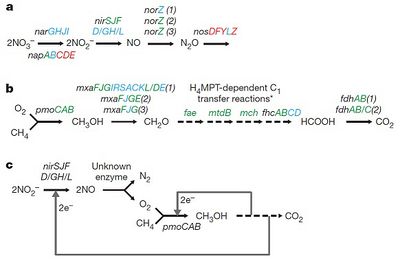
Nitrate/nitrite methane oxidation
Introduction
Methane (CH4) is a potent greenhouse gas. Marine and freshwater sources are important sources of methane. In oceans, methane seeps out of fissures along the ocean floor and from methane hydrates. The methane eventually bubbles out of the ocean and enters the atmosphere. It is estimated approximately 33 million tons of methane enters the atmosphere in this manner per year (Biello, 2006). Methane is also released from freshwater sediments (i.e. from lakes and wetlands) as a result of methanogenic activity.
Methane can be oxidized to carbon dioxide (CO2) by microbes in both marine and freshwater sediments. The ability of microbes to degrade methane means that there is a methane sink which can mediate fluxes of methane into the atmosphere. Although methane degradation results in greater CO2 flux into the atmosphere, CO2 has less impact than methane as a greenhouse gas. Bacteria and archaea that can metabolize methane are called methanotrophs and reverse methanogens, respectively.
Methane oxidation can occur in oxic and anoxic sediments. Aerobic metabolism of methane requires oxygen so that the methane molecule can be activated. The reaction with oxygen forms methanol, which is a better electron donor than methane. A series of oxidation reactions, where each reaction utilizes a different enzyme, results in the production of CO2. Methylococcus capsulatus is the most well-studied aerobic methanotroph (Oremland, 2010).
There are two known types of anaerobic oxidation of methane. One of the types of anaerobic oxidation entails the interaction of sulfate-reducing bacteria with reverse methanogens in marine sediments. It is thought that methane oxidation fuels sulfate reduction by providing H2 that sulfate-reducing bacteria can use as an electron donor. Sulfate reduction, in turn, creates thermodynamic conditions that allow for methane oxidation to occur (Valentine and Reeburgh, 2000); methane cannot be oxidized in unfavorable thermodynamic conditions because it is a poor electron donor. This symbiotic relationship is summarized as:
CH4 + SO42- --> HCO3- + HS- + H2O
The other type of anaerobic oxidation is microbial nitrate/ nitrite oxidation of methane. This entails the coupling of denitrification to methane oxidation to form CO2. Nitrate/nitrite oxidation of methane has become an important topic of research given the intensification of agriculture and the resulting increase in agricultural runoff (Raghoebarsing et al, 2006). In addition, Nauhaus et al (2005) and Raghoebarsing et al (2006) found that nitrate and nitrite were better substrates for anaerobic oxidation of methane than sulfate. This suggests that nitrate/ nitrite-dependent methane oxidation may be more prevalent in freshwater and marine ecosystems than sulfate-dependent methane oxidation.
There are two main theories that have been proposed for microbial nitrate/ nitrite oxidation of methane. The first theory proposes that a microbial consortium, including bacteria and archaea, is required for the coupling of both processes to occur (Raghoebarsing et al, 2006). The second theory proposes that methane oxidation can occur in the absence of archaea. The bacterium responsible for the coupling of both processes has been named Candidatus Methylomirabolis oxyfera, or M. oxyfera. The overall pathway utilized by M. oxyfera has been termed as ‘intra-aerobic’ because oxygen is produced from nitrite in anaerobic conditions and is then used to aerobically oxidize methane (Ettwig et al, 2010). Both theories are discussed in greater detail below. Research is still being conducted to determine whether just one theory or both theories explain the method(s) by which microbial nitrate oxidation of methane occurs.
