Rhodanobacter denitrificans
Classification
Taxa
Bacteria; Proteobacteria; Gammaproteobacteria; Xanthomonadales; Xanthomonadaceae; Rhodanobacter; denitrificans [8].
Strains
2APBS1 and 116-2 [8].
Description
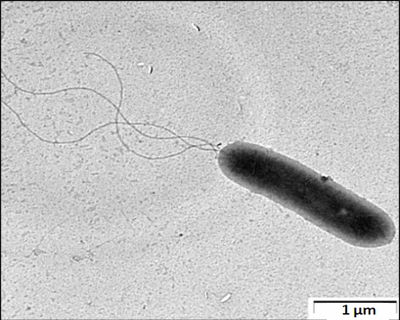
Rhodanobacter denitrificans is a yellow-pigmented, gram-negative, non-sporulating, motile,slow-growing, and facultatively anaerobic rod-shaped bacterium with the ability to perform complete denitrification [8]. For Rhodanobacter species, denitrification was not a characteristic until the discovery of R. denitrificans [6]. Denitrification is the conversion of nitrate to N2 through nitrite, nitric and nitrous oxide intermediates catalyzed by microbial enzymes [11]. Only two species of Rhodanobacter, one of them being Rhodanobacter denitrificans, is capable of this process [8]. In the absence of oxygen, growth is fueled by the use of nitrate, nitrite, and nitrous oxide as electron acceptors [8]. Rhodanobacter denitrificans has a salt tolerance up to 2% NaCl [8]. Growth was observed at pH 4-8 and 10-35ºC, optimally at pH 6.5 and 30ºC [8]. The two strains of R. denitrificans, 2APBS1 and 116-2, were found at Oak Ridge Integrated Field Research Challenge (ORIFRC) [8]. ORIFRC is a model nuclear legacy site with high levels of nitrate and acidity, with uranium and other heavy metal contaminants [8]. At ORIFRC, 2APBS1 was isolated from Area 2 (pH 6-7 and nitrate level <2 mM) and 116-2 was isolated from Area 3 (pH 3-4 and nitrate level 10s to 100s mM) [8]. The two strains are highly abundant and active in acidic, nitrate rich subsurface environments with high metal (e.g. uranium) concentrations [8]. The characteristic of survival in these harsh environments allow R. denitrificans to conduct bioremediation of the contamination sites by denitrification [8].
Ecology and Metabolism
Microbes can reduce contaminants in the subsurface ecosystem, such as in ORIFRC [5]. Nitrate concentration is inversely proportional to pH [11]. High levels of nitrate inhibits most microbes, but R. denitrificans can thrive in a nitrate rich environment conducting denitrification, which is important in subsurface ecology [5]. Low pH, high nitrate, and other co-contaminants cause strong selective pressure on the bacterial community [5]. Selection minimizes microbial diversity and leads to the survival of dominant species such as R. denitrificans [1]. pH is the strongest determination factor of abundance for Rhodanobacter [5]. Rhodanobacter thrives in conditions of high nitrate and uranium, and low pH [5]. Also, electron donors are the limiting factors of denitrification; nitrate levels exceed the number of electron donors [5]. The deaths of other native bacteria in the acidic subsurface led to favoring of horizontal gene transfers, giving complete denitrification capability and acid tolerance to R. denitrificans [5]. R. denitrificans also have a flexible metabolism and able to grow on many organic substrates [5]. They grow extremely well on acetate (great carbon source and electron donor), used to stimulate denitrification and neutralize liquid waste at ORIFRC [5]. All in all, low pH groundwater and high nitrate levels created a specific niche for Rhodanobacter denitrificans [5].
Significance in Bioremediation
Bioremediation is a cost effective process that microbes use to remove pollutants from the environment by enzymatically transforming the contaminants [2]. It is a natural solution to health and ecological impacts, and leads to nontoxic end-products such as N2 and precipitation of metals contaminants [2]. Contamination of aquifers is a major problem because clean water is an important resource for agriculture and industry [2]. For many countries, groundwater is a major source of drinking water [2]. Nitrate is one of the most widespread groundwater contaminants [2]. In ORIFRC, nitrate contamination is accompanied with uranium, which has no function biologically and is toxic to most bacteria and humans [12]. It has two oxidation states, one being the soluble U(VI) uranyl ion and the other is extremely insoluble U(IV) precipitate [12]. Uranium contamination is associated with the production of nuclear weapons, and is widespread across the world [1]. The growth of R. denitrificans at low pH is important because uranyl ion exists in the most toxic form at low pH [12]. Reductive immobilization of U(VI) is a currently favoured bioremediation process [9]. U(VI) contaminates aquifers and drinking water rapidly [9]. Nitrate is a competitive electron acceptor in the subsurface so U(VI) reduction is inhibited by its presence, and the ORIFRC research operation is used to test this correlation [11]. Therefore, uranium precipitation is most efficient when nitrate is depleted [10]. Rhodanobacter denitrificans effectively reduces nitrate in environments with low pH and high levels of nitrate and heavy metals. In ORIFRC, bioremediation of aquifers co-contaminated with nitrate and uranium can be stimulated by adding organic electron donors such as acetate [11]. Acetate facilitates the removal of U(VI) from contaminated aquifers [9], by U(VI) reduction which is strictly a biological reaction by microbes [3]. R. denitrificans are also able to reduce the emission of N2O from low pH soils, which is a strong greenhouse gas and a denitrification intermediate that depletes ozone and causes global warming [13]. This is due to their capability to undergo complete denitrification, therefore reducing nitrate and other harmful intermediates to N2 which is released into air [13].
