Eyes
Introduction
The eye, it has been said that the eyes are our window to the world. Of the five senses at our disposal, nothing more than the eyes have greatly infused us with an overwhelming abundance of the stimuli collected from the greater world. The eyes personify as the prevailing instrument that shapes our very individuality. Our perceptions, our interactions, our very definitions of who we are, stem from the visual cues received, processed and reflected back. Hence the eyes have been referred to as the window into our soul.
The evolution of the eyes dates back to the pre-Cambrian explosion when the first proto-eyes emerged. These simplest eyes used to detect light and darkness have evolved into wide ranging complexities and have emerged into myriad forms found today. From the compound eyes found in arthropods with its many facets permitting a wide ranging 360 degree field of vision, to the common single lens eyes that telescope and modulate to generate images, to eyes capable of registering light wavelength in the ultraviolet spectrum, the eyes today are all about the ever increasing information gathering, to provide a better edge and thus enhance survivability.
Our focus today looks not at the anatomy of the eye, but rather as an organ, the eye provide a niche for other organisms to explore, exploit and establish residence within this organ. Specifically, we will deal primarily with the human eye and the conditions it provides as the a suitable host. We will also examine some of the defense mechanisms employed by the human body to maintain law and order and thus the prevention of over infestation by undesirable colonization of micro organisms.
The Eye Niche
Physical Location of the Eyes
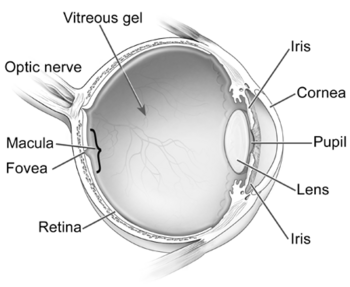
The human eye is located in the anterior portion of the cranium. The eyes are housed within the 7 bones that form the orbit of the eye; the frontal bone, the maxilla, the zygomatic bone, the lacrimal bone, the nasal bone (not part of orbit), the sphenoid bone, the ethmoid bone and the palatine bone.
Physical Conditions?
What are the conditions in your niche? Temperature, pressure, pH, moisture, etc.
Common bacteria found in Adjacent Communities
Ear: Staphylococcus (coagulase –neg.), Diptheroids, Pseudomonas spp., Enterobacteriaceae spp..
Nose: Staphylococcus (coagulase –neg.), S. aureus, Haemophilus spp., Viridians streptococci, S. pneumoniae
Oropharynx: Staphylococcus (coagulase –neg.), S. aureus, Haemophilus spp., Viridians streptococci, S. pneumonia, Veilonella spp., Prevotella spp., Fusobacterium spp., Candida spp., Moraxella spp., Actinomyces, Eikenella corrodens.
Skin: Staphylococcus (coagulase –neg.), S. aureus, Propionibacterium acnes, Haemophilus spp., Viridians streptococci, Mycobacterium spp., Bacillus spp., Candida spp.
Conditions under which the environment changes
Do any of the physical conditions change? Are there chemicals, other organisms, nutrients, etc. that might change the community of your niche.
Microscopic Organisms present in the Eye Niche
Microbe Interactions
Many normal bacteria are present in the human eye which includes Staphylococcus, Staphylococcus aureus, Haemophilus species, and Streptococcus species. However, many other outside bacteria compete for their stay in the eye niche that may lead to tragic diseases and unfavaorable results.
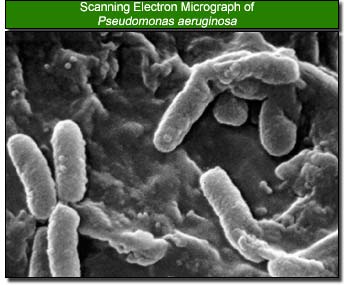
The most common bacteria that can cause corneal ulcer, which could lead to the development of eye blindness, are staphylococcal species, Pseudomonas aeruginosa, Streptococcus pneumoniae, and Moraxella species. Of these, Pseudomonas aeruginosa is found to be the primary bacterium when wearing contact lens, typically soft contacts, that can cause corneal ulcer. Pseudomonas Aeruginosa is a gram-negative, aerobic rod-shaped bacterium which optimally grows at 42 degrees celcius. Although classified as aerobic, it can adapt and live in anaerobic environments. Being an opportunist, this bacterium greatly affects the eye when the defensive mechanisms of the eye are weakened. Otherwise, it does not ordinarily cause disease. It is, however, a very durable bacterium, some of which includes minimal food requirement, a protective outer coating, and resistance to many antibiotics (P1).
