Metabolism in Neisseria meningitidis
Introduction
By Molly Folks
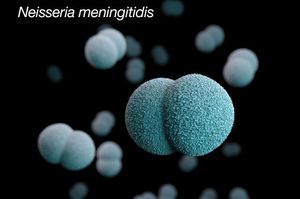
Neisseria meningitidis (N. meningitidis) is Gram-negative bacterium that is a member of the class, Betaproteobacteria. Neisseria are aerobic heterotrophic cocci, and the cocci of Neisseria genus form diplococci, distinctive pairs of cocci (Fig 1) [2]. Most members of the Neisseria genus are commensal organisms that colonize the nasal and oral mucosa of animals. There are 11 species of Neisseria that colonize humans and only 2 are pathogens, Neiserria meningitidis and Neiserria gonorrhoeae.
N. meningitidis was first discovered in 1887 by Anton Weichselbaum after analyzing the cerebrospinal fluid (CSF) of a patient who was infected with meningitis [3]. There are at least 12 known serotypes of N.meningitidis based on the unique capsular polysaccharides seen on the cell surface [3]. Serotypes are groups that are found within a single microorganism, such as viruses and bacteria, that share differentiating surface structures [4]. Serotypes A, B, C, W, X, and Y are the serotypes that cause most of the meningococcal infections [3]. Each of these common serotypes can be linked to various regions around the globe. The main serotypes causing meningococcal disease in Africa are serotypes A and C [3]. Serotypes B and C are the main serotypes seen in Europe and the Americas [3]. Serotype Y is what generally causes infection in the United States and Canada [3]. Epidemic outbreaks around the world is linked to serotype W [3].
N. meningitidis is one of the common and important causes of community-acquired bacterial meningitis in the United States [3]. Children and adults can be infected by this bacterium. The infection has a high mortality rate if not recognized and treated immediately following the diagnosis. N. meningitidis can lead to other infections as well such as; meningococcal septicemia, pneumonia, septic arthritis, pericarditis, and urethritis [3]. This bacterium can cause both endemic and epidemic infections, also having the ability to infect young, healthy adults [3].
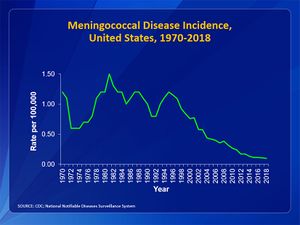
Due to the routine use of the meningococcal vaccination, the number of reported cases of meningococcal disease has decreased (Fig 1). In the United States, the number of cases each year is less than 1 case per 100000 [3]. February through March present the highest infection rates seen in the United States [3]. Infants less than 1 year old make up the highest rate of meningococcal disease seen with 5.38 cases per 100000 [3]. In sub-Saharan Africa, N.meningitidis has led to endemics and epidemic outbreaks and is an important cause of bacterial meningitis [3]. Mortality rate is very dependent on whether those who are infected have seeked treatment or not. The mortality rate for individuals who have received treatment ranges from approximately 10 to 14% [3]. In patients who do not receive treatment, the mortality rate rises up to 50% [3].
Invasive Behavior
N. meningitidis or meningococci are obligate commensals in humans, meaning that the bacterium is colonizing the nasal and oral mucosa of humans without causing them harm. This phenomenon of the bacterium remaining in the mucosa without harming the human is known as carriage [6]. Approximately 10% of the population may carry N. meningitidis in the upper airway during nonepidemic situations and act as asymptomatic carriers [6]. The time in which one person remains in the carrier state can vary between being chronic, lasting for several months, intermittent or transient [6]. It has been seen that colonization of the meningococci can induce an antibody response from the three major immunoglobulin classes and may act as an immunizing event [6]. This situation will occur within a few weeks of obtaining the bacterium.
However, the bacterium does have the ability to enter the bloodstream. On occasion, shortly after colonization in the upper airway, N. meningitidis strains can penetrate the mucosal membrane and from there enter the bloodstream [6]. Once the bacterium has entered the bloodstream, various forms of disease caused by the bacterium may develop. It is important to note that non-carriers are at high-risk for meningococcal disease because it is unknown about their capability to maintain a commensal relationship with an acquired strain of N. meningitidis [6].
Risk Factors, Causes, and Transmission
Signs and Symptoms
Include some current research, with at least one figure showing data.
Treatment and Prevention
Include some current research, with at least one figure showing data.
Metabolism and Virulence
Conclusion
References
- ↑ 1.0 1.1 Meningococcal Photos, CDC, Accessed April 20th, 2020
- ↑ Slonczewski, J., and Foster J. W.. Microbiology: An Evolving Science. New York
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 [Nguyen N, Ashong D. Neisseria Meningitidis. [Updated 2020 Mar 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK549849/ ]
- ↑ Serotypes and the Importnace of Serotyping Salmonella, CDC, Accessed April 20th, 2020
- ↑ Surveillance, CDC, Accessed April 20th, 2020
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Yazdankhah, S. P., & Caugant, D. A. (2004). Neisseria meningitidis: an overview of the carriage state. Journal of Medical Microbiology, 53(9), 821–832. doi: 10.1099/jmm.0.45529-0
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.
