Myxococcus xanthus - The Swarming Bacteria: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{Uncurated}} | |||
<br>By [Patrick Nally] <br> | <br>By [Patrick Nally] <br> | ||
[[Image:myx1.jpg|thumb|800px|right| A photo of Myxococcus xanthus cells. [https://en.wikipedia.org/wiki/Myxococcus_xanthus#/media/File:M._xanthus_development.png].]] | [[Image:myx1.jpg|thumb|800px|right| A photo of Myxococcus xanthus cells. [https://en.wikipedia.org/wiki/Myxococcus_xanthus#/media/File:M._xanthus_development.png].]] | ||
Latest revision as of 23:50, 19 July 2021
By [Patrick Nally]

Introduction
Myxococcus xanthus is a specific species of myxobacteria that are single celled but exist in “swarms” or “packs”. Myxococcus xanthus are rod-shaped and are gram-negative. M. xanthus was discovered in 1892 by Roland Thaxter. [1] M. xanthus are found in soil or any place plentiful with organic matter. They live best when the ph of the soil is around 7-8 and the temperature is around 30 degrees C. [2] M. xanthus is also a chemoorganotroph, which means they are organisms that oxidize the chemical bonds in organic compounds that they then use for energy. Myxococcus xanthus live together by living in a small biofilm or slime sheet. [3] They move as one in search of nutrients, but when nutrients are not immediately available the cells are able to transform and come together as one large mass in order to survive.
Classification
Kingdom: Bacteria
Phylum: Proteobacteria
Class: Delta Proteobacteria
Order: Myxococcales
Family: Myxococcaceae
Genus: Myxococcus
Species: M. xanthus
Ecology & Genome
Myxococcus xanthus is found in the Myxococcales order. Myxococcales can be characterized by the following: have specific ways of communicating with each other and are very social, they are morphogenetic, and they are able to move around via gliding. Many of these requirements for being in this order can be due to the habitat and nutrient availability. The gliding helps locate and attack other organisms within their general area. Myxococcus xanthus is also considered a predator since they eat other microbes in their environment. Myxococcus xanthus has been described as a social bacteria as well. [4]This means that each cell relies on each other in order to survive.
All myxobacteria, with one exception, have the largest genomes in comparison to other prokaryotes. Since myxobacteria are so complex and organized the large genome could be due to how they survive and live in harsh environments. [5]Since they are able to live together in tough conditions the genomes have high plasticity which results in changes in the genome such as the formation of the fruiting body. Four different myxobacteria species genomes were compared and it was found that 452 genes were found in all of them. [6]This means that these genes could be responsible for the formation of the fruiting body.
Myxococcus xanthus had its genome sequenced in 2001 and it was found to be 9.14 Mb. This is much larger than other deltaproteobacteria. It was found that specific genes were selected for rather than random gene duplication. These genes included sensing for small molecules, transcription control, and cell to cell signaling[7]The entire genome consists of 7,500 genes in total. Myxococcus xanthus also contains genes that aid in synthesizing amino acids as well as secreting growth inhibitors. There is also recent evidence that Myxococcus xanthus has a mechanism for breaking down protein. [8]It has also been found that fluids located within Myxococcus xanthus are able to lyse cell walls by hydrolyzing the peptidoglycan. [9] Each cell is only able to secrete so much, hence the reason they travel together in groups.
Fruiting

Fruiting is the method at which Mxyococcus xanthus reproduces. This occurs when nutrients are unavailable or scarce. When no food is obtainable the individual Myxococcus xanthus cells organize themselves into larger masses. On average about 100,000 cells assemble to form the larger mass. This large mass is called a fruiting body. [10] As the cells are forming the fruiting body they are actually being selected for based on related and unrelated cells. This creates uniform and species specific fruiting bodies among all of the cells. About 10% of the rod shaped cells that form the fruiting body then transform into sporangioles which also contain smaller myxospores. Around 30% of the original cells become the rods located on the outside of the mass of cells. The rest of the cells encased inside die off and become a source of nutrients for the remaining cells. It is believed that the cells come together to make sure a new colony is made by many cells opposed to one. Unlike other spore forming bacteria the rod-shaped cells directly transform into spherical myxospores.
