Copper Mining Using Acidothiobacillus: Difference between revisions
GipsonAndrew (talk | contribs) |
GipsonAndrew (talk | contribs) |
||
| Line 19: | Line 19: | ||
===Abiotic Solubilization and Regeneration of Bacterial Substrates=== | ===Abiotic Solubilization and Regeneration of Bacterial Substrates=== | ||
Copper sulfide ores are solubilized in a reaction where ferric iron complexed with sulfate attacks the copper compounds and becomes reduced to ferrous iron while generating copper sulfates and elemental sulfur (Hutchins <i>et al.</i> 1986): | Copper sulfide ores are solubilized in a reaction where ferric iron complexed with sulfate attacks the copper compounds and becomes reduced to ferrous iron while generating copper sulfates and elemental sulfur (Hutchins <i>et al.</i> 1986): | ||
<center>Cu<sub>2</sub>S (chalcocite), CuS (covellite), CuFeS<sub>2</sub> (chalcopyrite), Cu<sub>5</sub>FeS<sub>4</sub> (bornite) + Fe<sub>2</sub>(SO<sub></sub>)<sub>3</sub> --> CuSO<sub>4</sub> + FeSO<sub>4</sub> + S<sup>o</sup> [unbalanced equation]</center><br> | <center>Cu<sub>2</sub>S (chalcocite), CuS (covellite), CuFeS<sub>2</sub> (chalcopyrite), Cu<sub>5</sub>FeS<sub>4</sub> (bornite) + Fe<sub>2</sub>(SO<sub>4</sub>)<sub>3</sub> --> CuSO<sub>4</sub> + FeSO<sub>4</sub> + S<sup>o</sup> [unbalanced equation]</center><br> | ||
Copper oxides like azurite are somewhat less susceptible to attack by ferric iron; however, they are highly soluble in the low-pH solution produced by the bacterial production of sulfuric acid (Iasillo 1999):<br> | |||
<center>Cu<sub>3</sub>(OH)<sub>2</sub>(CO<sub>3</sub>)<sub>4</sub> (azurite) + 3H<sub>2</sub>O --> 3CuSO<sub>4</sub> + CO<sub>2</sub> + 4H<sub>2</sub>O</center><br> | |||
<center></center><br2 | |||
<center></center><br> | |||
==Section 3== | ==Section 3== | ||
Revision as of 03:27, 26 April 2011

Introduction
By Andrew Gipson
Introduce the topic of your paper. What microorganisms are of interest? Habitat? Applications for medicine and/or environment?
Section 1
Include some current research, with at least one figure showing data.
Mechanism of Bacterial Leaching
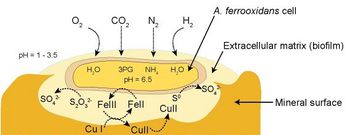
There has been much debate over how At. ferrooxidans contributes to the conversion of metal ores into soluble compounds. Although it has been proposed that the bacterium produces enzymes which directly mediate the reaction, studies have shown that iron is the only metal ion used to produce energy (Mignone and Donati 2004). Copper is not a metabolite of At. ferrooxidans, so the bacteria is not directly responsible for the solubilization of copper. Rather, it occurs through an abiotic reaction between the copper ores and the ferric ions and protons produced by the metabolic activities of the bacteria. These reactions regenerate the reduced iron and sulfur compounds that the bacteria reuptake to start the cycle anew.
Bacterial Metabolic Reactions
At. ferrooxidans has a diverse set of metabolic processes that allow it to be lithoautotrophic, but the most economically important activity this bacterium performs is the oxidation of ferrous iron (Fe2+) to ferric iron (Fe3+):
...and the oxidation of elemental sulfur (So) to sulfuric acid (H2SO4):
Abiotic Solubilization and Regeneration of Bacterial Substrates
Copper sulfide ores are solubilized in a reaction where ferric iron complexed with sulfate attacks the copper compounds and becomes reduced to ferrous iron while generating copper sulfates and elemental sulfur (Hutchins et al. 1986):
Copper oxides like azurite are somewhat less susceptible to attack by ferric iron; however, they are highly soluble in the low-pH solution produced by the bacterial production of sulfuric acid (Iasillo 1999):
<br2
Section 3
Include some current research, with at least one figure showing data.
Conclusion
Overall text length at least 3,000 words, with at least 3 figures.
References
Iasillo E (1999) Metallurgical test program for copper leaching projects. In Copper Leaching, Extraction, and Electrowinning Techniques, ed. Jergensen GV. 124-128. Littleton: SME.
Leduc LG and Ferroni GD (1994) The chemolithotrophic bacterium Thiobacillus ferrooxidans. FEMS Microbiology Reviews 14:103-120.
Mignone CF and Donati ER (2004)ATP requirements for growth and maintenance of iron-oxidizing bacteria. Biochemical Engineering Journal 18: 211–216.
Rawlings DE (2002) Heavy metal mining using microbes. Annu. Rev. Microbiol. 56: 65-91.
Valdés J, Pedroso I, Quatrini R, Dodson RJ,Tettelin H, Blake R, Eisen JA, Holmes DS (2008) Acidithiobacillus ferrooxidans metabolism: from genome sequence to industrial applications. BMC Genomics 9: 597.
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2011, Kenyon College.