Shock chlorination
Introduction
From swimming pools to wells, chlorine is a common chemical used to disinfect water sources. About a century ago, the process of chlorination was introduced in order to prevent the spread of diseases such as typhoid or cholera. Over time, the process has modified, with the EPA (Environmental Protection Agency) beginning to implement the use of chloramine, a closely related compound, instead1.
Due to safety concerns, hypochlorite (bleach) is the most commonly used compound to conduct shock chlorination2. Hypochlorite is used in one of three forms: commercial bleach (approx. 3.5-5% concentration), calcium hypochlorite (Ca(OCl)3; 65-70% concentrated), or sodium hypochlorite (NaOCl; about 12% concentration)2. These forms of hypochlorite are not as pure as chlorine gas, and will degrade in strength when in storage. However, like chlorine gas, they also react with water to form a disinfectant, hypochlorous acid (HOCl).
Microbial agents
Frequently, microbial factors infiltrate water sources through fecal matter. Many types of bacterial pathogens can initiate waterborne illnesses, including enteric bacteria, protozoa, or viruses4.
Due to the variety of species that can inhabit water sources, it comes as no surprise that chlorine has varying affects on the types of microbes. Though time and concentration can eliminate virtually every species, some species remain resistant to the process. Species such as Vibrio cholera have been tested to last approximately 30 days in drinking water sources, while toxigenic E.coli can last 90 days5. Survival techniques include cysts, spores, absorption, and intracellular survival and growth. Other species will remain viable, but no longer culturable.
Helicobacter pylori
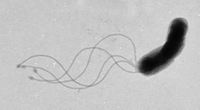
Helicobacter pylori is known to cause gastritis and peptic ulcers.
Studies done in Peru6 and Japan7 have shown the presence of the bacteria in public water sources, proving its possibility as a waterborne microbe.
Cryptosporidium
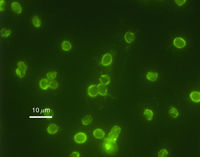
Cryptosporidium parvum is a type of parasite capable of causing gastrointestinal illness. Unlike Helicobacter pylori, however, Cryptosporidium has been proven to be unresponsive to most chlorination8.
In 1993, Milwaukee (Wisconsin) experienced one of the worst waterborne outbreaks after one of the city's water plants failed to remove Cryptosporidium oocysts from the Lake Michigan water collected for public use. Over 403,000 residents and visitors were affected, experiencing the watery diarrhea, nausea, fever, vomiting, and abdominal cramps associated with infection. Approximately 54 deaths occurred, although there is some debate regarding other preexisting conditions that affected patient survival9.
In October 2013, a resurgence of the Cryptosporidium protozoan appeared in Milwaukee, this time in local swimming pools10.
Methods

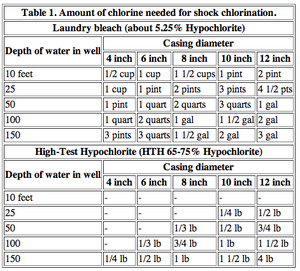
Shock chlorination of drinking water may be recommended for domestic use due to several different problems. In some instances, groundwater will be infiltrated by bacteria, requiring consistent disinfecting. Occasionally, shock chlorination may be an isolated necessity due to some foreign contamination during installation or repairs to a pump. Unlike the process for drinking water, which requires an initial shock and subsequent allowance of the chlorine concentration to drop, pool treatment usually requires a constant level to be maintained (barring some extreme contamination, such as waste or vomit). For example, the recommended amount of chlorine for disinfecting drinking water is 200 ppm, while swimming pools are usually maintained from 1.0-3.0 ppm11.
Safety during domestic shock chlorination requires protective gear such as gloves, goggles, and other clothing items, such as an apron. Concentrated chlorine levels are extreme irritants, so contact with skin should be avoided. Mixing chlorine bleach with other chemicals may produce toxic fumes. Thus, ensure that the area is well ventilated before beginning12.
Success rates
In 1998, using Raman and ultraviolet spectroscopic radiation, scientists proved that the bactericidal activity of chlorine treatment was quantitatively linked to the concentration of the hypochlorous acid in the chemical solution. Further, the pH dependence discovered indicates an equilibrium amongst the chlorine, hypochlorous, and hypochlorite13. Though typically chlorine activity decreases at higher pH, compromise with other factors (such as prevention of skin and eye irritation) places the ideal pH at around 7.2-7.814.
DNA microarray can be used to monitor water quality more precisely15. A study in 2011 established 26 different microbial probes, and claimed to drastically reduce the time and labor required with current methods16.
The success of shock chlorination, however, is not that simple. Due to the process of horizontal gene transfer (in which bacteria can pick up DNA from other species), some species of bacteria previously susceptible to chlorine are evolving to be resistant. Natural selection may also cause higher fitness in organisms that have mutations that allow higher survival in chlorine. For instance, scientists have discovered two forms of Cholera, rugose (referencing its wrinkly texture) and smooth. Rugose colonies, unlike smooth, are capable of producing a slime covering. This covering consists of long polysaccharides. Experimentation demonstrated that these colonies can withstand chlorine concentration ten to twenty times higher than their counterparts17, suggesting a situation similar to MRSA bacteria and antibiotics.
Related and Alternative methods
Various groups complain about the toxic residues created by chlorination. However, various alternatives that have been created appear to have faults of their own, producing the carcinogenic compounds similar to the ones scientists sought to remove. This is where other methods are used, such as carbon filtration and enhanced coagulation, physical-chemical processes, though this is expensive18.
Bromine is used as a disinfectant in swimming pools. However, unlike chlorine, it cannot be used to disinfect drinking water19. In contrast, the EPA references three different oxidizing disinfectants for drinking water: chlorine, chloramines, and ozone. Monochloramine, NH2Cl, is formed during a reaction between ammonia and chlorine (a process called chloramination). Ozone is a newer method, but offers greater levels of oxidation with quicker decomposition. In a study done in 1997, it was found that H.pylori and E.coli respond similarly to hypochlorous acid20. However, a different study in 2002 demonstrates the imperfection of any of the types suggested by the EPA. H.pylori is more resistant to chlorine and ozone than E.coli, but less resistant to monochloramine21.
One compound that seems to have the greatest success rate, chlorine dioxide, is capable of even killing Cryptosporidium. However, the compound is extremely expensive to make, and must be produced on site, making it inefficient22.
References
1 "Chlorine Substitutes In Water May Have Risks". 7 January 2011. NPR.
11 Dozier M., McFarland M. "Shock Chlorination of Stored Water Supplies". AgriLife Extension.
12 Wright M. "Disinfecting a Domestic Well with Shock Chlorination". October 1997.
14 "Your Disinfection Team: Chlorine & pH". Centers for Disease Control and Prevention.
18 Sedlak D., von Gunten U. "The Chlorine Dilemma". January 2011. Science. 331(6013):42-43
Edited by Erika Jensen, student of Joan Slonczewski for BIOL 116 Information in Living Systems, 2013, Kenyon College.
