Buchnera aphidicola: Difference between revisions
| Line 32: | Line 32: | ||
[[File:3137594 pone 0021944 g002.png|thumb|left|In situ hybridization of aphids is represented and "Buchnera aphidicola" is fluoresced in green[http://openi.nlm.nih.gov/detailedresult.php?img=3137594_pone.0021944.g002&query=buchnera aphidicola&fields=all&favor=none&it=none&sub=none&uniq=0&sp=none&req=4&npos=6&prt=]]] | [[File:3137594 pone 0021944 g002.png|thumb|left|In situ hybridization of aphids is represented and "Buchnera aphidicola" is fluoresced in green [http://openi.nlm.nih.gov/detailedresult.php?img=3137594_pone.0021944.g002&query=buchnera aphidicola&fields=all&favor=none&it=none&sub=none&uniq=0&sp=none&req=4&npos=6&prt=]]] | ||
===Pathogenicity=== | ===Pathogenicity=== | ||
Revision as of 04:32, 5 December 2012
A Microbial Biorealm page on the genus Buchnera aphidicola
Classification
Higher order taxa:
Kingdom: Bacteria Phylum: Proteobacteria Class: Gamma Proteobacteria Order: Enterobacteriales Family: Enterobacteriaceae Genus: Buchnera
Species
Buchnera aphidicola (4)
Description and Significance
Buchnera aphidicola are prokaryotes within the phylum gamma-Proteobacteria, where a great diversity of bacteria can be found (1). Proteobacteria are all Gram-negative bacteria, and contain various pathogenic bacteria and various nitrogen fixing bacteria (1). Buchnera aphidicola is a primary endosymbiont of aphids (A. pisum), a type of insect that digests plants (2). Researchers argue that the ancestors of Buchnera aphidicola were free living Gram-negative bacteria similar to a modern Enterobacteriaceae such as Escherichia coli (2). Various evidence supports that Buchnera aphidicola are closely related to Enterobacteriaceae family, such as studies using 16S rRNA sequence comparisons, which show that the two families share a closely related 16S rRNA structure (4). The size of Buchnera aphidicola is 3 µm in diameter and they share the most of the key components of their Gram-negative cell wall with related Enterobacteriaceae (3). However, unlike most other Gram-negative bacteria, Buchnera aphidicola do not have the genes to produce lipopolysaccharides (LPS) for their outer cell membrane(3). Since Buchnera aphidicola are closely related to some well known organisms such as Escherichia coli and Haemophilus influenzae, the study of Buchnera aphidicola can reveal the evolutionary mechanisms as well as molecular basis of intracellular endosymbiosis and the parasitic behavior of symbionts (4).
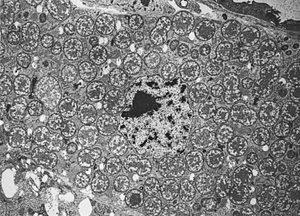
Genome Structure
Buchnera aphidicola have a very small genome (<1 Mb) compared to other living bacteria (6). As most Proteobacteria do, Buchnera aphidicola has one circular chromosome with few numbers of plasmids, two in most cases (1). Verification of the phylogeny of Buchnera aphidicola was done by comparing the genome sequence with various species in the family called Enterobacteriaceae, the bacteria researchers believe to be most closely related to Buchnera, and thought to be diverged from each other most recently (3). By comparing the genomes between Buchnera and general species of Enterobacteriaceae, one can find the genomic difference between symbiotic bacteria and free-living bacteria (4). There are several distinct features which Buchnera aphidicola hold. The ratio of G+C to A+T in their chromosome is fairly high due to the higher GC nucleotide substitution rates that have occurred since the divergence of the species(5). Furthermore, it is also one of the most genetically stable organism due to the lack of many enzymes and proteins involved in metabolism due to the symbiotic relationship with aphids (3). Besides a nutritional co-dependence with aphids, Buchnera have lost the ability to produce cell surface components such as lipopolysaccharides, which are a common characteristic of gram-negative bacteria. This is partly due to co-existing for millions of years(4). Lastly, Buchnera aphidicola have lost many genes that are involved in nutrient synthesis through deletions in their chromosomes(3). Researchers have found that the majority of genome reduction began shortly after the symbiosis between Buchnera and the aphids initiated, however before aphids began to diversify(7).
Cell Structure and Metabolism
Buchnera aphidicola does not have many metabolic enzymes which normal Gram-negative bacteria have. The symbiotic relationship with aphids and the lack of chromosome crossover events due to vertical transmission of genetic materials within its species have caused the deletion of genes required for various metabolism (3). Researchers have identified those enzymes, and they are found to be essential for anaerobic respiration, the synthesis of amino-sugars, fatty acids, phospholipids, and complex carbohydrates (3). Buchnera aphidicola also have lost regulatory factors allowing continuous overproduction of tryptophan and other amino acids (3). This loss of genes are thought to be the result of symbiotic relationship with aphids, as they share key nutrients together. It is not known how the aphids communicate with Buchnera what or how much nutrition it needs, but researchers have hypothesized a few things. They believe that it could be regulated by the amount of nitrogen and carbon substrates that the host produces (6). The symbiosis not only reduced the genome involved in metabolism but also reduced the genome which codes lipopolysaccharides (LPS), the key component of the cell walls of Gram-negative bacteria (4). The lack of lipopolysaccharides resulted in non-pathogenicity of Buchnera aphidicola(1).
