Viral Oncology: Difference between revisions
| Line 13: | Line 13: | ||
=Oncolytic Viruses= | =Oncolytic Viruses= | ||
<br>Oncolytic viruses are viruses that specifically target and kill cancer cells. This process usually relies on genetically modified viruses that | <br>Oncolytic viruses are viruses that specifically target and kill cancer cells. This process usually relies on genetically modified viruses that have lost their ability to replicate within somatic cells but maintain virulence in cancerous cells. Many of these cells are no longer able to infect somatic cells due to the loss or inactivation of genes that can suppress apoplectic mechanisms within the cell. Without these repression mechanisms the virus is only able to undergo the lytic cycle within cancer cells making them a potent and efficient method of cancer treatment. | ||
<br> | <br> | ||
Revision as of 18:45, 17 May 2016
Introduction
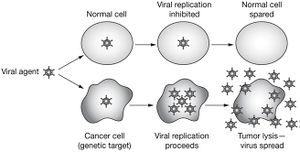
Viral oncology is a subsection of oncology concerned with the treatment of tumors with viral particles. While this field has gotten a lot of press in recent years, the idea of using viruses as oncolytic agents has been around since the early 1920’s, and the correlation between viruses and remission has been around singe the mid-to-late 1800s [1]. Most of the patients studied in the 1800s had blood-based cancers such as leukemia or lymphoma with significant immune suppression [3]. The most famous report of this type was made by Dr. Dock in 1904 [2] in which a 47 year old woman with myelogenous leukemia went into remission after a flu infection. Another more shocking case is that of a 4 year old boy with lymphatic leukemia who contracted chickenpox. Before his contraction of chickenpox, his liver, spleen and lymph nodes were all severely swollen and his leukocyte count was greatly elevated (200 cells/ul). After contracting chicken pox his liver and spleen returned to normal size and his white count fell back into normal levels (4.1 cells/ul). However, in both cases the remission was short lived and the cancer soon returned.
The first attempt to use viruses a curative was the treatment of chronic joint disease with hepatitis in 1897, with minimal success [1]. Despite the lack of success in the 1897 trials, in 1949 a clinical trial took place in which 22 patents with Hodgkin's lymphoma were treated with tissue samples from hepatitus positive individuals. While some did achieve remission for a short time, many in the study also contracted hepatitis B, and the study was discontinued [4]. A number of similar experiments were run throughout the 1950’s and 1960’s with minimal success.
Almost 50 years after the failed experiments of the 1950's and 60's a breakthrough came in the form of oncolytic adenovirus H101. This virus was approved by the Chinese Government for cancer treatments in 2005 and works by targeting p53 deficient cells (most cancers are p53 deficient)[5]. Today, thanks in large part to H101, a number of viral pathways are being investigated by medical researchers in an attempt to increase our cancer fighting toolbox.
Why Viruses?
While the medical community has been aware of the correlation between remission and viral infection since the mid-1800s, medical professionals were unsure as to why this correlation existed until we began to understand the molecular and genetic mechanisms behind cancer and cancer replication. Cancer cells are the product of small scale evolution, which is to say that the accumulation of point mutations and chromosomal shifts, along with chromosomal instability, have resulted in a phenotype drastically different from its ancestor. Typically, in cancer cells, we see a selection for growth advantages over somatic cells. These increased growth factors include immunity to density and anchorage dependence as well as up and down regulation of certain genes such as telomerase or p53.[6]
While these genetic and molecular changes result in an increase in fitness when compared to somatic cells, often times certain defensive mechanisms are sacrificed. For example in many cancer cells a number of genetic repair enzymes are shut off as well as proteins that defend against mutations in the genome and foreign DNA.
Without the ability to defend against foreign DNA many cancer cells are left vulnerable to viral attacks. By exploiting the innate viral sensitivity of most cancer cells scientists have been able to engineer viruses that can not only selectively kill cancer cells but also generate an cancer specific immune response within the patients body. [7]
Oncolytic Viruses
Oncolytic viruses are viruses that specifically target and kill cancer cells. This process usually relies on genetically modified viruses that have lost their ability to replicate within somatic cells but maintain virulence in cancerous cells. Many of these cells are no longer able to infect somatic cells due to the loss or inactivation of genes that can suppress apoplectic mechanisms within the cell. Without these repression mechanisms the virus is only able to undergo the lytic cycle within cancer cells making them a potent and efficient method of cancer treatment.
