Enzymatic decomposition of plant litter: Difference between revisions
Tim.philpott (talk | contribs) No edit summary |
Tim.philpott (talk | contribs) |
||
| Line 6: | Line 6: | ||
===Cellulose=== | ===Cellulose=== | ||
[[Cellulose]] is found primarily in the cell wall of plants (Figure 1), and is the most abundant biopolymer found on earth. It consists of [[D-glucose]] units linked by [[glycosidic bond| β-1,4-glycosidic bonds]], forming a linear polymer<ref name=Baldrian>{{cite book|last=Baldrian|first=P|title=Ecology of Saprotrophic Basidiomycetes|year=2008|publisher=Elsevier|location=San Francisco|pages=211–238}}</ref> . Cellulose becomes [[crystalline]] via [[hydrogen bonding]] and [[van der Waal forces]] between cellulose chains (fibrils)<ref name=Kogel>{{cite journal|last=Kogel-Kranber|first=Ingrid|title=The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter|journal=Soil Biology and Biochemistry|year=2002|volume=34|issue=2|pages=139–162|doi=10.1016/S0038-0717(01)00158-4}}</ref>,<ref name=Lynd>{{cite journal|last=Lynd|first=L|coauthors=Weimer PJ, van Zyl WH, and Pretorius IS|title=Microbial cellulose utilization: fundamentals and biotechnology|journal=Microbial cellulose utilization: fundamentals and biotechnology|year=2002|volume=66|issue=3|pages=506–577}}</ref>. Cellulose chains bound together are termed microfibrils. Some regions are less ordered and are termed amorphous regions.[[File:Fig 1.jpeg | [[Cellulose]] is found primarily in the cell wall of plants (Figure 1), and is the most abundant biopolymer found on earth. It consists of [[D-glucose]] units linked by [[glycosidic bond| β-1,4-glycosidic bonds]], forming a linear polymer<ref name=Baldrian>{{cite book|last=Baldrian|first=P|title=Ecology of Saprotrophic Basidiomycetes|year=2008|publisher=Elsevier|location=San Francisco|pages=211–238}}</ref> . Cellulose becomes [[crystalline]] via [[hydrogen bonding]] and [[van der Waal forces]] between cellulose chains (fibrils)<ref name=Kogel>{{cite journal|last=Kogel-Kranber|first=Ingrid|title=The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter|journal=Soil Biology and Biochemistry|year=2002|volume=34|issue=2|pages=139–162|doi=10.1016/S0038-0717(01)00158-4}}</ref>,<ref name=Lynd>{{cite journal|last=Lynd|first=L|coauthors=Weimer PJ, van Zyl WH, and Pretorius IS|title=Microbial cellulose utilization: fundamentals and biotechnology|journal=Microbial cellulose utilization: fundamentals and biotechnology|year=2002|volume=66|issue=3|pages=506–577}}</ref>. Cellulose chains bound together are termed microfibrils. Some regions are less ordered and are termed amorphous regions.[[File:Fig 1.jpeg |Figure 1 a) Arrangement of fibrils, microfibrils and cellulose in plant cell walls (source: Moore R, Clark D, Vodopich D. 1998. Botany Visual Resource Library, McGraw-Hill companies). b) Structure of plant cell wall (source: McCann and Roberts 1991, in Scheller and Ulvskov 2010)]] | ||
===Hemicellulose=== | ===Hemicellulose=== | ||
| Line 12: | Line 12: | ||
===Lignin=== | ===Lignin=== | ||
[[Lignin]] (Figure 2) is an amorphous polymer found in the cell walls of vascular plants that fills the voids between cellulose microfibrils, reinforcing the cell wall and protecting against microbial biodegradation<ref name=Kogel /> . It is a branched polymer of three [[aromatic]] precursors – p-coumaryl alcohol, coniferyl alcohol and sinapyl alcohol, which polymerize via carbon-carbon and ether bonds<ref name=Baldrian /> . Lignin, after cellulose, is the second most abundant biopolymer and is very resistant to [[mineralization]], largely due to its amorphous structure. | [[Lignin]] (Figure 2) is an amorphous polymer found in the cell walls of vascular plants that fills the voids between cellulose microfibrils, reinforcing the cell wall and protecting against microbial biodegradation<ref name=Kogel /> . It is a branched polymer of three [[aromatic]] precursors – p-coumaryl alcohol, coniferyl alcohol and sinapyl alcohol, which polymerize via carbon-carbon and ether bonds<ref name=Baldrian /> . Lignin, after cellulose, is the second most abundant biopolymer and is very resistant to [[mineralization]], largely due to its amorphous structure. | ||
==Organisms involved in plant litter decomposition== | ==Organisms involved in plant litter decomposition== | ||
Revision as of 01:38, 26 November 2013
Enzymatic decomposition of plant litter
Plant litter serves as the primary carbon source for terrestrial heterotrophic organisms. Microorganisms excrete enzymes that decompose the biopolymers found in plant litter, returning fixed carbon to the atmosphere through respiration, controlling the formation of soil organic matter, and influencing biogeochemical cycles. Additionally, the microbial mechanisms involved in biopolymer decomposition have important applications for biofuels such as cellulosic ethanol.
