Gloeobacter violaceus
A Microbial Biorealm page on the genus Gloeobacter violaceus
Classification
Higher order taxa
Bacteria; Cyanobacteria; Cyanophyceae; Chroococcales; Gloeobacteraceae
Species
|
NCBI: Taxonomy |
violaceus
Description and significance
It is a rod-shape unicellular cyanobacterium that has been isolated from calcareous rocks in Switzerland [3]. Gloeobacter violaceus is a Gram-negative, photoautotrophic, aquatic and photosynthetic cyanobacteria. Thus Gloeobacter violaceus is commonly labelled as a blue-green algae since it has the ability to live in the water and can manufacture it's own food. Although, it is small and unicellular, Gloeobacter violaceus tend to grow in colonies which are surrounded by a sticky mucous sac that plays a role in adhesion, thus these colonies are large enough to be visible with the human eye [2]. Gloeobacter violaceus is sensitive to strong light and a photoautotroph that contains chlorophyll a, caarotenoids, and phycobiliproteins. Molecular phylogenetic analysis of gloeobacter violaceus has revealed that this linage has diverged from other cyanobacteria and also, that it possesses oxygenic photosynthesis characteristics. Another distinct quality of gloeobacter violaceus is that its' composition of fatty acids is different because it has a high content of polyunsaturated fatty acids (PUFA). A large content of PUFA is unusual for because it was thought that instead there should be a larger composition of (sulfoquinovosyl diacylglycerol SQDG) to ensure photosystem stabilization. These remarkable qualities make it important to sequence the genome of Gloeobacter violaceus because it will reveal the genetic background that is responsible for the origin and evolution of oxygenic photosynthesis. The common method to determine the entire genome of Gloeobacter violaceus is the use of shotgun method in conjunction with the bridging shotgun strategy [3].
Genome structure
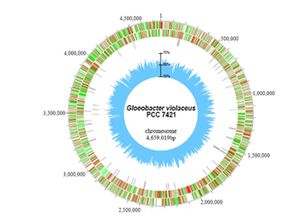
Scientist have revealed that the genome of Gloeobacter violaceus was comprised of a single circular chromosome that contained 4,659,019 base pairs. The average GC content was determined to be 62% and there was also no detection of plasmids during the course of sequencing. The total number of potential protein encoding genes for the entire genome was determined to be 4430 or on average one gene for evey 1052 base pairs. Only 610 genes of gloeobacter violaceus had matches to other cyanobacterial genomes, with about half of these having no known function. Also, about 20 % or 684 genes were found to be unique to this organism [1]. Other interesting features of G. violaceus has been unveiled in comparisons of the assigned gene components with those of other cyanobacteria. It has revealed distinctive features of the G. violaceus genome, such as the genes for PsaI, PsaJ, PsaK, and PsaX for Photosystem I and PsbY, PsbZ and Psb27 for Photosystem II were missing, and those for PsaF, PsbO, PsbU, and PsbV were poorly conserved. CpcG for a rod core linker peptide for phycobilisomes and nblA related to the degradation of phycobilisomes were also missing. These observations can be explained due to the fact that photosynthesis in G. violaceus takes place not in thylakoid membranes but in the cytoplasmic membrane [2].
Cell structure and metabolism
Gloeobacter Violaceus is unicellular organism that exhibits atypical characteristics compared to other cyanobacterium. Its unusual characteristic is the lack of an internal thylakoid membrane system [3]. The rod shaped phycobilisomes of this organism are heavily packed and attach to the plasmic surface of they cytoplasmic membrane. The cell walls of Gloeobacter violaceus are comprised of multiple layers. Starting from the exterior of the cell wall there is a sheath layer, then a double tracked structured outer membrane, intermediate cell wall layer that is electron dense, then lastly, a electron dense peptidoglycan layer. It gets its energy from oxygenic photosynthesis which is unique to this organism because usually the machinery for photosynthesis is found in the thylakoid membrane and not in the cytoplasmic membrane. Important products that G. violaceus produces are petJ and petE which are used for soluble electron transfer catalysts. Also, Gloeobacter violaceus has a lower light requirement for growth than other cyanobacteria, but a higher than normal light saturation level for electron transfer rate [4].
