genes for DNA repair, and even some of the aminoacyl-tRNA synthase genes, raising the question of how protein synthesis is accomplished.
Its genome displays a very low GC content, around 23.5%, which is a common feature of reduced genomes. This is speculated to be due to the universal mutational bias towards AT combined with low selection pressure and a lack of DNA repair machinery.
A disproportionate amount of Sulcia's genome is dedicated to amino acid synthesis, as one would expect based on the nature of its symbiosis.
Cell Structure, Metabolism and Life Cycle
Interesting features of cell structure; how it gains energy; what important molecules it produces.
S. muelleri uses aerobic respiration. Its ATP is synthesized from cytochrome c oxidase catalyzed termination. This bacteria has the genetic components necessary to produce all 20 amino acids, yet no strain actually synthesizes each one. Most strains are capable of synthesizing all 8 essential amino acids, which include leucine, valine, threonine, isoleucine, lysine, arginine, phenylalanine and tryptophan. For example, the strain Sulcia-CARI cannot synthesize tryptophan due to the loss of certain genetic components.These amino acids are essential for the bacteria's host, since sharpshooters do not get these essential amino acids from their xylem-sap diet.
Ecology and Pathogenesis
S. muelleri lives in specialized cells in the host and is involved in an obligate mutualism with its host and certain Gammaproteobacteria. The host primarily feeds on xylem sap which is low in nutrients and contains mostly inorganic components with little amino acids. In this tripartite symbiosis, a metabolic exchange of metabolites is occurring by each of the members. S. muelleri uses aerobic respiration with the electron donor being carbon found from components of the sap. It is responsible for synthesizing most essential amino acids found in the mutualism and in the host.
Although this bacteria isn't known to be the direct cause of any diseases, its host are considered pests and vectors of some plant diseases such as diseases caused by the pathogen Xylella fastidiosa.
Habitat; symbiosis; biogeochemical significance; contributions to environment.
If relevant, how does this organism cause disease? Human, animal, plant hosts? Virulence factors, as well as patient symptoms.
References
1. http://www.pnas.org/content/104/49/19392.full
2. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2953269/
Author
Page authored by _____, student of Prof. Jay Lennon at IndianaUniversity.