The fruiting body is formed when groups of cells start to spiral together and travel to certain points called aggregation centers.[11]As these aggregation centers form they become three-dimensional due to the meeting of different waves of cells. [12] After the fruiting body is completed two new cells have been formed. The myxospores which are found inside of the fruiting body and the peripheral rods which are found around the outside of the fruiting body.[13]
Around 6-12 hours after the cells were starved of nutrients and the fruiting body is formed the rods that were unused become lysed.[14] It has been found that the lysing cells are able to donate spore coated proteins to their own spores increasing the strength of the cell. [15]After 72 hours the spores are finally mature enough to germinate, and once they do it creates a new population of cells which start to swarm together in order to gain nutrients needed to grow. [16]In terms of fruiting body formation, the cells need to be stimulated by being close to another cell which initiates S-motility, also called twitching motility. [17]
In terms of why Myxococcus xanthus uses the fruiting body method there are a couple hypotheses. One is that it reduces the competition between other organisms by multiplying until they out number other organisms. [18]Another is that the spore may protect them from outside threats or stresses that may arise. [19] Lastly, it is possible that all of the processes that lead up to myxococcus formation is only to maintain their specialized lifestyle and make sure they are able to grow well. [20]
Life Cycle
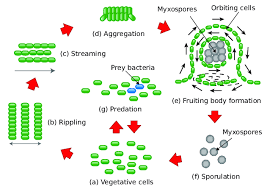
Myxococcus xanthus lives in a never-ending cycle. It “starts” when there are many individual vegetative cells, which just means a cell that is actively growing. The cells then scout for prey in order to obtain nutrients or they branch off together in much larger quantity. The cells then find a prey and attempt to attack it by rippling through the organism. If the cells do not find any prey they are forced to come together in order to begin fruiting. Once the aggregates are formed the cells within them start to become spores and then as soon as nutrients are available the spores are germinated and made into many vegetative cells again. Myxococcus xanthus has two ways of directly multiplying. They are able to attack neighboring cells and feed on the nutrients that the cells excreted. They can also come together when nutrients are infrequent and form fruiting bodies that contain myxospores. The myxospores are able to live for a long time while under harsh conditions giving them the best chance of survival.[12] The fact that Myxococcus xanthus is able to do perform either method makes it a highly studied organism due to its complexity.
Motility
Myxococcus xanthus use two separate forms of motility: A-motility and S-motility. A-motility which is also called gliding motility, is when single cells drive movement of the entire group of cells. This movement occurs when the cells are searching for nutrients and prey. This process of movement occurs very slowly through the environment. The result is many single cells distributed throughout the substrate. It has been found that the extracellular matrix is enough to facilitate the adhesion to other substrates in order to move in one direction. [21] While slowly moving across the substrates, a small trail of slime is left behind. Other cells are then able to use the trails almost as a small highway to travel on. These trails also facilitate close cell to cell contact and interactions. [21] Myxococcus xanthus has been compared to ants in terms of pattern of movement as a group. S-motility is the second form of motility and is sometimes called twitching motility. S-motility is when a large group of cells move together by swarming but single cells do not move on their own. This motility is used for fruiting as well as predation. This movement results in a few clusters of cells as well as a few single cells throughout the substrate. S-motility is possible thanks to the type IV pili which is able to extend and retract.
Swarming is mentioned a lot when it comes to Myxococcus xanthus. Swarming is when many cells come together to form a multicellular structure. While in the swarm the rod cells are moving around initiating direction. The organization of the swarm is so complex that each cell is able to move without getting into the way of other cells. [22]
Another movement associated with Myxococcus xanthus is rippling. Rippling is when cells organize themselves together and move as small waves over a substrate or organism. It has been found that when contacted with a larger molecule or prey the Myxococcus xanthus bacteria will start rippling over in order to feed on the nutrients. Rippling has been seen in preying on other organisms as well as forming the fruiting body. [23] Rippling is able to occur on proteobacteria, yeast, phages, as well as gram-positive bacteria.
Predation
Myxococcus xanthus use other organisms as energy so they are able to grow. The cells are constantly looking for bacteria and other organisms to kill. It has been found that Myxococcus xanthus is able to release antibiotics and enzymes that aid in breaking down the prey. [24] The secretions of the antibiotics and enzymes cause the outer membrane of the prey to become lysed. The biomass of the organism is then excreted and picked up by the Myxococcus xanthus cells via rippling.
Some bacteria are able to protect themselves from the predation of Myxococcus xanthus. This occurs when bacteria have built up defense mechanisms such as forming biofilms, making a mechanical barrier, or secreting antibiotics.
Both A-motility and S-motility have been found to be very productive in terms of predation. [25] It has also been found that in laboratory conditions most prey species aid in growth of Myxococcus xanthus.[26] Myxococcus xanthus cells are able to recognize dead cells and avoid them and identify cells of prey and then attack them. [27] Several other theories are currently in circulation such as the theory that Myxococcus xanthus can release chemo attractants to draw prey in but not enough studies have been performed to confirm nor deny.