Adenovirus
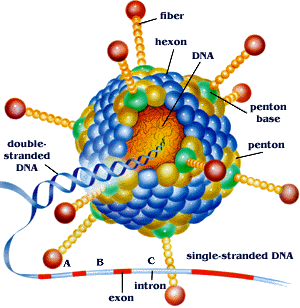
Adenoviruses are a class of nonenveloped (Without a lipid bilayer) viruses with a double stranded DNA genome. Adenoviruses enter the cell through endocytosis. Once in the virus enters the cell the capsid breaks apart and ruptures the endosome releasing its DNA into the host cell and eventually attaching to histone proteins to enable transcription and translation of viral genes. [8]
For an adenovirus to successfully replicate in a host cell a number of processes must be completed in succinct order. The most important of these events being the repression genes which lead to apoptosis or programmed cell death which if active would hinder the viruses ability to replicate within the cell. The adenovirus accomplishes this through 2 major gene products; E1B-19K and E1B-55K. E1B-19K does two major things in the host cell. [9] The first and most important function of this protein is its ability to bind and sequester BAK. BAK when activated induced apoptosis by binding to BAX and forming pores in the mitochondrial wall. Apoptogenic proteins then leech from the mitochondria ultimately releasing capase and lysing the cell. In addition, this protein works to stabilized viral and cellular DNA.[10]
E1B-55K functions on a p53 dependent pathway. p53, also known as the “Guardian of the Genome”, works to regulate the cell cycle, check for mutations in genes and initiate apoptosis. E1B-55K binds to p53 and adds a repression domain to the protein. This results in p53 acting as a repressor instead of an activator for the various apoptotic genes it would normally bind. Additionally, p53 with this repression domain has a binding affinity ten times higher than p53 alone. This repression domain effectively eliminates all p53 dependent apoptosis pathways allowing adenovirus to replicate without programmed cell death.[11]
In 1996 Frank McCormic was the first person to suggest that an adenovirus lacking its E1B-55K gene could selectively target cancer cells due to their lack of an active p53 gene but still maintain virulence and viral defenses through the E1B-19K gene. McCormic developed the Onyx-15 adenovirus while it proved effective in animal trials it never reached Phase III and was therefore never tested on human patients. However, the Sunway group in China took McCormics research and developed the H101 strain which contained a complete E1B-55KB deletion. This strain boasted a 79% response rate when coupled with chemotherapy while chemotherapy alone only resulted in a 40% response rate.[12,13,14,15]
Herpesvirus
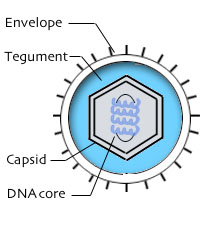
Herpesviridae is a class of enveloped viruses with a double stranded DNA genome that causes latent infections that, when triggered, can become lytic. Herpesvirus enters the cell using viral glycoproteins attached to the outside of its lipid bilayer that attach to the host cells cell membrane. When attached these glycoproteins cause the cell to internalize the herpes virion. Once the viron enters the cell it rapidly decays and its viral DNA migrates to the nucleus where transcription and replication of viral genes begins to take place.[16]
Herpesvirus was one of the first classes of viruses that was successfully turned oncogenic with the HSV1716 strain, a mutant form of the herpes simplex virus 1. HSV1716 takes advantage of innate somatic cell defense systems targeting cancer cells in a mechanism relatively similar to many other oncolytic viruses like H101. HSV1716 contains a full deletion of the gene ICP34.5 which allows for neurovirulence (viral replication within nerve cells)[17,18]. The ICP34.5 gene functions by counteracting PKR mediated blocks on viral replication and apoptosis. PKR is typically activated by double-stranded RNA, PACT or heparin. Once activated PKR deactivates cellular transcription, halting viral replication. Under extreme viral stress PKR is able to induce apoptosis through the activation of inferon cytokines [19]. PKR also interacts with p53 which is which is also typically inactivated in cancer cells[20].
A knockout of this gene results in a virus that while unable to infect somatic cells maintains virulence in most cancerous cells due to the inactivation of PKR (PKR is known to activate PP2A, a tumor suppression protein).