The Microbial Consortia Theory
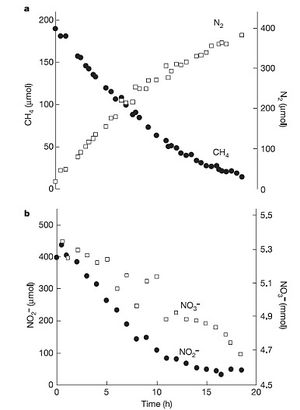
This microbial consortia theory hypothesizes that an archaeon, Methanosarcinales, and bacteria, from the NC10 phylum, is required for the coupling of denitrification and anaerobic oxidation of methane (Raghoebarsing et al, 2006). Freshwater, anoxic sediment was collected from a canal in the Netherlands. The sediment was incubated and provided with carbon-13 labeled methane (electron donor), nitrite (electron acceptor), nitrate (electron acceptor), bicarbonate and essential trace elements. They found that nitrate, nitrite and methane were being consumed and that dinitrogen gas (N2) was being produced (Figure 1). Figure 1 shows that more nitrite was consumed by the culture than nitrate, indicating that nitrite is a better and therefore preferred electron acceptor. In addition, Raghoebarsing and colleagues found that the denitrification rate was 290.1% faster with methane addition, suggesting that denitrification and methane oxidation are coupled.
To ensure that both archaea and bacteria were participating in the coupling of denitrification and methane oxidation, Raghoebarsing et al (2006) measured the absorption of methane into the membrane lipids of the bacteria and archaea. In order to measure membrane methane absorption, they measured depletion of 13C in the membrane and compared it to that of methane before and after exposure to methane. They found that bacterial biomarkers were labeled more quickly and were more depleted in 13C than archaeal biomarkers; in fact only one archaeal biomarker showed signs of significant 13C depletion. The slow incorporation of methane into biomass by the archaeon suggests slow growth. Fluorescence in situ hybridization (FISH) analysis of culture biomass showed the presence of bacteria and archaea in the consortium. Archaea only comprised of 10% of the consortium, also indicating slow growth.
The mechanism by which the coupling occurs has not been determined, but is thought to occur in a manner similar to sulphate-dependent methane oxidation. It is proposed that the archaea oxidize methane to CO2 after which they transfer electrons to the bacteria which can then reduce nitrate/ nitrite to N2 (Conrad, 2009). The coupling provides the bacteria and archaea with the thermodynamic conditions required for denitrification and methane oxidation, respectively. The following equations represent the overall reactions of methane with nitrate (NO3-) and nitrite (NO2-) (Raghoebarsing et al, 2006):
5CH4 + 8NO3- + 8H+ --> 5CO2 + 4N2 + 14H2O (ΔG0' = -765 kJ mol-1 CH4)
3CH4 + 8NO2-+ 8H+ --> 3CO2 + 4N2 + 10H2O (ΔG0' = -928 kJ mol-1 CH4)
The Intra-aerobic Denitrification Pathway Theory
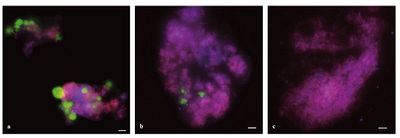
Ettwig et al (2008) found that methane could be anaerobically oxidized in the absence of archaea. The researchers experimented with the same enriched culture, in anoxic conditions, used by Raghoebarsing and colleagues (2006) over a 22 month period. The culture was also provided with the same nutrients, electron donors and electron acceptors. They found that archaeal composition reduced over the time period and could not be detected by the fifteenth month using FISH analysis of biomass (Figure 2). Despite the decrease in archaeal biomass, the bacteria continued to oxidize methane, which suggests one of two things:
1. Archaea and bacteria are both able to anaerobically oxidize methane completely separately, but the bacteria is able to outcompete the archaea or,
2. If archaea and bacteria are present, it is beneficial for them to form a syntrophic relationship and divide the labour; in an event where the archaea disappears, the bacteria can revert to an alternative pathway which allows it to continue with denitrification coupled to methane oxidation.
