Vibrio cholerae pathogenesis: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
[http://en.wikipedia.org/wiki/Vibrio_cholerae <i>Vibrio cholerae</i>] is the causative agent of the diarrheal disease [http://en.wikipedia.org/wiki/Cholera cholera]. A disease affecting 2.8 million people per year and resulting in the deaths of 91,000, cholera is most common in areas with high population density and low sanitation quality.<sup>[1] [2]</sup> Not all <i>V. cholerae</i> are pathogenic: only two strains of [http://en.wikipedia.org/wiki/Serotype serogroups] O1 and O139 cause cholera.<sup>[1]</sup> Furthermore, it is considered a facultative human pathogen, as it primarily inhabits [http://en.wikipedia.org/wiki/Surface_water surface waters], as opposed to the [http://en.wikipedia.org/wiki/Small_intestine small intestine].<sup>[3]</sup> | [http://en.wikipedia.org/wiki/Vibrio_cholerae <i>Vibrio cholerae</i>] is the causative agent of the diarrheal disease [http://en.wikipedia.org/wiki/Cholera cholera]. A disease affecting 2.8 million people per year and resulting in the deaths of 91,000, cholera is most common in areas with high population density and low sanitation quality.<sup>[1] [2]</sup> Not all <i>V. cholerae</i> are pathogenic: only two strains of [http://en.wikipedia.org/wiki/Serotype serogroups] O1 and O139 cause cholera.<sup>[1]</sup> Furthermore, it is considered a facultative human [https://en.wikipedia.org/wiki/Pathogen pathogen], as it primarily inhabits [http://en.wikipedia.org/wiki/Surface_water surface waters], as opposed to the [http://en.wikipedia.org/wiki/Small_intestine small intestine].<sup>[3]</sup> | ||
<br> | <br> | ||
<br>When entering and colonizing the human host, <i>V. cholerae</i> must endure changing environmental factors such as temperature, acidity, osmolarity, intestinal growth inhibitory substances, and immune system factors.<sup>[1]</sup> After sufficient colonization, an autoregulatory loop controlling ToxT, [http://en.wikipedia.org/wiki/Cholera_toxin cholera toxin], and the toxin-coregulated pilus (TCP) results.<sup>[4]</sup> Understanding pathogenesis of <i>V. cholerae</i> requires the understanding of colonization and this loop. | <br>When entering and colonizing the human host, <i>V. cholerae</i> must endure changing environmental factors such as temperature, acidity, osmolarity, intestinal growth inhibitory substances, and immune system factors.<sup>[1]</sup> After sufficient colonization, an autoregulatory loop controlling ToxT, [http://en.wikipedia.org/wiki/Cholera_toxin cholera toxin], and the toxin-coregulated pilus (TCP) results.<sup>[4]</sup> Understanding pathogenesis of <i>V. cholerae</i> requires the understanding of colonization and this loop. | ||
Revision as of 17:21, 26 March 2015
Vibrio cholerae is the causative agent of the diarrheal disease cholera. A disease affecting 2.8 million people per year and resulting in the deaths of 91,000, cholera is most common in areas with high population density and low sanitation quality.[1] [2] Not all V. cholerae are pathogenic: only two strains of serogroups O1 and O139 cause cholera.[1] Furthermore, it is considered a facultative human pathogen, as it primarily inhabits surface waters, as opposed to the small intestine.[3]
When entering and colonizing the human host, V. cholerae must endure changing environmental factors such as temperature, acidity, osmolarity, intestinal growth inhibitory substances, and immune system factors.[1] After sufficient colonization, an autoregulatory loop controlling ToxT, cholera toxin, and the toxin-coregulated pilus (TCP) results.[4] Understanding pathogenesis of V. cholerae requires the understanding of colonization and this loop.