HSV1716 is currently undergoing a number of clinical trials to determine its usefulness in humans and recently finished phase III trials.[21] Recently some have reported that by upregulating US11, GM-CSF and downregulating ICP47 rates of tumor destruction, tumor reduction and antigen presentation can be increased as well as enhancing an anti-tumor immune response. [22]
Cancer Immunotherapy
Cancer immunotherapy is, broadly, the use of one’s own immune system to treat cancer. These therapies are wide ranging but are typically centered around T-cell Costimulation. T-cell mediated immune responses of this type are triggered by cancer antigens and enhanced by immune adjuvants. Through introduction of an attenuated or genetically modified viruses carrying either the genetic material for cancer antigens or the cancer antigens themselves, the bodies defense systems can be triggered in a fashion that targets cells that express these cancer antigens.
Poxvirus
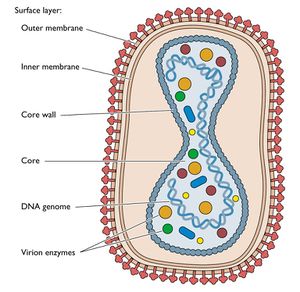
Poxviridae is a class of enveloped viruses with a double stranded DNA genome. Poxviruses enter the cell by attaching to glycoaminoglycans on the surface of the host cells lipid bilayer. Once inside the cell the viral capsid falls apart and viral genes are transcribed in the cytoplasm starting the lytic cycle. Poxviruses were the first class of viruses scientists were able to create vaccines for and are among the best understood viruses on earth. Additionally, Poxviruses, across all classes, contain a number of phenotypes that make them ideal candidates for vaccine-mediated cancer immunotherapy.[23,24]
The first and one of the most important factors in poxviruses utility as a cancer vaccine agent is its method of transcription and translation. Because Poxviruses replicate within the cytoplasm there is no risk of genetic integration thereby avoiding a constant low level immune response which could lead to a number of serious, chronic conditions and even, ironically, cancer. Additionally, cytoplasmic transcription and translation means that translation is independent of many host factors allowing for quick and efficient replication in most patients.[25]
Poxvirus is also among the most diverse viruses in the world and is found in many different species of animal. This means that there are classes of poxvirus that integrate into a human cell but are nonpathogenic. Modified vaccina Ankara (MVA) is a perfect example of this. The original vaccina was originally grown in chicken embryos, however after passing the virus through cell culture 570 times the MVA developed a number of mutations in the pathogenic regions of its genome (INF-a, IFN-b and TNF-a). This means, while it infect mammalian cell lines it cannot cause illness, even in the immune compromised. This makes MVA a perfect vector for introducing cancer antigens into the body [26]. Additionally, again due to the diversity these viruses, Poxvirus can be engineered to enter virtually all cell types within the body. Finally, because of its large size poxvirus is able to accommodate large genes that code for large proteins including antigens and other immunomodulation molecules. The translation of immune adjuvants in conjunction with the antigens causes an immune response against not only the virus but the host cell as well. Because the antigen that triggered this response was originally from a cancer cell, memory T cells are formed and the body develops an adaptive immunity to that specific type of cancer.
Currently a species of poxvirus containing the tumor-associated antigen PSA, i.e. PSA-TRICOM (PROSTVAC-V/F) is currently in phase III clinical trials in metastatic castration-resistant prostate cancer[28].
HIV
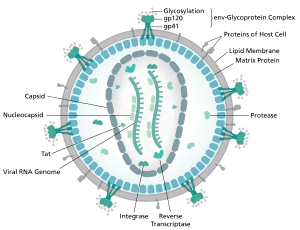
HIV or Human Immunodeficiency Virus is a retrovirus that causes progressive immunodeficiency in the form of AIDS. HIV only infects cells that play a role in the human immune response, most famously the CD4+ T cell but also macrophages and dendritic cells. HIV contains 2 RNA “chromosomes” chat code for 9 total proteins. The HIV capsid is surrounded by a lipid bilayer made up of its previous host cell’s cell membrane. This membrane makes the virus difficult to detect in the body and increases HIVs infectivity. HIV enters its host cell by fusing its cellular membrane to the cellular membrane of its host cell and injecting its viron into the cytoplasm. Once there the capsid falls apart and the viral RNA is turned into double stranded DNA using reverse transcriptase, this segment of DNA is then integrated into the host cells genome where it can sit in perpetuity. [29]
HIV immunotherapy focuses on dendritic cells. Dendritic cells are antigen presenting cells typically found in regions of the body most likely to be exposed to foreign invaders, i.e. the lungs, skin, nose and stomach. These cells are designed to recognize and present foreign antigens. When a dendritic cell recognizes a foreign antigen it moves to the lymph nodes and attaches to T and B cells where the antigen is attached to MHC proteins. The MHC protiens trigger a targeted immune response against that antigen and whatever is producing it. HIV uses these cells as a transportation system, infecting a dendritic cell then lysing it when it moves into the lymph nodes.[30,32]
Dendritic cells are of special importance to cancer immunotherapy as they seem to be able to effectively create a tumor specific immune response when compared with direct presentation of the antigen to a T or B cell. This means there is a stronger response to the cancer cells and a greater likelihood for remission.[31]
HIV is used as the vaccine vector for a number of reasons. HIV as discussed earlier, is selective and only infects certain cell types, including the dendritic cell. This means that the vaccine will be more effective per unit than a non-specific virus. Additionally, because HIV is a retrovirus it is able to be engineered as a lentiviral vector. A lentiviral vector is a modified retrovirus that has been given a new series of genes to transcribe within a cell. These vectors are very effective in gene therapy because the genes encoded within the lentiviral factor can continue to transcribed and translated long after the original dose. HIV is especially effective as its genes actually integrate into the host’s genome.
