Micrococcus
A Microbial Biorealm page on the genus Micrococcus
Classification
Higher order taxa:
Bacteria; Actinobacteria; Actinobacteria (class); Actinobacteridae; Actinomycetales; Micrococcineae; Micrococcaceae
Species:
Micrococcus antarcticus; M. luteus; M. lylae; M. roseus; M. sp.
|
NCBI: Taxonomy Genome: -Micrococcus sp. 28 plasmid pSD10 -Micrococcus luteus plasmid pMLU1. |
Description and Significance
M. luteus can be found in many places such as the human skin, water, dust, and soil. Micrococcus is generally thought of as harmless bacterium, but there have been rare cases of Micrococcus infections in people with compromised immune systems, as occurs with HIV patients.
Genome Structure
Hybridization studies show no close genetic relationship among the species of Micrococcus. For example, M. luteus and M. lylae are 40-50% genetically different. M. luteus has a G-C content of 65-75 mol%. About half of the strains of M. luteus were found to carry plasmids 1 to 100MDa in size. So far two genome sequences have been done, one on Micrococcus sp. 28 plasmid pSD10 and another on Micrococcus luteus plasmid pMLU1.
Cell Structure and Metabolism

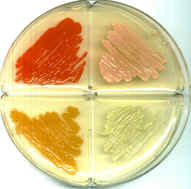
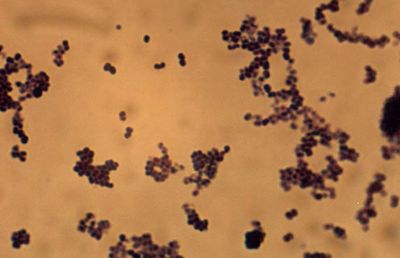
Micrococcus are Gram-positive cocci that are 0.5 to 3.5 micrometers in diameter and usually arranged in tetrads or irregular clusters. They are generally strict aerobes and can generally reduce nitrate. M. luteus oxidizes carbohydrates to CO2 and water, and it does not produce acid from glucose as well as it does not make arginine dihydrolase or b-galactosidase. Some Micrococcus are pigmented bacteria; for example, M. luteus produces yellow colonies and M. roseus produces redish colonies. Micrococcus species are oxidase-positive, which can be used to distinguish them from other bacteria like most Staphylococcus species, which are generally oxidase-negative. Defining characteristics of Micrococcus are the ability to aerobically produce acid from glucose glycerol, aesculin hydrolysis, arginine dihydrolase, major pigment production, motility, and conversion of nitrate to nitrite (Smith et al. 1999).
Ecology
M. luteus has been isolated from human skin, animal and dairy products, and beer. It can be found in many other places in the environment, as well, like water, dust, and soil. M. luteus on human skin breaks down compounds in sweat into compounds with bad odor. M. luteus can grow well in environments with little water or high salt concentrations. They grow optimally at 37oC and can be easily grown on inorganic nitrogen agar or Simmon's citrate agar. Although some, like Micrococcus antarcticus, are cold-adapted, and have been found living in Antarctica and in marine environments.
Pathology
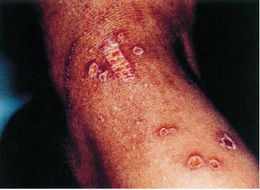
Although Micrococcus rarely causes infections or problems in the body, those with compromised immune systems, such as occurs with HIV+ patients, have been known to get skin infections caused by Micrococcus luteus. The skin infections, or chronic cutaneous infections, result in pruritic eruptions of the skin in some areas as well as scattered papule lesions with or without central ulcerations. Micrococcus as the cause of infections is easy to overlook because infections caused by this bacterium are rare as well as the bacterium is a natural part of the skin's bacterial flora. Thus, when dealing with Micrococcus infections, it usually takes several cultures being grown and examined (at about 35oC on blood agar) before one realizes that Micrococcus is indeed the culprit. Most Micrococcus infections are discovered through process of elimination (all other bacterial, fungal, etc. tests showing up negative) along with the presence of abundant Micrococcus tetrads in the lesions or cysts (which would normally have been overlooked because Micrococcus is a natural part of the skin's microbial flora) (Smith, et al. 1999). Though today immunocompromised patients the risk of infection has grown. There have been several deaths in immunocompromised children (caused by leukemia) from pulmonary hemorrhages because of Micrococcus. Recently, this organism was recognized as an opportunistic pathogen and has been implicated in recurrent bacteremia, septic shock, septic arthritis, endocarditis, meningitis, intracranial suppuration, and cavitating pneumonia in immunosuppressed patients.
References
Encyclopedia Britannica: Micrococcus
Health Canada: Material Safety Data Sheet: Micrococcus spp.
MadSci Network: What are the effects of Micrococcus luteus on the human body?
