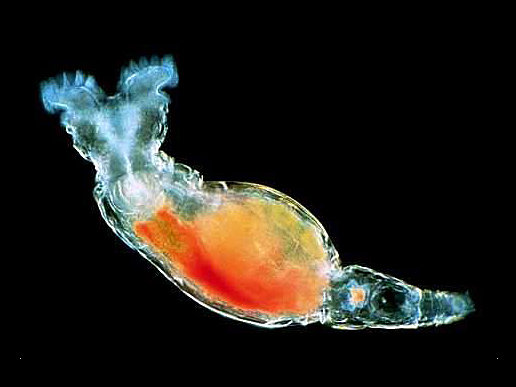
Bdelloid rotifer
Classification
Domain: Eukaryota
Kingdom: Animalia
Phylum: Rotifera
Superclass: Eurotaria
Class
Bdelloidea are a class of rotifers. There are over 450 species of bdelloid rotifers distinguished by their morphology. This includes species such as Adineta ricciae, Rotaria rotatoria, and Philodina acuticornis. Bdelloidea's species concept is based on molecular and morphological data due to their asexual nature.
Description and Significance
Appearance:
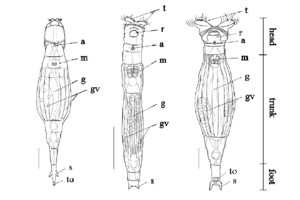
Bdelloid rotifers are microscopic worm-like organisms that are usually between 150 to 750 µm long. Their bodies are made up of three main regions: head, trunk, and foot (seen in Figure 2). Bdelloids have a well developed corona that is divided into two parts on a retractable head. Bdelloid rotifers can only be identified by eye while they are alive in order to see how they feed and crawl, which are their defining characteristics for classification. Through a weak hand lens, bdelloid rotifers appear as tiny white dots (Fontaneto et. al., 2004).
Habitat:
Bdelloidea can be found in fresh and brackish water all over the world. They can also be found in moss, lichen, and soil while also being able to survive dry, harsh environments through desiccation-induced dormancy, also known as anhydrobiosis. Bdelloid rotifers are considered to be cosmopolitan (Ricci et. al., 2006).
Significance:
Bdelloid rotifers are the oldest and most diverse asexuals to be discovered, surviving for over 80 millions years. Besides reproducing asexually, bdelloid rotifers are all female therefore reproducing through parthenogenesis. Bdelloidea defy biologist's ideas surrounding the centrality of sex by creating genetic diversity asexually. Daughter bdelloids inherit both copies of each gene from their mother, but these genes never intermix. This allows the genes to remain distinct and evolve to take on new roles regardless of their partner gene's destiny. Researcher's believe that these two-for-one gene helps bdelloids compensate for the loss of diversity that typically occurs without sex (Yong 2009).
Genome Structure
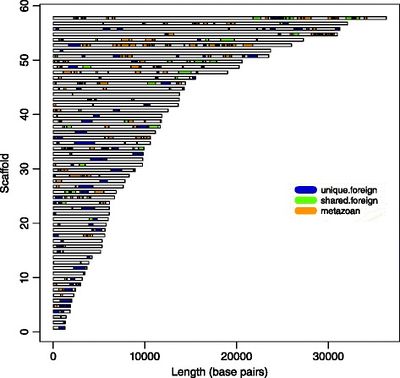
Bdelloid rotifers are found to have colinear chromosomes, with a mitochondrial genome sequence of 15,319 bp for R. rotatoria (Min 2009).
Horizontal gene transfer is rare in multi-cellular eukaryotes, but it has been found that bdelloid rotifers contain a high proportion of horizontally transferred, non-metazoan genes. The bdelloid rotifer incorporates foreign DNA from fungi, plants, and bacteria creating a mosaic of DNA (seen in Figure 3). The foreign DNA is incorporated when repairing their chromosomes from double strand breaks. The efficient repairing of double strand breaks allows the bdelloid rotifers to be resistant from ionization radiation. HGT is higher in species that desiccate more frequently in order to repair the double strand breaks in their chromosomes (Eyers 2015).
Lea Gene:
Evidence of the two-for-one gene was found in the lea gene from the bdelloid Adineta ricciae. The sequence of the two copies of the lea gene differ by 14%. This difference allows for translations of different structures and functions for LEA proteins. Both LEA proteins protect the bdelloid when it is in its desiccated-dormant state. Each version of the protein complements each other. One version acts as a molecular shield to prevent the bdelloid's sensitive proteins from balling into useless clumps, while the other version of the protein inserts itself into the fatty membrane surrounding the cell for stability (Yong 2009).
Metabolism and Life Cycle
Bdelloid rotifers are mostly organotrophs that feed by filtering or scraping bacteria, algae, or yeast from their environment. The species Abrochtha carnivora is the exception by feeding on other rotifers.
Under laboratory conditions bdelloid rotifers can survive for up to 30 days while laying 30-40 eggs. Their lifespan in nature is unknown. In response to stress, bdelloids will enter anhydrobiosis, a dormant state induced by drought in which an organism becomes almost completely dehydrated and reduces its metabolic activity to an imperceptible level. The bdelloid rotifer will resume metabolic activity when the conditons become suitable again (Fontaneto 2004).
Dormancy is a blind area for bdelloidea life because they resume their life without considering the time spent dormant. It has been shown that bdelloid rotifers benefit from dormancy with increase fecundity and fitness. The longest recorded bdelloid dormancy was 9 years (Ricci 2006).
Ecology
As stated above, bdelloid rotifers can be collected in fresh and brackish waters including lichen and moss. During extreme conditons such as droughts, bdelloid rotifers desiccate and enter a dormant state, anhydrobiosis. Desiccation of bdelloid rotifers allows for easy dispersal of the microorganism since it can easily be transported by the wind. Bdelloidea are able to outlive their predators and pathogens that are unable to survive desiccation. Therefore enabling bdelloid rotifers to persist without any major challenges (Fontaneto et. al., 2004).
There have been few bdelloidea symbiosises documented besides the mutualistic service-resource relationship between the bdelloid rotifers P. roseola and A. bengalensis in its embryonic stage. In this association, P. roseola feeds on the jelly coat of the A. bengalensis eggs and in return it enables the larvae of the ectoparasite to hatch out. This creates a furrow through the envelopes of the dejellied eggs, therefore contributing and benefiting from the relationship (Banerjee et. al., 2016).
Bdelloid rotifers are known as "ancient asexuals" due to them being the world's oldest and most diverse species to produce asexually. They are able to forego sex and obtain diversity by allowing the copies of their genes to evolve independently of each other (Yong 20009). Horizontal gene transfer also creates genetic diversity amongst bdelloid rotifers by incorporating foreign DNA when repairing double strand breaks caused by desiccation. The efficient double strand break repairs also allow for bdelloid rotifers to resistant ionization radiation damage (Eyers et. al., 2015).
References
Author
Page authored by Larynn Hall, student of Prof. Jay Lennon at IndianaUniversity.