The bacterium responsible for the coupling of nitrate/nitrite reduction and methane oxidation is a denitrifying methanotrophic bacterium in the NC10 phylum named Candidatus Methylomirabolis oxyfera, or M. oxyfera. It is a slow-growing, rod-shaped and gram negative bacterium. It was isolated by Ettwig and colleagues (2008, 2010). To date, it has only been found in freshwater sediments at mesophilic temperatures and pH.
The metagenomes of two cultures of M. oxyfera from enriched freshwater sediments (collected from Australia and the Netherlands) were sequenced to piece together the mechanism of nitrite-dependent anaerobic oxidation of methane (Ettwig et al, 2010). The researchers found that M. oxyfera expressed genes for the conversion of NO3- to NO2-, NO2- to NO (nitric oxide) and NO to N2O (nitrous oxide), but no genes for the conversion of N2O to N2 (Figure 3a). Despite this, N2O could only be found in trace amounts which in turn suggests that the NO is being converted to N2 using an alternative mechanism where N2O is not an intermediate. On the other hand, it may be that N2O is being reduced to N2 using, not N2O reductase, but a different enzyme.
Ettwig et al (2010) also found that the metagenome did not contain genes coding fumarate-adding glycl-radical enzymes or methyl-coenzyme M reductase, either of which can be used for anaerobic oxidation of methane. Rather, they found that the metagenome coded for the aerobic oxidation of methane (Figure 3b). First, methane reacts with oxygen, using the membrane-bound form of the enzyme methane mono-oxygenase (pMMO), to form methanol (CH3OH) and water. The pMMO complex is thought to function either by aiding oxygen production and then consuming the oxygen produced or by using NO directly as an oxygen source. Further research needs to be conducted to establish the mechanism by which pMMO works in M. oxyfera.
A separate enzyme, methanol dehydrogenase, is used to form formaldehyde (CH2O) from CH3OH. CH2O, in turn, is oxidized to form formate (HCOO-). The conversion of CH2O to HCOO- requires a tetrahydromethanopterin-dependent C1 transfer module which is thought to be a primordial metabolic molecule, indicating that M. oxyfera is from a particularly deep-branching lineage. Finally, HCOO- is converted to CO2 using the enzyme formate dehydrogenase. The electron flow resulting from the oxidation of methane generates a proton motive force and therefore allows for ATP synthesis (Ettwig et al, 2010).
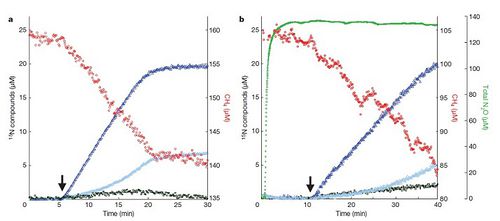
A peculiar aspect of the pathway is that nitrite is required for the mechanism to proceed even though M. oxyfera contains genes that code nitrate reductase (Figure 3a). Ettwig et al (2010) conducted an experiment where they provided an enrichment culture, under anoxic conditions, with nitrate and carbon-13 labeled CH4. Methane oxidation did not occur and N2 gas was not produced when the culture was incubated. However, as soon as nitrogen-15 labeled nitrite was added, methane oxidation commenced and labeled N2 gas was formed in stoichiometric quantities, indicating that N2 was produced solely from the added nitrite (Figure 4a). Is nitrate reductase not produced even though the metagenome codes for it?
Nevertheless, the findings suggest that incomplete denitrification somehow results in the production of oxygen and N2 gas (Figure 3c). Ettwig et al (2010) suggest that NO is converted to N2 and oxygen in a one-step mechanism using an ‘NO dismutase’ enzyme. The proposed NO dismutase enzyme, however, could not be isolated by the researchers. Further research needs to be conducted to isolate and identify the enzyme(s) responsible for the conversion of NO to N2 and oxygen.