A study from the United Kingdom reported that people who wear soft contact lenses result in 8 times more of a chance for bacteria to invade in the cornea when they sleep than the people who only wear them while they are awake (P2).
Dr. Gifford Jones of canadafreepress.com had reported that people who wore contact lens are at risk of developing blinding eye infections during the first six months of use. He proposed that it depended on what the contact lenses were made of. It was said that Pseudomonas aeruginosa infection for soft contact lenses is one in 2,500 and one in 500 if it was worn overnight. For hard contact lenses, infection is one in 10,000. Biodeposits build up on the contact lenses which can cause irritation and corneal ulcers. When contact lenses are worn, it covers up the cornea which restricts oxygen flow to underlying tissues. In an anaerobic environment, Pseudomonas aeruginosa is then able to reside properly. Dr. Dwight Cavanagh, a professor of Opthlalmology at the University of Texas Southern Medical School, stated that new cells on the cornea cannot be produced when the contact lenses cover it, which may decrease the thickness of the cornea by 10 percent (P3).
In 1993, The Department of Ophthalmology at University of Patras medical School in Greece, performed an experiment to determine the clinical microbiological characteristics of corneal ulcers in contact lens wearers. Results showed that, of the ulcerative keratitis patients, 26.74% of them wore contacts. In addition, Pseudomonas aeruginosa was the most frequent isolated bacterium. This experiment concluded that the use of contact lens has a high risk factor for ulcerative keratitis (P4).
Also, a study in Sydney of 2003, Australia found that Pseudomonas aeruginosa produce quorum-sensing signal moleucules which contributed to the induction of the inflammatory response in Pseudomonas keratitis (P5).
Pseudomonas aeruginosa in the eye is targeted by the use of an antibiotic called ciprofloxacin. Current research shows that ciprofloxacin is very effective in treating ulcerative keratitis, although there have been rare results of the bacterium being resistant to the antibiotic, possibly due to the bacterium’s adaptation (6). Pseudomonas aeruginosa has increased its resistance to many antibiotics up to today. Patients who are treats Pseudomonas aeruginosa often have to take multiple antibiotics due to the bacterium’s high barrier of antibiotic resistance (P1).
Fungal Infestation of the Eye Niche
Common fungi found in the Eye: - Acremonium spp. - Aspergillus flavus - Aspergillus fumigatus - Aspergillus niger - Bipolaris spp. - Candida albicans - Curvularia spp - Exserohilum spp. - Fusarium oxysporum - Fusarium solani - Lasiodiplodia theobromae
The warm and moist environment of the eye is subject to mycotic infections from fungal species such as Fusarium, Aspergillus, Acremonium, and the Candida species of yeasts. Due to the preferences for moisture-rich environments and warm weather, the prevalence of fungal infections of the eye is higher in tropical areas and in areas with large agrarian communities [9]. Despite the relatively sterile environment of the eye, factors such as eye “trauma (generally with plant material), chronic ocular surface diseases, contact lens usage, corneal anesthetic abuse and immunodeficiencies” can result in infection of the eye by filamentous fungi or yeasts [8]. Fusarium solani, a species of fungi that is associated with contact lens wearers, normally inhabits warmer climates in soil and organic matter. Invasion of the inner layers of contact lenses and transfer to the surface of the eye, however, is possible due to the ability of Fusarium solani to reside in the moisture-rich inner matrix of soft contact lenses.
