Thermoresistance mechanisms of hyperthermophilic archaea
Hyperthermophile identification
The Earth contains many types of environments some of which, due to certain features, are inhospitable to most cellular life. These environments include, but are not limited to: areas of extremely high/low temperature, extremely high/low pH levels, high concentrations of NaCl and high radiation levels Extreme environments. Species that are able to thrive in these environments are called extremophiles. A particular subset of these types is the hyperthermophile. Hyperthermophilic organisms are defined as those that thrive above temperatures of 60 degrees Celsius ThermophileHyperthermophiles, with the other temperature range being populated by psychrophiles, mesophiles and thermophiles Extremophiles The most temperature resistant organisms discovered so far have been Archaea, although several hyperthermophilic bacteria are known. These Archaea are often found around superheated hydrothermal vents at the bottom of the ocean floor. This is also where the most current temperature resistant Archaeon, species 121, was discovered Strain 121. Thus, microbial life has been discovered in even the most inhospitable regions of the earth, and has evolved to survive these environments.
Figure 1: Thermophilic Algae and Bacteria are responsible for the blue hue of this thermal spring.
Origins and factors of stability
The origin of hyperthermophilic archaea is theorised to predate species that live at lower temperatures, as 16S and 18S rRNA gene sequence data analysis places hyperthermophiles near the root of the phylogenetic tree of life (Woese et al. 1990). The difficulties of a high-temperature environment stem from the need to maintain stable functioning of all cellular mechanisms at temperatures where un-adapted proteins would denature and regular membranes would become too permeable. Adaptations to deal with these particular requirements seem to have a profound effect on the thermal sensitivity of the cells, as hyperthermophilic organisms, which thrive in conditions of high temperatures, are effectively inactive at moderate-low temperatures (Koutsopoulos et al. 2005). Thus hyperthermophilic organisms have evolved to thrive in a very particular environment to the extent that they have lost the ability to function properly at lower temperatures.
Protein stability
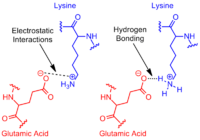
Proteins and enzymes represent an essential part of the machinery required for cellular life. Hyperthermophilic organisms have proteins with specific adaptations to survive the high temperatures in the environment. Fundamentally, protein stability is dependent on a protein’s tendency to denature, a properly folded form being absolutely essential for its function. Lower denaturing rates, i.e. slower unfolding, would be evidence of a more stable protein. This is exactly what has been observed in the proteins of hyperthermophilic Archaea. Their proteins have been found to have exceedingly high resistance to denaturation, sometimes in exchange for slower folding rates during initial protein construction (Luke et al. 2007). One of the ways in which the proteins achieve this is by burying hydrophobic groups deep within their structures. Hydrophobic residues provide stability for proteins along the entire temperature range by the interactions formed between these groups in the protein core (Dong et al. 2008). A recent study by Dong et al. working on the Ribonuclease HII from hyperthermophile Thermococcus kodakaraensis, showed that removing even a few residues from the hydrophobic core of the protein lead to a significant increase in its unfolding rate (Dong et al. 2008). Another factor that improves protein stability is the presence of salt bridges within their structure, formed during initial folding. Increased amounts of intrasubunit ion pairs lead to more salt bridges. This leads to interactions such as hydrogen bonding or electrostatic interactions between residues in different parts of the protein. These interactions help the protein stay folded. The salt bridges have been shown to have a greater effect on stability as temperatures increase (Elcock 1998). This is in line with recent findings that hyperthermophile proteins have higher amounts of ion pairs within their structures (Suhre, Claverie 2003, Cambillau, Claverie 2000).
Fig 2: Types of Salt bridge interactions
Macromolecular crowding may also play a role in stabilizing proteins. The effects of multiple stabilizing molecules, many of them sugar derivatives present in the cells, are thought to be an important factor in maintaining protein stability at high temperatures (Ellis 2001). Aside from proteins, ribozymes and regular enzymes, Archaeal RNA is suspected to have a higher temperature resistance. This is thought to be due to higher levels of guanine and cytosine, which have been found to increase temperature resistance in RNA (Unsworth et al. 2007). In conclusion there is significant evidence showing that hyperthermophilic Archaea have evolved methods of retaining cellular function at high temperatures.