Ecology
The appearance of oxygenic photosynthetic organisms, such as Gloeobacter violaceus, on earth represents the largest discontinuous process in the evolution of photosynthetic organisms. Since the appearance of organisms that are oxygenic photosynthetic, scientist have revealed that they contribute the determining factors that has shifted the direction of global biological evolution through an increase in the oxygen concentration on the Earth [5]. Another important characteristis of the cyanobacterium Gloeobacter violaceus, contains Ths and GroEL and the recombination repair proteins RecA and RadA which enable it to exhibit characterisitics that are similar to Archaea in expressing multiple detoxification PHX bacterioferritin comigratory proteins and several Hsp20s. Also, circadian rhythms have intensively been studied in cyanobacteria, and genes involved in various processes of circadian timing and regulation have been identifiedin many species of cyanobacteria. These genes include kaiABC as the major genetic elements of the circadian clock, sasA, cikA, ldpA, and pex as input modifiers, and rpoD2 and cpmA as output modifiers. Even after scientist completed an intensive search, they could not detect kaiABC in the Gloeobacter genome. It is therefore likely that Gloeobacter does not have a genetic controlling system for circadian rhythms and that cyanobacteria have acquired this system after divergence of the Gloeobacter lineage. Alternatively, Gloeobacter might have lost such genes [2].
Pathology
Gloeobacter violaceus and all species of the genus Gloeobacter are non-pathogenic thus they do not cause any known diseases to humans, animals, or plant hosts.
Application to Biotechnology
Does this organism produce any useful compounds or enzymes? What are they and how are they used? In cyanobacteria many compounds, including chlorophylls, carotenoids, and hopanoids, are synthesized from the isoprenoid precursors isopentenyl diphosphate (IPP) and dimethylallyl diphosphate.
Current Research
In a study conducted by Steiger et., al 2005, they revealed that Gloeobacter violaceus is a cyanobacterium isolated from other groups by lack of thylakoids and unique structural features of its photosynthetic protein complexes. They investigated carotenoid biosynthesis with respect to the carotenoids formed and the genes and enzymes involved. Their carotenoid analysis identified ss-carotene as major carotenoid and echinenone as a minor component. This composition is quite unique and the cellular amounts are up to 10-fold lower than in other unicellular cyanobacteria. Also, carotenoid biosynthesis was also found to be up-regulated in a light-dependent manner. This enhanced biosynthesis partially compensates for photooxidation especially of ss-carotene. They also sequenced the genome of G. violaceus and analyzed several gene candidates homologous to carotenogenic genes from other organisms obtained. Functional expression of all candidates and complementation in Escherichia coli led to the identification of all genes involved in the biosynthesis of the G. violaceus carotenoids with the exception of the lycopene cyclase gene. An additional diketolase gene was found that functioned in E. coli but is silent in G. violaceus cells. The biggest difference from all other cyanobacteria is the existence of a single bacterial-type 4-step desaturase instead of the poly cis cyanobacterial desaturation pathway catalyzed by two cyanobacterial-type desaturases and an isomerase. The genes for these three enzymes are absent in G. violaceus [6].
In a study performed by Krogmann et., al they examined the complete genome sequence of Gloeobacter violaceus. This allowed them to understand better the structure of the phycobilisomes (PBS) of this cyanobacterium. In their study genomic analysis revealed peculiarities in these PBS: the presence of genes for two multidomain linker proteins, a core membrane linker with four repetitive sequences (REP domains), the absence of rod core linkers, two sets of phycocyanin (PC) alpha and beta subunits, two copies of a rod PC associated linker (CpcC), and two rod cap associated linkers (CpcD). They investigated the PBS proteins by gel electrophoresis, amino acid sequencing and peptide mass fingerprinting (PMF). They were able to conclude that two unique multidomain linkers contain three REP domains with high similarity and these were found to be in tandem and were separated by dissimilar Arms. One of these, with a mass of 81 kDa, is found in heavy PBS fragments rich in PC. They then proposed that it links six PC hexamers in two parallel rows in the rods. The other unique linker has a mass of 91 kDa and is easily released from the heavy fragments of PBS. We propose that this links the rods to the core. The presence of these multidomain linkers could explain the bundle shaped rods of the PBS. The presence of 4 REP domains in the core membrane linker protein (129 kDa) was established by PMF. This core linker may hold together 16 AP trimers of the pentacylindrical core, or alternatively, a tetracylindrical core of the PBS of G. violaceus.[7]
New linker proteins in phycobilisomes isolated from the cyanobacterium Gloeobacter violaceus PCC 7421.Koyama K, Tsuchiya T, Akimoto S, Yokono M, Miyashita H, Mimuro M. Department of Interdisciplinary Environment, Graduate School of Human and Environmental Studies, Kyoto University, Japan.