Ecology
The symbiotic relationship of Buchnera aphidicola with aphids began between 200 million and 150 million years ago, according to the study of genome sequences and phylogeny within closely related species and aphids (2). Buchnera aphidicola have persisted through maternal transmission and co-speciation, with very few lateral gene transfer, which suppress the diversity of their genome (2). Aphids have developed bacteriocyte cells to host Buchnera aphidicola (2). It is estimated that a fully grown aphid may host 5.6 × 10^6 Buchnera aphidicola cells (2). Each bacteriocyte contains many vesicles called symbiosomes, which are derived from the plasma membrane. (2) Most of the aphids contain maternally transmitted bacteriocyte cells, which themselves contain Buchnera aphidicola, explaining the nature of vertical gene transfer within Buchnera themselves. The aphids live on a restricted diet (plant sap), rich in carbohydrates, but poor in nitrogenous or other essential compounds (4). Their mutualistic relationship is beneficial to the both of them.The Buchnera provide amino acids to the aphid, while the aphid provides itself as a host to the Buchnera (8). It is believed that the Buchnera provide the essential nutrients that the host lacks, by keeping the enzymes necessary to synthesize those essential nutrients (4). In return, Buchnera lost other key metabolic enzymes because they are dependent on aphids to provide those nutrients (4). Furthermore, Buchnera have lost the ability to produce cell surface components such as lipopolysaccharides, which forces them to keep the endosymbiont relationship with aphids (4).
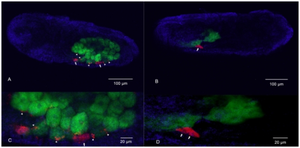
Pathogenicity
Proteobacteria contain various pathogenic bacteria such as Escherichia, Salmonella, Vibrio, and Helicobacter (1). But Buchnera aphidicola lacks lipopolysaccharides in their cell walls, which leads to a reduced-pathogenicity (4). There are no known pathogenic events found due to Buchnera aphidicola.
Application to Biotechnology
There aren’t any known applications to biotechnology involving Buchnera aphidicola, but the study of Buchnera aphidicola can result in various progressions in the field of biotechnology. For example, the study of Buchnera aphidicola and other Enterobacteriales can reveal key enzymes and proteins that are required for symbiosis (4). One can then artificially produce symbiotic bacteria and study various metabolic enzymes with knock-ins and knock-outs of specific genes.
Current Research
Buchnera aphidicola was first named by Paul Baumann and his graduate student, as well as the first molecular characterization of a symbiotic bacterium using Buchnera aphidicola (2). The studies on Buchnera aphidicola later contributed to many studies on symbionts of many types of insects, pursued by numerous researchers like Paul and Linda Baumann, Nancy Moran, Serap Aksoy, Roy Gross, those who studied symbionts of aphids, tsetse flies, ants, leafhoppers, mealybugs, whiteflies, psyllids, etc. (2)
References
1. Madigan M; Martinko J (editors). (2005). Brock Biology of Microorganisms, 11th ed., Prentice Hall. ISBN 0131443291
2. Douglas, A E (1998). "Nutritional interactions in insect-microbial symbioses: Aphids and their symbiotic bacteria Buchnera". Annual Review of Entomology 43: 17-38. ISSN 00664170.
3. Pérez-Brocal V, Gil R, Ramos S, Lamelas A, Postigo M, Michelena J, Silva F, Moya A, Latorre A (2006). "A small microbial genome: the end of a long symbiotic relationship?". Science 314 (5797): 312-3. PMID 17038625.
4. MUNSON (M.A.), BAUMANN (P.) and KINSEY (M.G.): Buchnera gen. nov. and Buchnera aphidicola sp. nov., a taxon consisting of the mycetocyte-associated, primary endosymbionts of aphids. Int. J. Syst. Bacteriol., 1991, 41, 566-568.
5. Gómez-Valero L, Silva FJ, Christophe Simon J, Latorre A. “Genome reduction of the aphid endosymbiont Buchnera aphidicola in a recent evolutionary time scale”. Institut Cavanilles de Biodiversitat i Biologia Evolutiva and Departament de Genètica, Universitat de València, Apartat 22085, 46071 Valencia, Spain
6. Thomas, G H, Zucker, J, Macdonald, S J, Sorokin, A, Goryanin, I, Douglas A E."A fragile megabollic network adapted for cooperation in the symbiotic bacterium Buchnera aphidicola." BMC Systems Biology 3:24, 2009.
7. Roeland, C H J, Kamerbeek, J, Palacios, C, Rausell, C, Abascal, F, Bastolla, U, Fernandez, J, Jimenez, L, Postigo, M, Silva, F J, Tamames J, Viguera, E, Latorre, A, Valencia, A, Moran, F, Moya, A."Reductive genome evolution in Buchnera aphidicola." Centro de Astrobiologia. Spain: Madrid, 2002.
8. Gil, R, Sabater-Munoz, B, Perez-Brocal, V, Silva F J, Latorre, A. 2006."Plasmids in the Aphid Endosymbiont Buchnera aphidicola with the smallest genomes." Gene 370:17-25. 2006. Edited by Brittany Mihalcoe student of Grace Lim-Fong Edited by Kyu Hahn student of Rachel Larsen and Kit Pogliano
kmg