Assuming that the aforementioned reaction occurs, how is the NO reductase enzyme (used to reduce NO to N2O) coded by the metagenome relevant in M. oxyfera's metabolic system? In other bacterial species, NO reductase has been used to detoxify NO as NO exposure can induce cell stress (Frey and Kallio, 2005). The prediction that N2O was not an intermediate for N2 production or a suitable electron acceptor for methane oxidation was tested by incubating the enrichment culture with 13CH4, nitrate and N2O (Ettwig et al, 2010). The researchers found that neither methane nor N2O were consumed, as per the prediction (Figure 4b). The role of NO reductase needs to be determined in future experiments.
Implications
Evolutionary Significance
Microbial nitrate/nitrite oxidation of methane is likely to have evolutionary significance because it is an oxygen-producing process and therefore may have contributed to the oxygenation of the atmosphere 2.45-1.85 billion years ago (Holland, 2006). The oxygenation of the atmosphere is important because it allowed for the evolution of aerobic organisms on Earth. The processes originally attributed to oxygenation of the atmosphere are photosynthesis, chlorate respiration and the detoxification of reactive oxygen species (Ettwig et al, 2010). Although oxygen produced from NO in the intra-aerobic pathway, discovered by Ettwig et al (2010), is used for methane oxidation, it is possible that some of the oxygen molecules escaped before they could be used for methane oxidation. The oxygen would have eventually dissolved out of the ocean and entered the atmosphere, thereby contributing to atmospheric oxygenation.
On what basis can we say that the intra-aerobic pathway is likely to be very old? M. oxyfera has the tetrahydromethanopterin-dependent C1 transfer module, a primordial metabolic molecule, suggesting that M. oxyfera is from a deep-branching lineage. Therefore, the pathways proposed by Ettwig et al (2010) and Raghoebarsing et al (2006) may have evolved before photosynthesis and respiration. M. oxyfera would have thrived in the Archaean Eon conditions were anaerobic and the atmosphere largely consisted of methane. In addition, Ducluzeau et al (2009) proposed that NO, nitrite and nitrate were widely available during the Archaen Eon as strong oxidants. Microorganisms using nitrate-dependent oxidation of methane could have thrived in such conditions. The pathways, therefore, also likely preceded aerobic respiration as aerobic respiration is thought to have evolved after photosynthesis (Ducluzeau et al, 2009). The intra-aerobic pathway, especially, may have actually allowed for the development of niches for the evolution of aerobic metabolism due to the local production of oxygen (Ettwig et al, 2010).
Methane Gas Hydrates
The rise in atmospheric temperature as a result of climate change has caused the oceans to warm. As the oceans continue to warm, methane hydrates become more and more unstable. Methane hydrates, or methane clathrates, are structures that can be found on the ocean floor. They are made up of water crystals and can trap enormous amounts of methane gas within the resulting lattice. It is estimated that these methane reservoirs hold between 500,000 and 1,000,000 Tg of methane (Conrad, 2009). Methane hydrates require low temperatures and low pressures or moderate temperatures and high pressures to remain stable (Biello, 2006).
At present, a large proportion of the methane they release (70-300 Tg CH4 per year) is consumed by methane-oxidizing microbes, meaning that methane hydrate emissions only compose about 1% of the total atmospheric methane budget (Conrad, 2009). The actions of methane-oxidizing microbes are, therefore, vital in mediating upward methane fluxes from oceans. However, if the oceans continue to warm, it may well be that there will be explosive releases of methane from the methane hydrates which cannot be contained by methane-oxidizing microbial species. The resulting drastic increases in atmospheric methane would prove to be catastrophic for all ecosystems and the life that they sustain.
Ettwig et al (2008, 2010) and Raghoebarsing et al (2006) isolated the enriched samples of nitrate/ nitrite-dependent anaerobic methane oxidizers from freshwater systems. So far, none have been isolated from marine sediments. Part of this may be due to the fact that gas hydrates tend to be in parts of the ocean that are difficult and dangerous to access. It is possible that there are nitrate/ nitrite-dependent anaerobic methane oxidizers in the deep ocean (Valentine, 2011). It would be interesting to study the ability of M. oxyfera to survive and function in deep ocean-like conditions.