Colonization of the Human Small Intestine
Biofilm Formation
V. cholerae’s journey through the acidic stomach is aided by its assembly into a biofilm, a structure consisting of bacteria in a matrix of sugars and proteins.[1] [3] 100% of V. cholerae cells in biofilms have been shown to withstand acid shock characteristic of the human stomach. Exposure to an environment with a pH of 4.5 for 15 to 30 minutes improves cell survival 50 and 1000-fold, respectively, compared to non-biofilm-associated cells.[5]
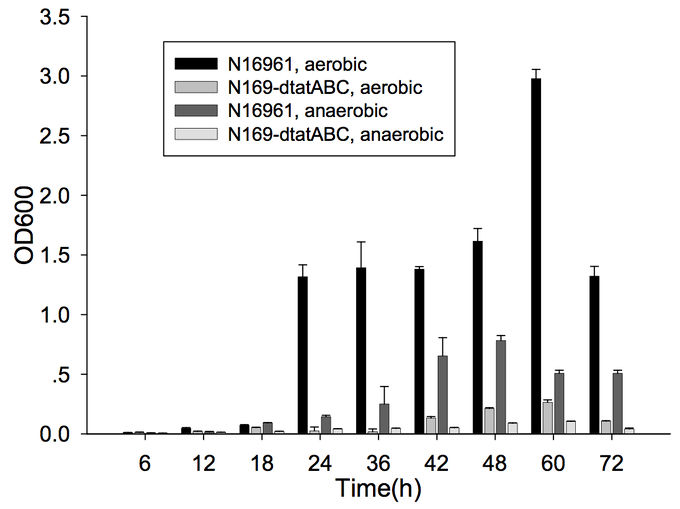
Furthermore, cells capable of forming biofilms have greater intestinal colonization capacity in the suckling mouse model. The rugose variant of V. cholerae, which has superior biofilm-forming abilities compared to the smooth variant, is also more successful at colonizing the intestine.[6] In addition, the twin-arginine translocation (Tat) system has also been shown to be important for biofilm formation and for colonization. The Tat system is an export system that recognizes the twin arginine signal peptide on proteins that are intended to be translocated across the cell membrane (these are often enzymes involved in the electron transport chain). This system has already been associated with virulence of Pseudomonas aeruginosa, Escherichia coli, Yersinia pseudotuberuclosis, and other pathogenic bacteria. The Tat system has recently been shown to be functional in V. cholerae; deletion of tatABC causes a decrease in biofilm formation (Figure 1) as well as in colonization of the suckling mouse.[7] Finally, the absence of vibrio polysaccharides (encoded by the vps operon), the major exopolysaccharide component of biofilms, or the absence of RbmA, one of the matrix proteins, leads to defective colonization.[6]
However, quorum sensing studies in V. cholerae have shown that the biofilm is not necessary during colonization, but instead during the bacteria’s travel through the acidic stomach, during which it needs a protective structure. Once they reach the intestine, they will then utilize virulence factors, not the biofilm, to colonize and infect the host.
Quorum Sensing
Once in the intestine, the biofilm is no longer of help to V. cholerae, and a new mechanism is necessary for colonization. Quorum sensing, a bacterial mechanism of communication with downstream effects on gene regulation based on population density, is what allows the bacteria to colonize the intestine by expressing virulence factors, such as cholera toxin (CT) and the toxin-coregulated pilus (TCP).[8]
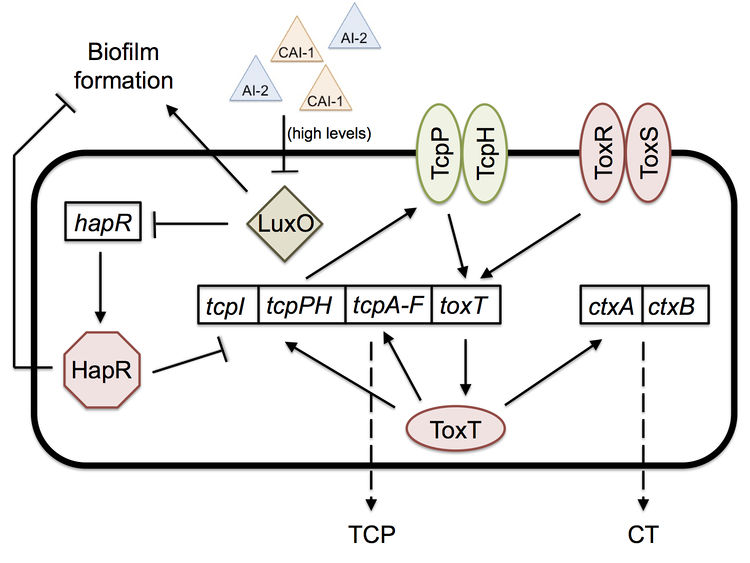
Two important quorum-sensing related proteins of note are LuxO and HapR, which work together in a system similar to that of Vibrio harveyi, the most common bacterium used in quorum sensing studies.[9] LuxO is a regulator that takes the information provided by levels of autoinducer 2 (AI-2) and cholera autoinducer 1 (CAI-1) – detected by LuxPQ and CqsS membrane receptors, respectively – and represses the master transcription factor HapR.[10] [11] In V. harveyi, this activation of LuxO and repression of HapR leads to a decrease in luminescence (as HapR in the bacterium transcriptionally activates the luciferase operon). V. cholerae does not have a bioluminescence system and HapR transcriptionally represses virulence genes.[11] When LuxO is active, it represses expression of hapR, thus allowing the transcription of tcpP, a gene necessary for the biosynthesis (though not itself a building block) of the TCP. CT (responsible for the diarrhea characteristic of cholera) expression is dependent upon the presence of TCP, so active LuxO and inactive HapR result in an increased amount of both TCP and CT.[11] LuxO and HapR’s effect on virulence has been demonstrated in the CD-1 suckling mouse model: deletion of luxO results in no cell recovery from the intestine of the biopsied mouse (no colonization occurred), while deletion of hapR does not affect virulence and colonization at all.[11] Furthermore, toxigenic strains of V. cholerae have hapR mutations, which may serve to improve their colonization of humans by increasing TCP and CT levels.
