Bovine spongiform encephalopathy
By Kevin Pan
Overview
By Kevin Pan
Transmissible spongiform encephalopathies are known as prion diseases that are progressive neurological disorders. This group of diseases includes Kuru, which is a disease found among New Guinea natives, Gerstmann-Straussler-Scheinker disease and bovine spongiform encephalopathy. Bovine spongiform encephalopathy is a progressive neurological disorder in cows that arose in the 1980’s. Humans can be infected with this disease after eating products that have been tainted with this disease. While the exact method of transfer is still not understood, the disease is not believed to be a product of bacteria, viruses, parasites and fungus (bseinfo) It is believed that a misfolded protein known as the prion protein in cattle is the cause of transfer. Prion proteins are commonly found throughout the body, but they can be modified if there is an accumulation of the modified prion proteins Eventually, a high accumulation of these protein can lead to damaged and disease. A major issue with this protein is that it is difficult to kill as it is able to survive heat, UV light, ionizing radiation and normal sterilization techniques (USDA 2015). The prion is also partially resistant to Proteinase K, which is a type of amino acid breakdown. This leads scientists to believe that the prion is not destroyed in the gastrointestinal tract (Campbell 2006).
It is believed that the variant Creutzfeldt-Jakob disease, which is the human form of BSE, is caused by BSE. However, studies have shown a similarity between prions of BSE and TSE, but not a directly correlation to support that theory2. Symptoms of CJD are dementia, memory loss, personality changes and hallucinations.
Mad Cow Disease was first found in cattle in England in 1986 and thousands of cattle died due to the disease. Heavy restrictions were placed on cattle export throughout the world, with heavy restrictions occurring in Europe. The USDA implemented stringent measures, banning products from nations where BSE was found. Between 1985-95, it is believed that almost 1 million cattle have been infected in the UKK and about ¾ of those were fed to humans (Campbell 2006). This ban on UK beef was removed in May 2006.
While the disease has appeared to be almost eradicated, it is still relevant to current research. Recent studies have shown a link between bovine prion disease and Alzheimer’s disease.
Prion Diseases
A prion is a type of protein that triggers normal proteins in the brain to fold abnormally, which affect the nervous system in humans and animals and can cause numerous afflictions (Prion Diseases Johns Hopkins). One of the earliest known prion diseases is called “scrapie,” which has been around since 1732. This degenerative disease affects the nervous systems of sheep and goats and can e characterized by animals scraping off their fur or wool. Other symptoms of the disease are changes in behavior, tremors and locked muscles and a loss of movement. Scrapie affects sheep between the ages of three to five years old and once infected, the animals generally die within 2 weeks to 6 months of onset. Dissections of afflicted sheep indicate the proteins’ presence throughout the nervous system and various important organs. Healthy mammals have normal prion proteins (PrPC), which have amino acid sequences that are highly conserved in mammalian species. This protein consists of 254 amino acids, which includes a signal peptide at the N-terminus, as well as the c-terminus. The protein also includes two asparagines for nitrogen glycosylation (Hagiwara 2013). Unlike other infectious agents, prions are unique in that they do not have nucleic acids (Korth et al 2001). Comparing the two proteins with each other can elicit several differences. Firstly, it was shown through infrared spectroscopy that the normal prion protein consists of roughly 40% α-helixes, whereas PrPSc was shown to have a higher β-sheet (54%) content than α-helices (21%) content. It is believed that this high β-sheet content is what makes the misfolded proteins pathogenic (Prusiner 1997). It was found that human prion diseases are closely related with two types of scrapie prion proteins that can be detected using a Western blot based on the size of PrPSc (Gambetti et al 2003).
The symptoms that are associated with scrapie (figure 2) are also commonly associated with other prion diseases. Common symptoms of these other diseases include impaired brain function, memory changes, personality changes and behavioral changes. These conditions commonly arise in adulthood and cause a very rapid deterioration (Genetics Home reference). These disorders are very rare affect about one person per million worldwide—The united States has had relatively low amounts of reported cases with roughly 350 cases reported per year.
The most common type of Prion Diseases are Creutzfeldt-Jakob disease (CJD), Gerstmann-Sträussler-Scheinker syndrome (GSS), and fatal familial insomnia (FFI). 10-15% of these disorders are caused by mutations to the PRNP gene on chromosome 20, which encode the prion protein (PrPC). The other 85-90% of cases are classified as sporadic or acquired. The actual function of the prion protein is unknown, but researchers believe that this protein is responsible for transport of copper into cells, protect the brain cells from injury and communicate between neurons (Prion Disease 2014). Mutations to this gene form produce an abnormally shaped protein called PrPSc, which binds to PrPc. This binding to normal protein forms protein-protein interactions and recruits more PrPC into the mutated form. A build-up of this abnormal protein leads to clumping in the brain, which forms sponge-like holes in the brain (Prion Disease 2014).
Analysis of people affected with variant Creutzfeldt-Jackob disease has shown that affected individuals were homozygous for methionine at PRNP codon 129. In a study conducted by Asante et al 2002, they were able to show that mice homozygous for methionine at 129 acquired the prion disease after inoculation with BSE. Amongst the Caucasian population, roughly 50% of the individuals are homozygous for M, about 35% are heterozygous for MV and the rest are homozygous for valine.
Prion diseases that are termed acquired prion diseases are due to exposure to PrPSc causing the disease. An example of an acquired disease is Variant Creutzfeldt-Jakob disease, which can be obtained from eating cows that have been afflicted with BSE.

Prion Structure
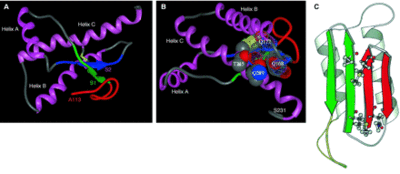
Prions are commonly found in other organisms and play a role in nonmammalisn organisms such as yeast and fungi. In Saccharomyces cerevisiae, the prions are composed, respectively, of Sup35p, a translation termination factor, and Ure2p, a transcriptional regulator involved in nitrogen metabolism. In a study done by Nelson et al (2005), they utilized X-ray crystallographic analysis to study amyloid polymers of a peptide derived from the N-terminal of Sup35p. The analysis showed that the structure consists of a pair of pair of β sheets running parallel to the fiber axis and are held together by hydrogen bonds. This structure differs in many ways from mammalian prions. Harris et al (2006) studied the mammalian prion structure through the use of NMR spectroscopy and X-ray crystallographic analysis. They found that PrPC consists of a N-terminal region encoded by the residue region between 23-142 and a C-terminal region encoded by the residues 125-228. Further analysis showed that the C-terminal is up of three α-helices and two short β-strands. Hagiwara et al (2012) also showed that the N-terminus is supposed to be flexible in a normal prion protein. Prusiner (1997) believes it is the NH2 terminal where PrPSc binds to PrPC. The structure of PrPSc is a little more difficult to determine. The tertiary structure of PrPSc can be difficult to determine due to the tendency of these proteins to aggregate together. Models of PrPSc indicate that the main cause of the prion diseases is the refolding of the NH2 terminal helices into β-sheets.