Fungal infections, such as fungal keratitis, can occur on the anterior surface of the eye or the cornea, and may also infect the posterior of the eye as a result of mycosis. Fungi that inhabit the eye produce proteases that are specific for the surface on which they grow. As a result, protein catabolism by fungal proteases results in tissue damage to the eye, ulceration as well as an inflammatory response around the infected area [7]. Aggregation of fungal microbes into hyphae enlarges the infected region and prevents host inflammatory cells from ingesting the fungi. As a result of the pattern of fungi to join together and form hyphae, fungi that inhabit the eye may extend throughout the entire depth of the cornea, and tend to grow parallel to the lamellae of the cornea [7][8]. As a result of catabolic degradation of the eye environment by the fungi, an inflammatory response by the host is accompanied by necrosis of the surrounding eye tissues.
Fungal infection of the eye may occur as a result of fungal growth on contact lenses. Due to tendency of fungi to inhabit moisture-rich locations, contact lens cases and the soft inner matrix of contact lenses are subject to fungal growth. Fungi have the ability to degrade hydrophilic polymers of contact lenses, and may be transferred from contaminated lenses to the surface of the eye, often causing the implantation of fungi into the anterior regions of the eye or the cornea and result in fungal keratitis [7].
Do the microbes that are present interact with each other?
Describe any negative (competition) or positive (symbiosis) behavior
Do the microbes change their environment?
Do they alter pH, attach to surfaces, secrete anything, etc. etc.
Do the microbes carry out any metabolism that affects their environment?
Do they ferment sugars to produce acid, break down large molecules, fix nitrogen, etc. etc.
Defense Mechanisms of the Eye
Barely any microbes live in the eye. This stems from the fact that each section within the eye has mechanisms that defend against infection and microbe colonization.
Blink Reflex
The blink reflex is a mechanical defense against particles in the air or trauma. Eyelashes and the sensitive cornea both participate in this reflex. Tears, debris, allergens, microbes, etc. are moved over to the lacrimal excretory system with the motion from the eyelid.
Barriers
The orbital septum, cornea and conjunctiva all provide a protective barrier against pathogens. The orbit and the eyelid are separated into preseptal and postseptal spaces by the orbital septum which creates a physical barrier against infections. The various layers of the cornea limit permeability of items into the eye. Also, native flora of the lids and mucosal surface limit possible pathogenic colonization. [H1]
Tears
Tears are one of the most important defense systems that our eyes have. Tears are secretion from tear glands to lubricate and protect eyes from infection. The tear film allows gas exchange between the air and the epithelium and also keeps the cornea wet. The substance of the tear film is retinol to provide the transparent nature of the epithelium. (M1) Tear fluid is composed of water, electrolytes (sodium and potassium), proteins (immunoglobulin and lysozyme), lipid, mucins, and others, such as lactoferrin, lipocalin, and lacritin.
These components of tears help form a defense system that guards the eyes from infection. For example, IgA is the main immunoglobulin of tears and is synthesized by subconjunctival plasma cells. Because of the dimer structure of this protein, it is very effective in aggregating targets to hampers their penetration of the body surface (C1). Besides immunoglobulin, another protein called lysozyme has similar functions. “Lysozyme is an enzyme that is naturally abundant in tears and plays an important role in maintaining healthy eyes” (C2). Lysozymes are known to have anti-inflammatory and bacteriolytic functions because they can dissolve the outer coating of certain bacteria; and thus, killing the bacteria so that it cannot infect the eye.
In addition, lactoferrin is an iron binding protein secreted by the epithelial acinar cells of the lacrimal glands. Lactoferrin is one of the key molecules that regulate the inflammatory response and the protection against infections. The sequestration of iron by lactoferrin can firmly control bacterial flora and inhibits the iron catalyzed production of hydroxyl radical. Generally, hydroxyl radical may lead to major tissue damage by producing peroxidation of cell membrane lipids, oxidative damage to proteins, and generation of other free radicals. Hence, lactoferrin’s ability to deplete free iron can inhibits the pro-inflammatory effects of hydroxyl radical; and therefore, protects the mucosal surfaces from oxidative damage. Moreover, lactoferrin inhibits the ability of bacteria and viruses to attach to cell membrane of the host cell (C3). Furthermore, lipocalin is another component of tears that form the eye’s defense system. Lipocalin is the major lipid binding protein in tears. Lipocalin can bind to wide variety of lipids such as fatty acids, cholesterol, phospholipids and glycolipids. By interacting with lipids, tear lipocalin can contribute to the surface pressure in the tear film. Moreover, lipocalin also help to promote optical clarity and may transport lipids from the lacrimal gland to the tear film. It plays a significant role in the protection of the ocular surface from desiccation because the lipids that are bounded to the tear lipocalin can serve to provide a reservoir of lipid molecules in equilibrium with the surface and could reduce evaporation of water. Moreover, tear lipocalin can gather lipid from the cornea to prevent dry spots from forming on the cornea (C4).