Quorum sensing and biofilm formation have been shown to have a reciprocal relationship in V. cholerae, unlike in most other biofilm-forming species.[5] The current working model of colonization is that the biofilm is necessary to protect V. cholerae as it goes through the acidic stomach environment, since it has been shown to increase tolerance to acid shock.[5] However, once the bacterium is in the small intestine, in which it no longer needs the biofilm’s protection, the high density of cells causes high levels of quorum sensing autoinducers. High levels of the autoinducer CAI-1 represses LuxO and activates HapR, which then represses transcription of the vps operon, thus preventing further biofilm formation and improving the bacterium’s colonization ability; hapR mutants have shown to have thicker biofilms with 10-fold lower colonization in suckling mice.[5] The cells disperse from the biofilm and as a result have lower levels of autoinducers. Low levels allow the activation of LuxO, the repression of HapR, and thus the expression of tcpP and the ctx operon.[5] [11] Production of TCP and CT allow for infection to occur. Once enough V. cholerae accumulate throughout the infection, high levels of autoinducers will repress TCP and CT production, thus activating protease production, which leads to the detachment of bacteria from the intestinal mucosa and their exit from the host.[5]
Toxin-Coregulating Pilus
Cholera Toxin
ToxR and ToxT Regulon
References
[1] Reidl, J. and K.E. Klose, Vibrio cholerae and cholera: out of the water and into the host. Fems Microbiology Reviews, 2002. 26: p. 125-139.
[2] Ali, M., et al., The global burden of cholera. Bulletin of the World Health Organization, 2012. 90: p. 209-218.
[3] Sack, D.A., et al., Cholera. Lancet, 2004. 363: p. 223-233.
[4] Yu, R.R., and V.J. DiRita, Analysis of an autoregulatory loop controlling ToxT, cholera toxin, and toxin-coregulated pilus production in Vibrio cholerae. Journal of Bacteriology, 1999. 181: p. 2584-2592.
[5] Zhu, J. and J.J. Mekalanos, Quorum sensing-dependent biofilms enhance colonization in Vibrio cholerae. Developmental Cell, 2003. 5: p. 647-656.
[6] Fong, J.C.N., et al., Role of Vibrio polysaccharide (vps) genes in VPS production, biofilm formation and Vibrio cholerae pathogenesis. Microbiology, 2010. 156: p. 2757-2769.
[7] Zhang, L., et al., Pleiotropic effects of the twin-arginine translocation system on biofilm formation, colonization, and virulence in Vibrio cholerae. Biomed Central Microbiology, 2009. 9: p. 1-13.
[8] Camilli, A. and B.L. Bassler, Bacterial small-molecule signaling pathways. Science, 2006. 311: p. 1113-1116.
[9] Miller, M.B., et al., Parallel quorum sensing systems converge to regulate virulence in Vibrio cholerae. Cell, 2002. 110: p. 303-314.
[10] Higgins, D.A., et al., The major Vibrio cholerae autoinducer and its role in virulence factor production. Nature, 2007. 450: p. 883-886.
[11] Zhu, J., et al., Quorum-sensing regulators control virulence gene expression in Vibrio cholerae. Proceedings of the national Academy of Sciences USA, 2002. 99: p. 3129-3134.
Edited by Tina Solvik, a student of Suzanne Kern in BIOL168L (Microbiology) in The Keck Science Department of the Claremont Colleges Spring 2015.