When in the soil Myxococcus xanthus feed on different bacteria throughout the soil, cyanobacteria, yeasts, fungi, E. coli, and S. aureus. When taking up the contents of the cells that have been lysed the Myxococcus xanthus cells feed on the cytoplasm, carbon molecules, nitrogen molecules, and of course ATP.
It has also recently been found that Myxococcus xanthus acts cannibalistic as well as killing other species. This means that the healthy cells are able to kill weaker or less fit cells which maintains the healthy community of Myxococcus xanthus. It has more recently been found that the Myxococcus xanthus cells selectively choose their siblings or relatives and fuse together with them while killing other cells as well. [28]
Communication
The communication methods of Myxococcus xanthus are not very well known currently. It is believed that they are able to communicate cell to cell and have complex social networks. Cell to cell communication is believed to be happening due to the complexity of the fruiting and sporulation processes. Messages or signals are sent and received by the cells in order to organize themselves especially when fruiting.
It has been found that many prey of Myxococcus xanthus use acyl homoserine lactones as quorum signals. Quorum signals are signals that are sent and received and acted upon. Myxococcus xanthus cells are unable to produce their own acyl homoserine lactones but they are able to identify the presence of them in their immediate area. The acyl homoserine lactone was found to increase the chances of being overtaken by Myxococcus xanthus.[29]
Other Myxococcus Strains
Myxococcus xanthus DK 1622; Myxococcus xanthus DZ2; Myxococcus xanthus DZF1; Myxococcus xanthus NewJersey2; Myxococcus xanthus DSM16526
Research
Many studies have been done and continue to be done on Myxococcus xanthus because of the complexity of their lives. Although it is continuously being studied there is a lot of information we do not know about how the cells do what they do.
A recent study found that the rod shaped cells are tightly packed together and form a active nematic liquid crystal. This means that the cell to cell interactions as well as the different motilities are enough to start the layering of cells when in starvation. [30]
Another study found that the secreted proteins from the Myxococcus xanthus only lysed Gram-positive species. When the Myxococcus xanthus cells were in contact with four test strains they were able to kill all four of them. The only cell envelopes that were not able to be disintegrated by the Myxococcus xanthus cells were gram-positive cells. [31]
A study was done in 2003 found that Myxococcus xanthus lineages that did not contain a type IV pili, which is used to swarm and move, instead had a separate mechanism that evolved in order to still swarm. They found that there was an overproduction of extracellular fibril matrix when the type IV pili was unavailable. This extracellular fibril matrix is what helps hold individual cells together, therefore the cells were still able to group up and move together. [32]Specific relationships with other organisms have been discovered as well. A well-known bacteria that serves as a prey for Myxococcus xanthus is Escherichia coli. Their relationship is known as the Red Queen hypothesis, which states that a species has to constantly evolve in order to survive against other constantly evolving species. [33] So in this case Myxococcus xanthus usually preys on E. coli but E. coli will eventually evolve to defend itself. The Myxococcus xanthus will then also evolve in order to successfully grow.
Bacteriophages that are able to lyse Myxococcus xanthus have also been discovered. They are specifically called myxophages. The myxophage called MX8 was found to start up a lysogenic cycle in the Myxococcus xanthus cells. The DNA of the MX8 myxophage is able to be inserted via recombination. [34]
A lot is still unknown about Myxococcus xanthus and there is hope that we could possibly use Myxococcus xanthus to kill unwanted bacteria. The killing of unwanted bacteria could be used for food storage, agriculture, as well as healthcare settings.
Conclusion
Myxococcus xanthus has been a heavily studied bacteria for many may years now. It is studied so much because there is still so much unknown information about how they live. Myxococcus xanthus serves as a model organism due to the complexity of their genome and the fact that the cells are able to metamorphically adapt to different environments and situations.
The processes of killing prey, communication between cells, and the exact way in which the cells are able to move are still not entirely known. Recent studies are discovering more and more information about the way in which they perform certain tasks such as identifying offspring or siblings and then being able to communicate and link up with that cell while also killing cells that are not the same species or strain. It is also amazing that these small Myxococcus xanthus cells are able to listen and eavesdrop on other cells communicating with each other in the immediate area in order to try to kill and lyse them for their nutrients. There are currently only 58 known myxobacteria species that have been discovered. This is itself is very surprising and gives hope to the fact that new species could easily be found in soil environments just like Myxococcus xanthus and possibly will have similar structure and methods of growing.