Two new linker proteins were identified by peptide mass fingerprinting in phycobilisomes isolated from the cyanobacterium Gloeobacter violaceus PCC 7421. The proteins were products of glr1262 and glr2806. Three tandem phycocyanin linker motifs similar to CpcC were present in each. The glr1262 product most probably functions as a rod linker connecting phycoerythrin and phycocyanin, while the glr2806 product may function as a rod-core linker. We have designated these two proteins CpeG and CpcJ, respectively. The morphology of phycobilisomes in G. violaceus has been reported to be a bundle-like shape with six rods, consistent with the proposed functions of these linkers.
The secondary electron acceptor of photosystem I in Gloeobacter violaceus PCC 7421 is menaquinone-4 that is synthesized by a unique but unknown pathway.Mimuro M, Tsuchiya T, Inoue H, Sakuragi Y, Itoh Y, Gotoh T, Miyashita H, Bryant DA, Kobayashi M.
Department of Technology and Ecology, Hall of Global Environmental Research, Kyoto University, Kyoto 606-8501, Japan. mamo-mi@mm1.mbox.media.kyoto-u.ac.jp
The secondary electron acceptor of photosystem (PS) I in the cyanobacterium Gloeobacter violaceus PCC 7421 was identified as menaquinone-4 (MQ-4) by comparing high performance liquid chromatograms and absorption spectra with an authentic compound. The MQ-4 content was estimated to be two molecules per one molecule of chlorophyll (Chl) a', a constituent of P700. Comparative genomic analyses showed that six of eight men genes, encoding phylloquinone/MQ biosynthetic enzymes, are missing from the G. violaceus genome. Since G. violaceus clearly synthesizes MQ-4, the combined results indicate that this cyanobacterium must have a novel pathway for the synthesis of 1,4-dihydroxy-2-naphthoic acid.
The cyanobacterium Gloeobacter violaceus PCC 7421 uses bacterial-type phytoene desaturase in carotenoid biosynthesis.Tsuchiya T, Takaichi S, Misawa N, Maoka T, Miyashita H, Mimuro M.
Department of Technology and Ecology, Hall of Global Environmental Research, Kyoto University, Kyoto 606-8501, Japan.
Carotenoid composition and its biosynthetic pathway in the cyanobacterium Gloeobacter violaceus PCC 7421 were investigated. beta-Carotene and (2S,2'S)-oscillol 2,2'-di(alpha-L-fucoside), and echinenone were major and minor carotenoids, respectively. We identified two unique genes for carotenoid biosynthesis using in vivo functional complementation experiments. In Gloeobacter, a bacterial-type phytoene desaturase (CrtI), rather than plant-type desaturases (CrtP and CrtQ), produced lycopene. This is the first demonstration of an oxygenic photosynthetic organism utilizing bacterial-type phytoene desaturase. We also revealed that echinenone synthesis is catalyzed by CrtW rather than CrtO. These findings indicated that Gloeobacter retains ancestral properties of carotenoid biosynthesis.
Unique fluorescence properties of a cyanobacterium Gloeobacter violaceus PCC 7421: reasons for absence of the long-wavelength PSI Chl a fluorescence at -196 degrees C.Mimuro M, Ookubo T, Takahashi D, Sakawa T, Akimoto S, Yamazaki I, Miyashita H.