Likewise, lacritin also help promote ocular surface wetting. Lacritin flows onto the ocular surface from the lacrimal and meibomian glands. It is capable of protecting epithelia of the lacrimal-corneal axis against inflammation that associated with cell death.
Tear production system The protection of tears has three separate layers: the lipid layer, the aqueous layer, and the mucous layer. The outermost lipid layer is a thin layer (0.1-0.2µm that is composed of hydrophobic surface. It contains oils (lipids) produced by the meibomian glands. This oil (lipids) function to retain moisture and prevent tear fluid loss due to evaporation. The temperature in meilbomain gland lipids has the low melting point range 19-32°C depending on the degree of fatty acids unsaturation. The meibomain gland lipids allow lipid get through the meibomian gland ducts and excretion to the tear film.
The aqueous layer is a tear film including water and proteins produced by the lacrimal gland. The middle layer of the lacrimal gland is the thickest (7-8µm) and produces 0.9% saline tears. It functions delivery the main nutrients such as oxygen to the cornea and carrying waste products away from the cornea
The innermost mucous layer (30µm) contains mucin produced by goblet cells. The mucin provides a protective barrier for the surface and serves as a means for the aqueous layer to adhere to the surface of the eye. The mucous’s thin layer of hydrophilic surface can make even on the corneal surface to spread water. (M1, M2, M3, M4 and M5)
Conditions which affect the lipid layer or the mucin layer can cause a dry eye.
[LIPID MISSING PICTURE HERE] [LIPID RESTORED PICTURE HERE]
People with dry eyes lack a complete lipid layer which result in an inadequate amount of tear fluid. Without this tear fluid, the person may experience a burning, gritty sensation and/or blurred vision. When lipids are restored, fluid is then replenished and moisture is restored in the eyes. (C5)
Many species of bacteria are found in the eyelid and conjunctiva as symbiotic relationships. These are Staphylococcus aureus (S. aureus), Staphylococcus epidermidis, Streptococcus, Corynebacterium, Propionibacterium, and Propionibacterium acnes. These bacteria produce lipase that harm to the tear film lipid layer especially, S. epidermidis and S. aureus produce triglyceride lipase and cholesterol and wax esterase. Accumulated cholesterol is used for the growth of certain ocular bacteria. Increased bacteria are regulated by anti-microbial peptides from the lacrimal gland and conjunctiva. (M6)
Immune Response
The cornea, due to lack of a vascular system, contains limited immune defenses. Immune defenses are provided by Langerhans cells and immunoglobulins. Langerhans cells modify B and T cell in the cornea while glycocalx coupled with a mucous glycoprotein layer covers the corneal surface for added protection.