The common nickname for Myxococcus xanthus is the “wolf pack”. This is due to their behaviors of swarming and hunting together as packs as opposed to hunting by themselves. They seem to have such a complex social network system due to the way cells link with each other.
Future studies of Myxococcus xanthus could find that they could be very useful in our daily lives. Whether that’s exposing Myxococcus xanthus to crops to control for unwanted bacteria that could damage food or whether its exposing Myxococcus xanthus to unwanted bacteria in a healthcare setting which could possibly help treat potential diseases. The possibility to purposely mutate the Myxococcus xanthus cells have not been talked about much but could have positive consequences. There is so little known about not just Myxococcus xanthus but the entire family of Myxococcus because there has only been 11 different species discovered. Future studies will hopefully let us understand the mechanisms of multicellularity vs single cell structure of Myxococcus xanthus as well as a deeper look into the formation of the fruiting body and why it evolved to make that structure.
- ↑ Thaxter, R On the Myxobacteriaceae, a new order of Schizomycetes. Botanical Gazette. 1892, 17:12, 389-406.
- ↑ Janssen GR, Wireman JW, Dworkin M Effect of temperature on the growth of Myxococcus xanthus. Journal of Bacteriology. 1977, 130(1) 561-562.
- ↑ Shimkets L.J., Dworkin M., Reichenbach H. (2006) The Myxobacteria. In: Dworkin M., Falkow S., Rosenberg E., Schleifer KH., Stackebrandt E. (eds) The Prokaryotes. Springer, New York, NY.
- ↑ Wolgemuth CW, Igoshin O, Oster, G. The Motility of Mollicutes. Biophysical Journal. 2003, 85(2)828:842.
- ↑ Zhou, X.‐w., Li, S.‐g., Li, W., Jiang, D.‐m., Han, K., Wu, Z.‐h. and Li, Y.‐z. (2014), Predominant soil myxobacteria. Environmental Microbiology Reports, 6: 45-56.
- ↑ Stuart Huntley, Nils Hamann, Sigrun Wegener-Feldbrügge, Anke Treuner-Lange, Michael Kube, Richard Reinhardt, Sven Klages, Rolf Müller, Catherine M. Ronning, William C. Nierman, Lotte Søgaard-Andersen, Comparative Genomic Analysis of Fruiting Body Formation in Myxococcales, Molecular Biology and Evolution, Volume 28, Issue 2, February 2011, Pages 1083–1097.
- ↑ B. S. Goldman, W. C. Nierman, D. Kaiser, S. C. Slater, A. S. Durkin, J. A.Eisen, C. M. Ronning, W. B. Barbazuk, M. Blanchard, C. Field, C. Halling, G.Hinkle, O. Iartchuk, H. S. Kim, C. Mackenzie, R. Madupu, N. Miller, A.Shvartsbeyn, S. A. Sullivan, M. Vaudin, R. Wiegand, H. B. Kaplan. Evolution of sensory complexity recorded in a myxobacterial genome. Proceedings of the National Academy of Sciences Oct 2006, 103 (41) 15200-15205
- ↑ Krug D, Zurek G, Revermann O, Vos M, Velicer GJ, Müller R. Discovering the hidden secondary metabolome of Myxococcus xanthus: a study of intraspecific diversity. Appl Environ Microbiol. 2008;74(10):3058-3068. doi:10.1128/AEM.02863-07.
- ↑ Rosenberg E, Keller KH, Dworkin M. Cell density-dependent growth of Myxococcus xanthus on casein. J Bacteriol. 1977 Feb;129(2):770-7. doi: 10.1128/JB.129.2.770-777.1977. PMID: 402357; PMCID: PMC235010.
- ↑ White, D. 1993. Myxospore and fruiting body morphogenesis, p. 307-332. In M. Dworkin and D. Kaiser (ed.), Myxobacteria II. ASM Press, Washington, D.C.
- ↑ Reichenbach, H. 1966. Myxococcus spp. (Myxobacteriales). Schwarmentwicklung und Bildung von Protocysten. Publikationen zu wissenschafilichen Filmen, Gottingen. lA:557-578.
- ↑ 12.0 12.1 Kaiser, D., and R. Welch. 2004. Dynamics of fruiting body morphogenesis. J Bacteriol 186:919-27.
- ↑ O'Connor, K. A., and D. R. Zusman. 1991. Development in Myxococcus xanthus involves differentiation into two cell types, peripheral rods and spores. J Bacteriol 173:3318-33.
- ↑ Wireman, J. W., and M. Dworkin. 1975. Morphogenesis and developmental interactions in myxobacteria. Science 189:516-23.