Methods of adaptation
As all cellular function is encoded in an organism’s DNA, an organism must change its DNA to evolve. Thus, DNA transfer is a vital part of several microbial processes. Horizontal gene transfer seems to have played a significant role in the adaptation of hyperthermophilic Achaea to their environment (van Wolferen et al. 2013) Similarly, homologous recombination of DNA allows for species to repair their own genome (van Wolferen et al. 2013).Three known methods of DNA transfer exist: natural transformation (uptake of naked DNA by the organism), conjugation (DNA transfer with two cells in direct contact) and transduction (viral-mediated DNA transfer). All three have been shown to take place in a hyperthermophilic environment, although research shows that conjugation, involving direct contact is favoured (van Wolferen et al. 2013). This is reasonable as the extreme environment will degrade naked DNA at a rapid rate. Furthermore other non-classic pathways, such as vesicle-mediated DNA transfer, are also found in hyperthermophiles, although little is yet known about these processes. (Gaudin et al 2013). Hyperthermophiles are constantly seeking to improve their capability to thrive in their environment via all the known methods of DNA transfer.
References
Cambillau, C., & Claverie, J. M. (2000). Structural and genomic correlates of hyperthermostability. The Journal of Biological Chemistry, 275(42), 32383-32386. doi: 10.1074/jbc.C000497200
Dong, H., Mukaiyama, A., Tadokoro, T., Koga, Y., Takano, K., & Kanaya, S. (2008). Hydrophobic effect on the stability and folding of a hyperthermophilic protein. Journal of Molecular Biology, 378(1), 264-272. doi: 10.1016/j.jmb.2008.02.039
Elcock, A. H. (1998). The stability of salt bridges at high temperatures: Implications for hyperthermophilic proteins1. Journal of Molecular Biology, 284(2), 489-502. doi: 10.1006/jmbi.1998.2159
Ellis, R. J. (2001). Macromolecular crowding: An important but neglected aspect of the intracellular environment. Current Opinion in Structural Biology, 11(1), 114-119. doi: 10.1016/S0959-440X(00)00172-X
Gaudin, M., Gauliard, E., Schouten, S., Houel-Renault, L., Lenormand, P., Marguet, E., & Forterre, P. (2013). Hyperthermophilic archaea produce membrane vesicles that can transfer DNA. United States: WILEY-BLACKWELL. doi: 10.1111/j.1758-2229.2012.00348.x
Karsten Suhre, & Jean-Michel Claverie. (2003). Genomic correlates of hyperthermostability, an update. Journal of Biological Chemistry, 278(19), 17198-17202. doi: 10.1074/jbc.M301327200
Koutsopoulos, S., van der Oost, J., & Norde, W. (2005). Temperature-dependent structural and functional features of a hyperthermostable enzyme using elastic neutron scattering. Proteins, 61(2), 377-384. doi: 10.1002/prot.20606
Luke, K. A., Higgins, C. L., & Wittung‐Stafshede, P. (2007). Thermodynamic stability and folding of proteins from hyperthermophilic organisms. FEBS Journal, 274(16), 4023-4033. doi: 10.1111/j.1742-4658.2007.05955.x
Unsworth, L. D., Van Der Oost, J., & Koutsopoulos, S. (2007). Hyperthermophilic enzymes − stability, activity and implementation strategies for high temperature applications. FEBS Journal, 274(16), 4044-4056. doi: 10.1111/j.1742-4658.2007.05954.x
van Wolferen, M., Ajon, M., Driessen, A. J. M., & Albers, S. (2013). How hyperthermophiles adapt to change their lives: DNA exchange in extreme conditions. Extremophiles : Life Under Extreme Conditions, 17(4), 545-563. doi: 10.1007/s00792-013-0552-6
Woese, C. R., Kandler, O., & Wheelis, M. L. (1990). Towards a natural system of organisms: Proposal for the domains archaea, bacteria, and eucarya. Proceedings of the National Academy of Sciences of the United States of America, 87(12), 4576-4579. doi: 10.1073/pnas.87.12.4576