There have been a number of cases of modified HIV being used to treat cancer most of which were performed by Dr. Carl H. June. He is currently working on using the virus in treating pancreatic cancer.[33]
Problems Still Facing Viral Oncology
While viruses are certainly promising as a cancer fighting tool there are still a number of challenges still facing viral oncology. The first and most obvious of these problems is that viral oncology involves injecting a person with a live virus, which carries with it a risk of contracting disease. While almost all viruses are attenuated significantly or genetically modified to the point where they should be totally non-pathogenic to somatic cells there is still a risk, especially as many cancer patients are immunocompromised. Additionally, the patient’s body often mounts a defense against the virus itself causing the immune system to attack and destroy circulating viral particles before they can infect target cells[34,35].
While the problems intrinsic to using a virus are significant, the majority of the ptoblems facing viral oncology have to do with the way cancer exists inside of the human body. Most cancers form a large mass of cells called a tumor. This tumor is surrounded by connective tissue that differs significantly from the cell membrane. This connective tissue blocks many viruses that enter the cell by binding to specific proteins, significantly reducing the efficiency of the viral particles and resulting in an incomplete infection of the cancerous mass. This problem gets exponentially worse as the cancerous tumor becomes larger. While direct injection of viral particles into the tumor coupled with increased dose does reduce this effect larger tumors still prove difficult to fully infect.[36] Another problem with viral oncology stems from the necrotic region contained within many cancerous tumors. This necrotic region is made up of cancerous cells that have died and lysed due to increased cellular density and inefficient distribution of nutrients. As the tumor continues to expand and grow the necrotic region also expands. This expansion can kill cancer cells in the interior of the tumor before the virus is able to adequately replicate. Premature cell death coupled with many cancers accelerated division rate often means that the virus simply cannot replicate fast enough to keep up with the tumor.[37]
Lastly, due to the inherent instability of a cancer cell’s genome, cancer cells are rapidly able to develop defenses against viruses. While oncolytic viruses are designed to exploit innate defensive shortcomings within the cancer cells most of these shortcomings concern genome regulation and internal defense not external defense. In some trials doctors have observed cancer cells becoming totally resistant to a certain strain of virus by no longer producing surface binding proteins that the virus needs to enter the cell.[38]
Possible Solutions
As discussed earlier a major problem facing oncolytic viruses is the inability of the virus to move thorough the connective tissue surrounding the target tumor. Recently, doctors have begun experimenting with pre-treatments before dosing someone with a virus, such as hyaluronidase or collagenase, which would work to break up the connective tissue allowing the virus to attach to the cancer cell and infect it.[39]
Doctors are also thinking about employing a “viral cocktail” much like those used in chemotherapy or in treating retroviruses. These would contain a number of different types of viruses that function using different pathways making it difficult for the cancers to develop an immunity.
References
1. Still, GF (1897). On a form of chronic joint disease in children. Med Chir Soc 80: 52.
2. Dock, G (1904). The influence of complicating diseases upon leukemia. Am J Med Sci
127: 563–592.
3. Pelner, L, Fowler, GA and Nauts, HC (1958). Effects of concurrent infections and their
toxins on the course of leukemia. Acta Med Scand Suppl 338: 1–47.
4. Hoster, H, Zanes R and vonHaam E (1949). The association of “viral” hepatitis and
Hodgkin’s disease. Cancer Res 9: 473–480.
5. Garber, K (2006). China approves world’s first oncolytic virus therapy for cancer
treatment. J Natl Cancer Inst 98: 298–300.