Department of Physics, Biology and Informatics, Faculty of Science, Yamaguchi University, Yoshida, Yamaguchi, 753-8512 Japan. mamo_mi@bio.h.kyoto-u.ac.jp
We investigated the reason for the absence of the long-wavelength PSI Chl a fluorescence at -196 degrees C in the cyanobacterium Gloeobacter violaceus using two methods: p-nitrothiophenol (p-NTP) treatment and time-resolved fluorescence spectra. The p-NTP treatment showed that PSII Chl a fluorescence was specifically affected in a manner similar to that for Synechocystis sp. PCC 6803 and spinach chloroplasts, although there were no components modified by the p-NTP treatment, indicating an absence of the long-wavelength PSI Chl a fluorescence. The time-resolved fluorescence spectra with a time resolution of 1.3 ps and spectral resolution of 1.0 nm gave no indication of the presence of the long-wavelength PSI fluorescence in the wavelength region between 700 nm and 760 nm, indicating that a very fast energy transfer among Chl a molecules could not account for the absence of the long-wavelength PSI fluorescence. From these data, it seems that the absence of the long-wavelength PSI fluorescence is due to a lack of the formation of a component responsible for the fluorescence at -196 degrees C, which may originate from a difference in the amino acid sequence. We discuss the significance of this phenomenon and interpret our findings in terms of the evolution of cyanobacteria.
Proton gradients in intact cyanobacteria.Belkin S, Mehlhorn RJ, Packer L.
Department of Physiology-Anatomy, Lawrence Berkeley Laboratory, University of California, Berkeley 94720, USA.
The internal pH values of two unicellular cyanobacterial strains were determined with electron spin resonance probes, over an external pH range of 6 to 9, in the light and in the dark. The slow growing, thylakoid-lacking Gloeobacter violaceus was found to have a low capacity for maintaining a constant internal pH. The distribution pattern of weak acid and amine nitroxide spin probes across the cell membranes of this organism, in the light and in the dark, was consistent with the assumption that it contains a single intracellular compartment. At an external pH of 7.0, intracellular pH was 6.8 in the dark and 7.2 in the light. The cells of Agmenellum quadruplicatum, a marine species, were found to contain two separate compartments; in the dark, the pH of the cytoplasmic and the intrathylakoid spaces were calculated to be 7.2 and 5.5, respectively. Upon illumination, the former increased and the latter decreased by about 0.5 pH units.
References
1.Jackisch, Y., Sandmann, G., Steiger, S., "Carotenoid biosynthesis in Gloeobacter violaceus PCC4721 involves a single crtI-type phytoene desaturase instead of typical cyanobacterial enzymes". Archives of Microbiology. 2005. Volume 184
2.Jurgens, U., Schneider, S., "Cell Wall and Sheath Constituents of the cyanobacterium gloeobacter violaceus". Archives of Microbiology. 1991. Volume 155. p. 312-318.
3.Nakamura, Y., Kaneko, T., Sato, S., "Complete Genome Structure of Gloeobacter violaceus PCC 7421, a Cyanobacterium that lacks Thylakoids". DNA Research. 2003. Volume 10. p. 137-145.
4.Rippka, R., "A cyanobacterium which lacks thylakoids." (1974) Arch Microbiol 100, 419-436
5.Mimuro, M., "Photosynthetic properties of a cyanobacterium, Gloeobacter violaceus PCC7421."Department of Physics, Biology and informatics, Faculty of Science, Yamaguchi University. 1-4
6.Steiger, S., "Carotenoid biosynthesis in Gloeobacter violaceus PCC4721 involves a single crtI-type phytoene desaturase instead of typical cyanobacterial enzymes." Arch Microbiology. 2005;184(4):207-14
7.Krogmann DW, Pérez-Gómez B, Gutiérrez-Cirlos EB, Chagolla-López A, González de la Vara L, Gómez-Lojero C. "The presence of multidomain linkers determines the bundle-shape structure of the phycobilisome of the cyanobacterium Gloeobacter violaceus PCC 7421." Department of Biochemistry, Purdue University, West Lafayette, IN, 47907-1157, USA.