Langerhans cells have receptors for immunoglobulins, antigen, and complement. They work like macrophages to destroy foreign invaders. The B and T cells that are modified by Langerhans cells work in conjunction with lymphocytes, resulting in a strong immune reponse. [H1]
Leukocyte Defense: Leukocytes consume and destroy microorganisms via and oxygen-dependent pathway and an oxygen-independent pathway. The oxygen-dependent pathway uses oxygen radicals while the oxygen-independent pathway utilizes defensins which are peptides that have a broad range of antibacterial, antifungal, and some antiviral activities. [H3]
Current Research
1. Iris Pigment Epithelium Expressing CD86 (B7-2) Directly Suppresses T Cell Activation In Vitro via Binding to Cytotoxic T Lymphocyte–associated Antigen 4
A current study at Schepens Eye Research Insitute at Harvard Medical School identified the thin layer of cells lining the iris of the eye, the iris pigment epithelium (IPE), as a tissue that contributes to ocular-immune privilege by secreting immunosuppressive factors and expressing surface molecules that trigger apoptosis by T cells. Scientists exposed PE cells from various regions of the eye to purified T Cells and found that only the IPE cells from the iris responded to CD86 by stopping T cell activity. Normally, T cell activity in the body promotes inflammation. By suppressing its activity, however, IPE cells are able to prevent inflammation in the eye. The results demonstrated that IPE cells contribute to immune privilege and prevent eye inflammation in the iris. The study identified that disruption of IPE cell activity in the eye, perhaps by infection or other factors, may lead to immunogenic inflammation, disruption of the visual axis and blindness [10].
2. Viral Infection of the Lungs through the Eye
Respiratory syncytial virus (RSV) is a virus that is greatly known to affect the respiratory system of newborns. Researchers discovered an association of this virus with allergic conjunctivitis, also known as Pink Eye, which allowed the virus to flow toward the respiratory system initially from the eyes. They experimented on a live mouse by applying RSV to its eyes. RSV not only replicated but also drifted to the lungs, which produced a respiratory disease. Treatment of RSV with anticytokine was successful. They concluded that respiratory infections could be involved with the eye and that it can be treated. [P7]
3. Phospholipase A2 in Rabbit Tears: A Host Defense against Staphylococcus aureus
Staphylococcus aureus causes keratitis in many human populations. Tear films have many nutrients that are beneficial for bacteria growth. Tear films also, however, have an effective defense system to reduce replication of bacteria and regulate bacterial growth. One of inhibitors for the host defense molecule is phosholipase A2 (PLA2). Moreau et al. studied the PLA2 effect on Staphylococcal activity. They found PLA2 killed Staphylococcus by hydrolyzing bacterial membranes to release fatty acids and destroyed the bacterial cell wall. [M7]
4. Antimicrobial Contact Lenses
Current research on antibacterial contact lenses to prevent microbial colonization into the eye niche, are going through various trials to measure the effectiveness of such lenses in comparison to regular lenses. The lenses were created to address the problem ofmicrobial contamination of lenses ranging from acute red eye to microbial keratitis. The lenses work by "jamming" the signaling systems that bacteria use to form biofilms. [H4] The technology involves using a furanone technology to jam the signals and was discovered from a seaweed that generates the anti-biofilm compound. The drug-resistance problem of current antimicrobial agents should not affect these lenses since the technology is not designed to kill bacteria. [H5]
References
(C1) “Biological Functions of Immunoglobulins.” Structure, Function, & Genetics of Immunoglobulins. 20 Aug. 2008. <http://www.lib.mcg.edu/edu/esimmuno/ch3/biologic.htm>
(C2) “Lion to Release ‘Smile 40 Mediclear’.” Lion. 30 July 2007. 19 Aug. 2008. <http://www.lion.co.jp/en/press/html/2007017f.htm>