Global Climate Change
The global budget of methane in the atmosphere is 500-600 Tg CH4 per year. Its levels have increased between pre-industrial times and now from 715 ppbv to 1770 ppbv. A significant proportion of the increase can be attributed to fossil fuel burning and the intensification of agriculture – ruminants and large-scale rice plantation produce substantial amounts of methane (Conrad, 2009). The increase of methane in the atmosphere has significantly contributed to climate change, given its status as a greenhouse gas with a much higher global warming potential that carbon dioxide.
Climate warming has a positive feedback loop. In the case of wetlands, for example, warming has accelerated the metabolism of soil methanogens, thereby increasing the release of Warming, in turn, will further increase methane release from the soil provided that soil moisture levels remain high (Cao et al, 1998). Warming, as discussed earlier, is also expected to increase methane fluxes from methane hydrates in oceans.
Methane-oxidizing bacteria, as mentioned earlier, can mediate methane fluxes and nitrate levels. It may, therefore, be possible to use them for bioremediation. Nitrate/ nitrite-dependent methane-oxidizing microbes can be used to offset increases in methane production (Raghoebarsing et al, 2006). In the case of agricultural run-off, the same microbes can be used to consume the nitrate/ nitrite in soils and aquatic and marine sediments. The microbes can also be used to restore eutrophic marine and freshwater ecosystems as they thrive in anoxic conditions where there is a lot of nitrite and methane available. The use of the intra-aerobic pathway may help re-oxygenate the ecosystem, allowing the ecosystem to sustain life once again. This is, however, difficult to say given that relatively little oxygen escapes the intra-aerobic pathway (Ettwig et al, 2010).
Conclusion
It is evident that microbial nitrate/ nitrite oxidation of methane has important implications for methane fluxes, making it an important area of research. Firstly, it needs to be determined whether both theories, the microbial consortium theory and the intra-aerobic denitrification pathway theory, or just one theory serves as a viable pathway for the coupling of anaerobic methane oxidation and nitrate/ nitrite reduction. To a large extent, the existence of the intra-aerobic denitrification pathway can be proven by the isolation and identification of the enzyme, or enzymes, responsible for the conversion of NO to N2 and O2. The microbial consortium theory, on the other hand, can be tested by removing either the bacteria or the archaea from the enrichment culture and evaluating how the remaining species survives. Does anaerobic oxidation of methane continue? Does denitrification continue?
Scientists also need to determine what conditions anaerobic methane-oxidizing species such as M. oxyfera require for their survival. The enrichment cultures obtained by Raghoebarsing et al (2006) and Ettwig et al (2008, 2010) were from shallow freshwater sediments; pH and temperature conditions were both mesophilic meaning that pH was close to neutral and temperatures were moderate. Can the microbial species involved in nitrate/ nitrite oxidation of methane survive in high pressure and high salinity environments? What about thermophilic or psychrophilic environments?
Another important question is whether or not M. oxyfera can be cultured in enormous quantities for the purposes of bioremediation. In order to be able to culture bacteria, knowledge of nutrient requirements (macro- and micro-nutrients, inorganic ions) and optimal environmental conditions (temperature, pH and levels of oxygen) is required. Once cultured, the effect of the species on the biodiversity of environments needing to be restored in terms of methane and nitrate/ nitrite flux needs to be determined. Adding non-native species to environments or drastically increasing their population in environments that they already exist in can result in food web and ecosystem collapse. Therefore, it is only with such holistic understanding that species such as M. oxyfera can be used for bioremediation purposes; such understanding requires the collaboration of microbiologists, biochemists and ecologists. Despite this, microbes specializing in anaerobic nitrate/ nitrite oxidation of methane hold a lot of purpose as a means of offsetting climate change resulting from large outward fluxes of methane.
References
Oremland, RS. 2010. NO connection with methane. Nature 464: 500-501.
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2009, Kenyon College.