6. Malkin, D (2011). "Li-fraumeni syndrome". Genes & cancer 2 (4): 475–84. doi:10.1177/1947601911413466. PMC 3135649. PMID 21779515.
7. Fillat, Cristina (2010). "Controlling Adenoviral Replication to Induce Oncolytic Efficacy" (PDF). The Open Gene Therapy Journal 3: 15–23. doi:10.2174/1875037001003010015.
8. ICTV. "Virus Taxonomy: 2014 Release". Retrieved 15 June 2015
9. Rodriguez, R; Schuur, ER; Lim, HY; Henderson, GA; Simons, JW; Henderson, DR (1997). "Prostate attenuated replication competent adenovirus (ARCA) CN706: A selective cytotoxic for prostate-specific antigen-positive prostate cancer cells". Cancer Research 57 (13): 2559–63. PMID 9205053.
10. Gilbert, Scott F. Developmental Biology, 10th ed. Sunderland, MA USA: Sinauer Associates, Inc. Publishers. p. 588.
11. H. Jiang, F. McCormick, F.F. Lang, C. Gomez-Manzano, J. Fueyo, Oncolytic adenoviruses as antiglioma agents, Expert. Rev. Anticancer Ther. 6 (2006) 697–708.
12. Barker, Douglas D.; Berk, Arnold J. (1987). "Adenovirus proteins from both E1B reading frames are required for transformation of rodent cells by viral infection and DNA transfection". Virology 156 (1): 107–121
13. Heise, Carla; Sampson-Johannes, Adam; Williams, Angelica; Mccormick, Frank; Von Hoff, Daniel D.; Kirn, David H. (June 1997). "ONYX-015, an E1B gene-attenuated adenovirus, causes tumor-specific cytolysis and antitumoral efficacy that can be augmented by standard chemotherapeutic agents". Nature Medicine 3 (6): 639–645.
14. Definition of ONYX-015 - National Cancer Institute Drug Dictionary
15. John Nemunaitis, Ian Ganly, Fadlo Khuri, James Arseneau, Joseph Kuhn, Todd McCarty, Stephen Landers, Phillip Maples, Larry Rome, Britta Randlev, Tony Reid, Sam Kaye and David Kirn (2000). "Selective Replication and Oncolysis in p53 Mutant Tumors with ONYX-015, an E1B-55kD Gene-deleted Adenovirus, in Patients with Advanced Head and Neck Cancer: A Phase II Trial". Cancer Res 60 (22): 6359–66. PMID 11103798
16. Trus BL, Cheng N, Newcomb WW, Homa FL, Brown JC, Steven AC (November 2004). "Structure and Polymorphism of the UL6 Portal Protein of Herpes Simplex Virus Type 1". Journal of Virology 78 (22): 12668–71. doi:10.1128/JVI.78.22.12668-12671.2004. PMC 525097. PMID 15507654.
17. Varghese, Susan; Rabkin, Samuel D (2002). "Oncolytic herpes simplex virus vectors for cancer virotherapy". Cancer Gene Therapy 9 (12): 967–78. doi:10.1038/sj.cgt.7700537. PMID 12522436
18. Liu, B L; Robinson, M; Han, Z-Q; Branston, R H; English, C; Reay, P; McGrath, Y; Thomas, S K; et al. (2003). "ICP34.5 deleted herpes simplex virus with enhanced oncolytic, immune stimulating, and anti-tumour properties". Gene Therapy 10 (4): 292–303. doi:10.1038/sj.gt.3301885. PMID 12595888.
19. García MA, Gil J, Ventoso I, Guerra S, Domingo E, Rivas C, Esteban M (December 2006). "Impact of protein kinase PKR in cell biology: from antiviral to antiproliferative action". Microbiol. Mol. Biol. Rev. 70 (4): 1032–60. doi:10.1128/MMBR.00027-06. PMC 1698511. PMID 17158706.
20. Cuddihy AR, Wong AH, Tam NW, Li S, Koromilas AE (April 1999). "The double-stranded RNA activated protein kinase PKR physically associates with the tumor suppressor p53 protein and phosphorylates human p53 on serine 392 in vitro". Oncogene 18 (17): 2690–702.
21. "Amgen Presents Interim Overall Survival Data From Phase 3 Study Of Talimogene Laherparepvec In Patients With Metastatic Melanoma" (Press release). November 18, 2013. Retrieved 30 October 2015.