(C3) “Tear Lactoferrin.” 20 Aug. 2008. <http://www.touchscientific.com/lfnfact2.htm>
(C4) “Dry Eye Disease.” Mission for Vision – Complications of LASIK. 31 Dec. 2005. 19 Aug. 2008. <http://www.missionforvisionusa.org/content/2005/12/dry-eye-disease.html>
(C5) “Almera Soothe Eye Drops.” Matheson Optometrists. 25 Aug. 2008. <http://www.matheson-optometrists.com/Products/Almera-Bausch-and-Lomb-Soothe.htm>
(H1)
(H4) "First Human Trial of Antibacterial Contact Lenses." ScienceDaily. 29 June 2006. 27 Aug. 2008. <http://www.sciencedaily.com /releases/2006/06/060629085000.htm>
(H5)"Biosignal's Furanone Technology." MedGaget. 30 June 2006. 27 Aug. 2008. <http://medgadget.com/archives/2006/06/biosignal_furan.html>
(M1) Walcott, Benjamin "The Lacrimal Gland and Its Veil of Tears" April 1998. 27 August 2008. <http://physiologyonline.physiology.org/cgi/content/full/13/2/97>
(M2) Tiffany, M. J "Tears in health and disease" 5 September 2002. 27 August 2008. <http://www.nature.com/eye/journal/v17/n8/full/6700566a.html>
(M3) The Cornea 27 August 2008. <http://www.tedmontgomery.com/the_eye/cornea.html>
(M4) Eyelids and Tear channels 27 August 2008. <http://www.lea-test.fi/en/eyes/lidsncha.html>
(M5) "Comparative bacteriology of chronic blepharitis" 1984. 27 August 2008. <http://bjo.bmj.com/cgi/content/abstract/68/8/524>
(M6) McCulley, P. James and Ward E. Shine "The lipid layer of tears: dependent on meibomian gland function" 11 September 2003. 27 August 2008. <http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6WFD-49H6Y6X-1&_user=10&_rdoc=1&_fmt=&_orig=search&_sort=d&view=c&_version=1&_urlVersion=0&_userid=10&md5=cae06adc263756d8bb890e5f40005b89#toc1>
(M7) Moreau, M. Judy, et al "Phospholipase A2 in Rabbit Tears: A Host Defense against Staphylococcus aureus" 2001. 27 August 2008. <http://www.iovs.org/cgi/content/abstract/42/10/2347>
(7) Wilson LA, Ajello L. Agents of oculomycosis: fungal infections of the eye. In Collier L, Balows A, Sussman M, eds. Microbiology and microbial infections. 9th ed. Vol. 4, London, Arnold, 1998.
(8) Tanure, M. A., E. J. Cohen, S. Sudesh, C. J. Rapuano, and P. R. Laibson. 2000. Spectrum of fungal keratitis at Wills Eye Hospital, Philadelphia, Pennsylvania. Cornea. 19:307-12.
(9) L. Alcazar-Fuoli, E. Mellado, A. Alastruey-Izquierdo, M. Cuenca-Estrella, and J. L. Rodriguez-Tudela Aspergillus Section Fumigati: Antifungal Susceptibility Patterns and Sequence-Based Identification Antimicrob. Agents Chemother., April 1, 2008; 52(4): 1244 - 1251.
(10) Sugita, S., J. Streilein. “Iris Pigment Epithelium Expressing CD86 (B7-2) Directly Suppresses T Cell Activation In Vitro via Binding to Cytotoxic T Lymphocyte–associated Antigen 4.” J. Exp. Med., Jul 2003; 198: 161 - 171.
(P1) Rowland, Belinda. “Pseudomonas Infections.” August 2006. http://www.healthatoz.com/healthatoz/Atoz/common/standard/transform.jsp?requestURI=/healthatoz/Atoz/ency/pseudomonas_infections.jsp
(P2) Naradzay, Jerome. “Corneal Ulceration and Ulcerative Keratitis.” November 2006. http://www.emedicine.com/emerg/topic115.htm
(P3) W. Gifford, Jones. “Careless Use of Contact Lenses Can Cause Blindness”. March 2003. http://www.canadafreepress.com/medical/ear-nose-throat030203.htm
(P4) EK, Mela, et.al. “Ulcerative keratitis in contact lens wearers.” October 2003. http://www.ncbi.nlm.nih.gov/pubmed/14555893?ordinalpos=1&itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_DiscoveryPanel.Pubmed_Discovery_RA&linkpos=1&log$=relatedarticles&logdbfrom=pubmed
(P5) H, Zhu, et.al. “Pseudomonas aeruginosa quorum-sensing molecules induce IL-8 production by human corneal epithelial cells.” May 2008. http://www.ncbi.nlm.nih.gov/pubmed/18463485?ordinalpos=6&itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_RVDocSum
(P6) Lomholt, JA and M Kilian. “Ciprofloxacin susceptibility of Pseudomonas aeruginosa isolates from keratitis.” October 2003. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1920786
(P7) Bitko, Vira, et.al. "Viral Infection of the Lungs through the Eye." January 2007. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1797451