Genome
The complete genome of R. denitrificans 2APBS1 is 4.23 Mb and 116-2 is 3.98 Mb [8]. 2APBS1 strain contains nitrate, nitrite, nitric oxide, and nitrous oxide reductase genes [4]. Comparing 16S rRNA gene sequences, the two strains have high sequence similarity to R. thiooxydans LCS2; 99.2% match for 2APBS1 and 98.7% match for 116-2 [8]. LCS2 is a thiosulfate-oxidizing bacterium with almost the same phenotype as Rhodanobacter denitrificans [7]. However, nitrate is reduced, but nitrite is not for this bacterium [7]. The highest rRNA sequence similarity is between the two strains, with a 99.4% match [8]. Using DNA-DNA hybridization, results show that 2APBS1 has the highest DNA similarity of 86% to 116-2, whereas the next two closest only have 56.7% (R. soli) and 41.1% (R. thiooxydans) similarity [8]. Overall, knowing that the DNA-DNA hybridization threshold is 70% for determining species, and with the genus-unique capability of denitrification, the two strains represent a novel species of Rhodanobacter with the proposed name Rhodanobacter denitrificans [8].
References
[1] Akob, D.M., Mills, H.J., and Kostka, J.E. "Metabolically active microbial communities in uranium-contaminated subsurface sediments." FEMS Microbiology Ecology, 2006, DOI: 10.1111/j.1574-6941.2006.00203.x
[2] Atlas, R.M., and Philp, J. "Bioremediation: Applied Microbial Solutions for Real-World Environmental Cleanup." American Society for Microbiology, 2005, DOI: not given
[3] Finneran, K.T., Anderson, R.T., Nevin, K.P., and Lovley, D.R. "Potential for Bioremediation of Uranium-Contaminated Aquifers with Microbial U(VI) Reduction." Soil and Sediment Contamination: An International Journal, 2010, DOI: 10.1080/20025891106781
[4] Green, S.J., Prakash, O., Gihring, T.M., Akob, D.M., Jasrotia, P., Jardine, P.M., Watson, D.B., Brown, S.D., Palumbo, A.V., and Kostka, J.E. "Denitrifying Bacteria Isolated from Terrestrial Subsurface Sediments Exposed to Mixed-Waste Contamination." Applied and Environmental Microbiology, 2010, DOI: 10.1128/AEM.03069-09
[5] Green, S.J., Prakash, O., Jasrotia, P., Overholt, W.A., Cardenas, E., Hubbard, D., Tiedje, J.M., Watson, D.B., Schadt, C.W., Brooks, S.C., and Kostka, J.E. "Denitrifying Bacteria from the Genus Rhodanobacter Dominate Bacterial Communities in the Highly Contaminated Subsurface of a Nuclear Legacy Waste Site." Applied and Environmental Microbiology, 2012, DOI: 10.1128/AEM.06435-11
[6] Kostka, J.E., Green, S.J., Rishishwar, L., Prakash, O., Katz, L.S., Marino-Ramirez, L., Jordan, I.K., Munk, C., Ivanova, N., Mikhailova, N., Watson, D.B., Brown, S.D., Palumbo, A.V., and Brooks, S.C. "Genome sequences for six Rhodanobacter strains, isolated from soils and the terrestrial subsurface, with variable denitrification capabilities." Journal of Bacteriology, 2012, DOI: 10.1128/JB.00871-12
[7] Lee, C.S., Kim, K.K., Aslam, Z., and Lee, S. "Rhodanobacter thiooxydans sp. nov., isolated from a biofilm on sulfur particles used in an autotrophic denitrification process." International Journal of Systematic and Evolutionary Microbiology, 2007, DOI: 10.1099/ijs.0.65086-0
[8] Prakash, O., Green, S.J., Jasrotia, P., Overholt, W.A., Canion, A., Watson, D.B., Brooks, S.C., and Kostka, J.E. "Rhodanobacter denitrificans sp. nov., isolated from nitrate-rich zones of a contaminated aquifer." International Journal of Systematic and Evolutionary Microbiology, 2012, DOI: 10.1099/ijs.0.035840-0
[9] Williams, K.H., Long, P.E., Davis, J.A., Wilkins, M.J., N'Guessan, A.L., Steefel, C.I., Yang, L., Newcomer, D., Spane, F.A., Kerkhof, L.J., McGuinness, L., Dayvault, R., and Lovley, D.R. "Acetate Availability and its Influence on Sustainable Bioremediation of Uranium-Contaminated Groundwater." Geomicrobiology Journal, 2011, DOI: 10.1080/01490451.2010.520074
[10] Shelobolina, E.S., O'Neill, K., Finneran, K.T., Hayes, L.A., and Lovley, D.R. "Potential for In Situ Bioremediation of a Low-pH, High-Nitrate Uranium-Contaminated Groundwater." Soil and Sediment Contamination: An International Journal, 2010, DOI: 10.1080/714037720
[11] Spain, A.M., and Krumholz, L.R. "Nitrate-Reducing Bacteria at the Nitrate and Radionuclide Contaminated Oak Ridge Integrated Field Research Challenge Site: A Review." Geomicrobiology Journal, 2011, DOI: 10.1080/01490451.2010.507642
[12] Suzuki, Y., and Banfield, J.F. "Resistance to, and Accumulation of, Uranium by Bacteria from a Uranium-Contaminated Site." Geomicrobiology Journal, 2010, DOI: 10.1080/01490450490266361
[13] van den Heuvel, R.N., van der Biezen, E., Jetten, M.S.M., Hefting, M.M., and Kartal, B. "Denitrification at pH 4 by a soil-derived Rhodanobacter-dominated community." Environmental Microbiology, 2010, DOI: 10.1111/j.1462-2920.2010.02301.x