22. Varghese S, Rabkin SD (1 December 2002). "Oncolytic herpes simplex virus vectors for cancer virotherapy". Cancer Gene Therapy 9 (12): 967–978. doi:10.1038/sj.cgt.7700537. PMID 12522436.
23. Z.S. Guo, A. Naik, M.E. O’Malley, P. Popovic, R. Demarco, Y. Hu, X. Yin, S. Yang, H.J. Zeh, B. Moss, M.T. Lotze, D.L. Bartlett, The enhanced tumor selectivity of an oncolytic vaccinia lacking the host range and antiapoptosis genes SPI-1 and SPI-2, Cancer Res. 65 (2005) 9991–9998.
24. J.A. McCart, J.M. Ward, J. Lee, Y. Hu, H.R. Alexander, S.K. Libutti, B. Moss, D.L. Bartlett, Systemic cancer therapy with a tumor-selective vaccinia virus mutant lacking thymidine kinase and vaccinia growth factor genes, Cancer Res. 61 (2001) 8751–8757.
25. ICTV. "Virus Taxonomy: 2014 Release". Retrieved 15 June 2015.
26. C.A. Petrulio, H.L. Kaufman, Development of the PANVAC-VF vaccine for pancreatic cancer, Expert. Rev.Vaccines 5 (2006) 9–19.
27. Unanue ER (Jul 2013). "Perspectives on anti-CD47 antibody treatment for experimental cancer". Proceedings of the National Academy of Sciences of the United States of America 110 (27): 10886–7. doi:10.1073/pnas.1308463110. PMC 3704033. PMID 23784781.
28. Gully JL, et al. Immune impact induced by PROSTVAC (PSA-TRICOM), a therapeutic vaccine for prostate cancer. http://www.ncbi.nlm.nih.gov/pubmed/24778277, Pubmed 2013
29. Weiss RA (May 1993). "How does HIV cause AIDS?". Science 260 (5112): 1273–9. Bibcode:1993Sci...260.1273W. doi:10.1126/science.8493571. PMID 8493571.
30. Fong, Laurence, and Engleman Edgar. "Dendritic Cells in Cancer Immunotherapy." - Annual Review of Immunology, 18(1):245. Annual Reviews, 2000. Web. 14 Dec. 2015.
31. Steven A. Rosenberg, Nicholas P. Restifo, and James C. Yang. "Adoptive Cell Transfer: A Clinical Path to Effective Cancer Immunotherapy." Nature Reviews. Nature, 2008. Web.
32. "DENDRITIC CELL VACCINES FOR CANCER IMMUNOTHERAPY." - Annual Review of Medicine, 50(1):507. N.p., 1999. Web.
33. http://www.cancerresearch.org/news-publications/video-gallery/webinar-carl-june-engineering-t-cells-to-conquer-cancer
34. S. Krishnamurthy, T. Takimoto, R.A. Scroggs, A. Portner,Differentially regulated interferon response determines the outcome of Newcastle disease virus infection in normal and tumor cell lines, J. Virol. 80 (2006) 5145–5155.
35. Z.Y. Li, S. Ni, X. Yang, N. Kiviat, A. Lieber, Xenograft models for liver metastasis: relationship between tumor morphology and adenovirus vector transduction, Mol. Ther. 9 (2004) 650–657.
36. D.M. Shayakhmetov, Z.Y. Li, S. Ni, A. Lieber, Targeting of adenovirus vectors to tumor cells does not enable efficient transduction of breast cancer metastases, Cancer Res. 62 (2002) 1063–1068.
37. R. Cairns, I. Papandreou, N. Denko, Overcoming physiologic barriers to cancer treatment by molecularly targeting the tumor microenvironment, Mol. Cancer Res. 4 (2006) 61–70.
38. T. Alain, M. Kim, R.N. Johnston, S. Urbanski, A.E. Kossakowska, P.A. Forsyth, P.W. Lee, The oncolytic effect in vivo of reovirus on tumour cells that have survived reovirus cell killing in vitro, Br. J. Cancer 95 (2006) 1020–1027.
39. R. Bilbao, M. Bustos, P. Alzuguren, M.J. Pajares, M. Drozdzik, C. Qian, J. Prieto, A blood-tumor barrier limits gene transfer to experimental liver cancer: the effect of vasoactive compounds, Gene Ther. 7 (2000) 1824–1832.
Authored for BIOL 291.00 Health Service and Biomedical Analysis, taught by Joan Slonczewski, wirtten by Erick Ditmars, 2016, Kenyon College.
