Hantaviruses: Old World vs. New World
Code Notes:
By Raphael A. Melo
At right is a sample image insertion. It works for any image uploaded anywhere to MicrobeWiki.
The insertion code consists of:
Double brackets: [[
Filename: PHIL_1181_lores.jpg
Thumbnail status: |thumb|
Pixel size: |300px|
Placement on page: |right|
Legend/credit: Magnified 20,000X, this colorized scanning electron micrograph (SEM) depicts a grouping of methicillin resistant Staphylococcus aureus (MRSA) bacteria. Photo credit: CDC. Every image requires a link to the source.
Closed double brackets: ]]
Other examples:
Bold
Italic
Subscript: H2O
Superscript: Fe3+
Sample citations: [1]
[2]
A citation code consists of a hyperlinked reference within "ref" begin and end codes.
To repeat the citation for other statements, the reference needs to have a names: "<ref name=aa>"
The repeated citation works like this, with a forward slash.[1]
Hantaviruses
Background
Hantaviruses belong to the viral family Bunyaviradae, of which there are five genera: bunyavirus, phlebovirus, nairovirus, tospovirus, and hantavirus (HTV). Although there is no definitive number, there are around 35 known species of HTV that currently exist in the world, including both old-world and new-world strains. Hantaviruses are typically able to infect humans via urine and fecal matter of rodents (Clement et al., 2006). When bedding covered in rodent waste is disturbed, the waste particles become aerosolized and are able to be inhaled, potentially leading to infection (Clement et al., 2006). Rodents are the main vector of this disease into humans, and those who work with rodents that are infected are at an increased risk of becoming infected themselves. Transmission from human to human is far more rare than rodent to human, although it is not impossible. Infected aerosolized particles may also enter the human body through mucous membranes in the eyes, nose, and mouth, and through breaks in the skin, for example, from a rodent bite. Most infections and outbreaks arise within conditions that allow for these particles to become aerosolized, such as during land development, agriculture, and urbanization (CDC, 2021). The first recorded strain was documented as the Hantaan strain, an Old-World variant, between 1951 and 1953 in Korea, and was the first species of many to be documented since. Infecting more than 3000 Korean soldiers during the Korean war, the Hantaan virus outbreak was located near the Hantan river, providing the virus with its name that would later represent all future variants (Mir, 2010). In regards to healthcare, there is no effective cure or treatment for HTV (CDC). Patients will typically receive a broad range of antibiotic therapies and supportive care while being closely monitored (CDC). In regards to treatment based on the type of HTV, there are generally different procedures and drugs administered based on whether or not the species of HTV is “Old World” or “New World.” HTV human infections may result in two possible diseases: hemorrhagic fever with renal syndrome (HFRS) and HPS hantavirus pulmonary syndrome (Avsic-Zupanc et al., 2019). What determines whether the infection yields HFRS or HPS is the type of capillary bed that is infected. For HFRS, renal medulla capillaries are the origin of infection while for HPS, the pulmonary capillaries are the target (Avsic-Zupanc et. al., 2019). Despite this, most initial symptoms tend to be the same for both syndromes include typical flu-like symptoms in addition to aggressive fevers, malaise, and myalgia. Additional symptoms include increased vascular permeability which can lead to hypotension thrombocytopenia and leukocytosis with a left shift, meaning that an abundance of neutrophils are destroyed at the site of infection, causing an increase in production in the bone marrow (Avsic-Zupanc et. al., 2019)(Ishimine et al., 2013).
Genome
Hantaviruses are negative-sensed, enveloped, single-stranded RNA viruses. They have 3 segments of RNA within their genome, Small (S) , Medium (M), and Large (L). Each mRNA segment’s open reading frame is placed in between much smaller non-coding regions that are capped. The L segment plays a key protein in viral replication and codes for the production of L proteins, which facilitate transcriptase and replicase functions. Additionally, L proteins are thought to have a role in endonuclease cleavage of cellular mRNAs to produce primer caps used for transcription initiation of viral mRNAs in a process known as “cap snatching” (CDC, Hantaviruses). The M segment codes for the glycoprotein precursors Gn and Gc, also known as G1 and G2 proteins respectively. These proteins later form the glycoprotein outer membrane matrix and allow the virus to bind to host cell receptors. The S segment codes for N proteins, which serve a variety of functions needed to complete the viral life cycle. Arguably the most important of the proteins, N proteins aid in translation, trafficking, assembly, the possible modulation of the host’s immune system, and serve as the building material needed to form the viral nucleocapsid (Jonsson et al., 2010).