Major biopolymers in plant litter
Plant tissues consist of diverse compounds including intracellular components (proteins, starch, chlorophyll) and structural cell wall biopolymers (cellulose, hemicellulose, pectin, lignin). Non-structural components of plant litter are considered labile, whereas biopolymers require specialized enzymatic systems for decomposition.
Cellulose
Cellulose is found primarily in the cell wall of plants (Figure 1), and is the most abundant biopolymer found on earth. It consists of D-glucose units linked by β-1,4-glycosidic bonds, forming a linear polymer[1] . Cellulose becomes crystalline via hydrogen bonding and van der Waal forces between cellulose chains (fibrils)[2],[3]. Cellulose chains bound together are termed microfibrils. Some regions are less ordered and are termed amorphous regions.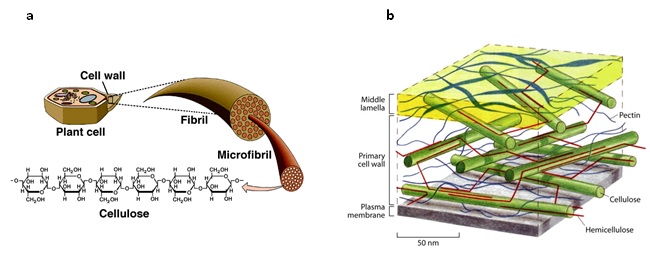
Hemicellulose
Hemicellulose refers to non-cellulosic polysaccharides that differ from cellulose in their sugar backbone, side chains, and branching. Hemicelluloses are bound together with glycosidic linkages but are more branched and shorter than cellulose[2]. The hemicelluloses in wood are mainly xylans (consisting of D-xylose units) and glucomannans (consisting of D-glucose and D-mannose units)[1] . Hemicelluloses tether cellulose microfibrils through cross-linkage via hydrogen bonding and by being trapped in the microfibril during cellulose biosynthesis, conferring further structural integrity to plant cell walls[4] .
Lignin
Lignin (Figure 2) is an amorphous polymer found in the cell walls of vascular plants that fills the voids between cellulose microfibrils, reinforcing the cell wall and protecting against microbial biodegradation[2] . It is a branched polymer of three aromatic precursors – p-coumaryl alcohol, coniferyl alcohol and sinapyl alcohol, which polymerize via carbon-carbon and ether bonds[1] . Lignin, after cellulose, is the second most abundant biopolymer and is very resistant to mineralization, largely due to its amorphous structure.
Organisms involved in plant litter decomposition
The ability to decompose cellulose (and hemicellulose) is widespread in microorganisms, with cellulolytic activity among the eubacteria concentrated in the Actinobacteria and Firmicutes phyla, and cellulolytic activity within the fungi distributed across the entire kingdom[3] . Fungi are better adapted to cellulose degradation due to their filamentous growth form (hyphae). However, the cellulosome is a bacterial adaptation allowing adhesion to cellulose, resulting in efficient cellulolysis.
Lignin degradation is predominately carried out by fungi because their mycelia directly contact lignocellulose and they have more diverse enzymatic capabilities. Fungal decomposition of plant biopolymers is classified in three categories; soft, white and brown rot, determined by their relative degradation of the biopolymers[5] . Soft rot fungi decompose cellulose and hemicellulose, but lignin is not appreciably decomposed and decay is often localized. Brown rot is similar to soft rot, but lignin is only modified and hemicelluloses and cellulose are selectively removed, leaving behind a brown residue consisting of oxidized lignin. White rot fungi are able to decompose all plant cell wall biopolymers[6].
Enzymatic decomposition of cellulose
Cellulose is decomposed via a cellulase enzymatic system composed of three enzyme groups (i) endoglucanases (ii) exoglucanases and (iii) β-glucosidases (Figure 3). Endoglucanases hydrolyze D-glucan units at random in the amorphous regions of the microfibril, producing cellulose oligosaccharides with exposed chain ends. Exoglucanases hydrolyze β-1,4-glycosidic bonds processively at reducing or non-reducing chain ends, producing cellobiose or glucose. Cellobiose is further broken down by cell wall bound or intracellular β-glucosidases, while glucose is taken up directly by the cell[1] [3].
Enzymatic decomposition of hemicellulose
Being a heterogeneous polysaccharide, hemicellulose decomposition is more complex relative to cellulose decomposition, but the general process involves degradation to monomeric sugars and acetic acid[7]. Glucuronoxylans are common hemicelluloses and four enzymes are required for their degradation (endoxylanase, acetylxylan esterase, α-glucuronidase and β-xylosidase) (Figure 4). Glucoronoxylan consists of a xylan backbone with acetyl and glucuronic acid side chains. Endoxylanase cleaves the xylan backbone into xylan oligosaccharides, acetylxylan esterase removes acetyl groups and α-glucuronidase removes glucoronic acid groups. Finally, β-xylosidase produces xylose from xylan, which is taken up by the cell.