- ↑ Rosenbluh, A., and E. Rosenberg. 1993. Developmental lysis and autocides, p. 213-233. In M. Dworkin and D. Kaiser (ed.), Myxobacteria II. American Society of Microbiology, Washington, D. C.
- ↑ L J Shimkets Correlation of energy-dependent cell cohesion with social motility in Myxococcus xanthus. Journal of Bacteriology Jun 1986, 166 (3) 837-841
- ↑ Diodati ME, Gill RE, Plamann L, Singer M. 2008. Initiation and early developmental events. In: Whitworth D. E (Ed). Myxobacteria: Multicellularity and Differentiation. ASM Press.
- ↑ GARDNER, A. and WEST, S.A. (2006), Demography, altruism, and the benefits of budding. Journal of Evolutionary Biology, 19: 1707-1716.
- ↑ Ward MJ, Lew H, Zusman DR. Disruption of aldA Influences the Developmental Process in Myxococcus xanthus. J. Bacteriol. 2000, 182(2):546.
- ↑ Keane R, Berleman J. The predatory life cycle of Myxococcus xanthus. Microbiology. 2016, 162:1.
- ↑ 21.0 21.1 Adrien Ducret, Marie-Pierre Valignat, Fabrice Mouhamar, Tâm Mignot, OlivierTheodoly Bacterial migration tracks at a glance. Proceedings of the National Academy of Sciences Jun 2012, 109 (25) 10036-10041.
- ↑ Dale Kaiser, Hans Warrick Myxococcus xanthus Swarms Are Driven by Growth and Regulated by a Pacemaker. Journal of Bacteriology Oct 2011, 193 (21) 5898-5904.
- ↑ Berleman JE, Scott J, Chumley T, Kirby JR. Predataxis behavior in Myxococcus xanthus. Proc Natl Acad Sci U S A. 2008;105(44):17127-17132.
- ↑ Thiery S, Kaimer C. The Predation Strategy of Myxococcus xanthus. Front Microbiol. 2020 Jan 14;11:2.
- ↑ Pham VD, Shebelut CW, Mukherjee B, Singer M. RasA is required for Myxococcus xanthus development and social motility. J Bacteriol. 2005;187(19):6845-6848. doi:10.1128/JB.187.19.6845-6848.2005.
- ↑ Morgan AD, MacLean RC, Hillesland KL, Velicer GJ. Comparative analysis of myxococcus predation on soil bacteria. Appl Environ Microbiol. 2010 Oct;76(20):6920-7.
- ↑ Mark J. McBride, David R. Zusman, Behavioral analysis of single cells of Myxococcus xanthus in response to prey cells of Escherichia coli, FEMS Microbiology Letters, Volume 137, Issue 2-3, April 1996, Pages 227–231.
- ↑ Dey A, Vassallo CN, Conklin AC, Pathak DT, Troselj V, Wall D. Sibling Rivalry in Myxococcus xanthus Is Mediated by Kin Recognition and a Polyploid Prophage. J Bacteriol. 2016 Jan 19;198(6):994-1004.
- ↑ Livingstone PG, Morphew RM, Whitworth DE. Myxobacteria Are Able to Prey Broadly upon Clinically-Relevant Pathogens, Exhibiting a Prey Range Which Cannot Be Explained by Phylogeny. Front Microbiol. 2017;8:1593. Published 2017 Aug 22.
- ↑ Copenhagen, K., Alert, R., Wingreen, N.S. et al. Topological defects promote layer formation in Myxococcus xanthus colonies. Nat. Phys. 17, 211–215 (2021).
- ↑ Kirstin I. Arend, Janka J. Schmidt, Tim Bentler, Carina Lüchtefeld, Daniel Eggerichs, Hannah M. Hexamer, Christine Kaimer Myxococcus xanthus predation of Gram-positive or Gram-negative bacteria is mediated by different bacteriolytic mechanisms. Applied and Environmental Microbiology Dec 2020, AEM.02382-20.
- ↑ Velicer GJ, Yu YT. Evolution of novel cooperative swarming in the bacterium Myxococcus xanthus. Nature. 2003 Sep 4;425(6953):75-8.
- ↑ Nair RR, Vasse M, Wielgoss S, Sun L, Yu YN, Velicer GJ. Bacterial predator-prey coevolution accelerates genome evolution and selects on virulence-associated prey defences. Nat Commun. 2019 Sep 20;10(1):4301.
- ↑ Dworkin M. Tactic behavior of Myxococcus xanthus. J Bacteriol. 1983 Apr;154(1):452-9.