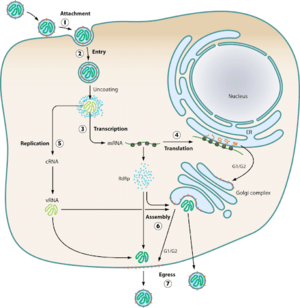
Viral Life Cycle and Structure
HTV virions typically infect endothelial, epithelial, macrophage, follicular dendritic, and lymphocyte cells (Jonsson et al., 2010). Using glycoproteins, the virions attach to the host cell’s surface membrane receptors and enter the cell via endocytosis. The most common receptors that allow for entry of the virion are β1 integrins, with some exceptions involving β3 receptors (Jonsson et al., 2010). Upon cellular uptake, the virions are enclosed in endosomes where the viral capsule is immediately uncoated. The L, M, and S mRNA segments are set up for transcription via the viral RdSp enzyme, and subsequent translation of the L and S mRNA occurs on free floating ribosomes, while the M mRNA undergoes translation on membrane-bound ribosomes (Jonssonet al., 2010). Assembly of all virion components at the golgi apparatus means the virus exclusively replicates within the host’s cytoplasm (Parvate, 2019). Beyond this, little more than speculation exists on how HTV RNPs are synthesized, trafficked to the golgi, and what drives the budding mechanisms of the golgi. Further speculation of alternative assembly methods for new world hantaviruses could point towards the plasma membrane as a potential assembly site (Jonsson et al., 2010). Golgi vesicles containing newly made viral particles then fuse with the host cell’s plasma membrane, and are then released into the extracellular environment.
Old World Hantaviruses
Hantaviruses in Asia and Europe
Hemorrhagic Fever with Renal Syndrome

HFRS are technically a group of very similar diseases that all hail from a small subset of hantaviruses: Korean hemorrhagic fever, epidemic hemorrhagic fever, and nephropathia epidemica. The species of Hantavirus that result in these syndromes include Hantaan, Dobrava, Saaremaa, Seoul, and the Puumala strain. Typically, HFRS is divided into 5 distinct phases that the patient may experience. The first phase involves an incubation period of 2 to 4 weeks, followed by a second, febrile phase that involves headache, backache, high fever, abdominal pains, chills, nausea and vomiting. Somnolence, lethargy, and visual distortion and blurring have also often been reported in addition. The end of this phase sees conjunctival hemorrhages in the eyes. The second stage can last as little as a couple of hours to a day or two, and involves hypotension. In some cases shock is also present, and about ⅓ of all deaths of HFRS result from imminent shock. Further hemorrhaging, thrombocytopenia and leukocytosis are characteristic of this phase especially. The third phase, the oliguric phase, lasts about 3 to 7 days, and generally sees a decline in kidney function yielding abdominal and back pains. This phase results in half of all HFRS associated deaths. The fourth phase, the polyuric phase, sees a reversal of the third phase, with a return of kidney function. This leads to the diuretic phase which is typically a sign of recovery, and can last anywhere from days to weeks, and sees a massive increase in micturition in the patient. Recovery can take up to half a year to complete, and there are very rarely lingering symptoms or complications once recovery is complete. (Whole paragraph - CDC and (Avsic-Zupanc et. al., 2019).
Include some current research, with at least one figure showing data.
New World Hantaviruses
Hantaviruses in the Americas
Hantavirus Pulmonary Syndrome
Further Treatments and Prevention
Critical care management of HTV infections that yield both HFRS and HPS/HCPS stress the avoidance of fluid overload due to the possibility of increasing fluid buildup in the lungs or swelling of the kidneys. Pressors to maintain cardiac output are also essential during the treatment process, and in the most extreme cases, the use of extracorporeal membrane oxygenation. There is no current vaccine or cure for HTV infections. One of the reasons why a vaccine is not currently being pursued is due to the dangers associated with mass-production of a virus under strict containment. Some work has been previously done but a major roadblock is that the smallpox vaccine greatly reduced the efficacy of the prototype HTV vaccine. Many attempts to create effective vaccines have approached using plasmids and viral genomes in particular to stimulate the desired type and number of antibodies. The antiviral drug ribavirin, also known as virazole, is the only known drug used for HTV infections, and efficacy of the drug depends entirely on how close to the initial time of infection the drug is taken as it targets the replication cycle. The general efficacy of ribavirin is heavily debated based on a slew of clinical trials from the 90s and early 2000s that yielded relatively non-conclusive results in regards to effectiveness. Despite that, it is one of the most broad-range antiviral agents available currently and is an effective early stage treatment for HPS, especially when combined with other supportive drugs. Ribavirin is able to successfully prevent growth in mammalian cells, and has other cytostatic properties that allow it to cause hyper mutations in RNA viruses, and disrupt the lifecycle of DNA viruses at the replication stage. Given the number of HtV infections that occur each year, it is necessary for further research to fully discern a proper and decisive method of treatment, however that number of infections is not large enough to warrant further research towards the particular production of a vaccine. Prevention of hantavirus involves preventing the disease vector itself from getting in contact with the individual, such as taking measures to seal up homes, maintain clean living environments, and providing traps for rodents.
References
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski,at Kenyon College,2024