Enzymatic decomposition of lignin
Lignin decomposition involves a series of enzymes that oxidize phenolic and non-phenolic lignin units. The first step in lignin decomposition produces aromatic radicals by oxidizing the lignin polymer. Laccases and lignin peroxidases (e.g. lignin peroxidase, manganese peroxidase) are extracellular enzymes involved in this first step. Laccases directly oxidize phenolic lignin units using molecular oxygen. However, phenolic lignin units comprise less than 10% of the lignin polymer, with non-phenolic units making up to 90% of the polymer. Lignin peroxidase (LiP) degrades non-phenolic lignin units by first being oxidized by hydrogen peroxide (H2O2), then oxidizing aromatic nuclei of soluble lignin units. LiP can be recycled through oxidation by H2O2. The oxidized aromatic radical species are further involved in non-enzymatic radical reactions resulting in polymer cleavages[1] [8]. Manganese peroxidase (MnP) oxidizes Mn2+ to Mn3+, Mn2+ being a commonly available element in soils and lignocellulose[9]. Mn3+ is unstable on its own and is chelated with organic acids such as oxalate. The chelated Mn3+ acts as a non-specific diffusible oxidizer on several substrates within the lignin complex[9][10]. Both LiP and MnP consume H2O2, which is replenished through oxidases (e.g. aryl-alcohol oxidase, AAO, and aryl-alcohol dehydrogenase, AAD).
The enzymes listed above are involved in the coordinated decomposition of lignin, as illustrated in Figure 5. LiP and MnP generate aromatic radicals (1), which are then involved in non-enzymatic reactions such as demethoxylation (2), C4-ether breakdown (3), aromatic ring cleavage (4), and Cα-Cβ breakdown (5). Production of H2O2 for use in LiP and MnP reactions occurs via AAO and AAD (6,7), with aromatic aldehydes from (5) as a substrate. Four pathways are available for phenoxy radicals, the product of C4-ether breakdown (3): they can be reduced to phenolic compounds (8), they can re-polymerize on the lignin polymer (9), they can be re-oxidized by laccases, MnP or LiP (10), or they can undergo Cα-Cβ breakdown (11) to yield p-quinones. Quinones are involved in generating the hydroxyl radical (·OH) (12) via the fenton reaction. The hydroxyl radical is a powerful oxidizer that likely initiates lignin degradation[8]. Generally, this complex biochemistry results in simpler carbon compounds that are taken up by hyphae for anabolism.
References
Chockalingam, Evvie, and S. Subramanian. “Utility of Eucalyptus Tereticornis (Smith) Bark and Desulfotomaculum Nigrificans for the Remediation of Acid Mine Drainage.” Bioresource Technology 100, no. 2 (January 2009): 615–621. doi:10.1016/j.biortech.2008.07.004.
“Genus Desulfotomaculum - Hierarchy - The Taxonomicon.” Accessed November 5, 2013. http://taxonomicon.taxonomy.nl/TaxonTree.aspx?id=229.
Kaksonen, Anna H., Stefan Spring, Peter Schumann, Reiner M. Kroppenstedt, and Jaakko A. Puhakka. “Desulfotomaculum Thermosubterraneum Sp. Nov., a Thermophilic Sulfate-reducer Isolated from an Underground Mine Located in a Geothermally Active Area.” International Journal of Systematic and Evolutionary Microbiology 56, no. 11 (November 1, 2006): 2603–2608. doi:10.1099/ijs.0.64439-0.
Liu, Yitai, Tim M. Karnauchow, Ken F. Jarrell, David L. Balkwill, Gwendolyn R. Drake, David Ringelberg, Ronald Clarno, and David R. Boone. “Description of Two New Thermophilic Desulfotomaculum Spp., Desulfotomaculum Putei Sp. Nov., from a Deep Terrestrial Subsurface, and Desulfotomaculum Luciae Sp. Nov., from a Hot Spring.” International Journal of Systematic Bacteriology 47, no. 3 (July 1, 1997): 615–621. doi:10.1099/00207713-47-3-615.
Moser, Duane P, Thomas M Gihring, Fred J Brockman, James K Fredrickson, David L Balkwill, Michael E Dollhopf, Barbara Sherwood Lollar, et al. “Desulfotomaculum and Methanobacterium Spp. Dominate a 4- to 5-kilometer-deep Fault.” Applied and Environmental Microbiology 71, no. 12 (December 2005): 8773–8783. doi:10.1128/AEM.71.12.8773-8783.2005.
Ogg, Christopher D, and Bharat K C Patel. “Desulfotomaculum Varum Sp. Nov., a Moderately Thermophilic Sulfate-reducing Bacterium Isolated from a Microbial Mat Colonizing a Great Artesian Basin Bore Well Runoff Channel.” 3 Biotech 1, no. 3 (October 2011): 139–149. doi:10.1007/s13205-011-0017-5.
Pikuta, E, A Lysenko, N Suzina, G Osipov, B Kuznetsov, T Tourova, V Akimenko, and K Laurinavichius. “Desulfotomaculum Alkaliphilum Sp. Nov., a New Alkaliphilic, Moderately Thermophilic, Sulfate-reducing Bacterium.” International Journal of Systematic and Evolutionary Microbiology 50 Pt 1 (January 2000): 25–33.
