A Beautiful Colonizer: Vibrio fischeri and its Host Euprymna scolopes
Vibrio fischeri is a Gram-negative luminous bacterium belonging to the Vibrionaceae family, which includes many bacterial species that are marine γ-proteobacteria and engage in a diversity of pathogenic or beneficial interactions with animal tissue. V. fischeri is best known for its symbiotic relationships with squids and fishes and is found throughout the world in temperate and subtropical regions. The bacterium exists as members of the microbial gut community in many marine animals or as free-living saprophytes. The symbiotic relationship between V. fischeri and the squid Euprymna scolopes has been extensively studied primarily because of the ability of V. fischeri to produce luminescence by expressing the lux operon, a cluster of genes found in several of the Vibrionaceae. Luminescence allows the squid to camouflage itself and evade bottom-dwelling predators of the ocean because the light produced is emitted downward. The squid can manipulate the light that V. fischeri produces to match the intensity of the light that the moon and starlight reflects downward onto its body. V. fischeri is housed within a specialized light organ within the squid's mantle cavity. The light organ consists of host tissues that include crypt spaces that contain the bacterial symbionts. 7
In the squid-vibrio symbiosis, the squid can select from different V. fischeri strains to find the most suitable symbionts. Establishing symbiotic relationships in aquatic environments can be a challenge because there are low to undetectable concentrations of the specific symbiont cells that colonize the tissues of animal hosts. The colonization of the E. scolopes light organ by V. fischeri cells usually occurs within twelve hours after the juvenile squid hatches and is the topic of current research. 2
Recruitment and Colonization
First Encounter
V. fischeri cells target the tissue on the ciliated organ for inoculation. The squid E. scolopes sheds mucus from a superficial ciliated epithelium shortly after hatching. V. fischeri cells are harvested from the environment along the tissue that makes up this ciliated organ.1 The organ is initially uncolonized and has an array of complex ciliated epithelial cells on each lateral surface. Successful colonization of the squid by V. fischeri will induce the loss of the ciliated epithelium. 2
Successful Association
The ciliated field of the surface epithelium surrounds a set of three pores and as shown in Figure 2, during initiation of the symbiosis, V. fischeri cells enter these pores, travel down ducts, and colonize the internal spaces of the organ.2 Specifically, V. fischeri cells associate with the host in two steps. Attachment to the cilia occurs first followed by aggregation of the harvested population. In the absence of V. fischeri cells, other Gram-negative but not Gram-positive bacteria will aggregate. When V. fischeri cells are present, they exhibit a competitive dominance over three or more hours, and are the only cells present in the aggregates. Elimination of other bacteria species requires a complex host-symbiont dialogue across the ciliated surface epithelium.1 Recent transcriptomic studies in the lab using green fluorescent protein-labeled V. fischeri cells have found that within a few hours after introducing V. fischeri cells into seawater containing E. scolopes, currents aggregated the bacteria and these aggregates were embedded in a mucus-like material above the light organ pores. Ciliary-mucus currents allow invertebrate tissues to interact with food particles in the environment. 2 The harvested V. fischeri cells travel toward the crypts after entering the pores and no matter how many cells migrate through the pores, only one or two make it to the crypts, similar to the bottleneck effect. Then, the bacterial cells begin to divide, eventually filling the crypt spaces. Thousands of species of bacteria interact with the surface of E. scolopes organ, but only one or two strains of V. fischeri cells inhabit the deep crypts. 1
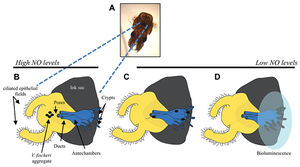
The Effect of Time on Normal Colonization
Furthermore, V. fischeri cells require a minimum amount of time to reside within the squid secreted mucus before colonization of the bacteria in its host was initiated. Normal colonization results when the bacterial cells accumulated in the mucus for the first few hours and after four to six hours after initial inoculation, V. fischeri cells migrated away from the mucus and into the pores, eventually proliferating into the crypts.2
Dominance of Symbiont Cells
Specificity of Colonization
V. fischeri is competitively dominant over other species of bacteria concerning space along the epithelial ciliated surface of E. scolopes. In one study, V. fischeri cells were added to unfiltered environmental seawater at concentrations ranging from 103 cells to 106 cells per milliliter. Hatchling squid were exposed for four hours to this water, and V. fischeri cells formed tightly packed aggregations, regardless of the proportions of non symbiotic bacteria. V. fischeri cells accounted for more than ninety-nine percent of the cells in the aggregations. The ability of one bacterial species to outcompete another for a space to adhere onto mucus is not uncommon among species of bacteria. Once inside the light organ, strains of V. fischeri compete to inhabit the deep crypts and strains with higher fitness dominate the light organ.3
Bacterial viability
Viability of V. fischeri is important for the formation of tight aggregations and for competition with non symbiotic bacterial strains for space within the light organ. Tight aggregations form above the pores on each side of the light organ when viable V. fischeri cells are added to seawater containing hatchling squid. On the other hand, V. fischeri cells that are heat inactivated or treated with azide stain vividly with propidium iodide and form significantly looser aggregations. Propidium iodide is used to determine which V. fischeri cells are viable because a low staining of propidium iodide by the symbiont cells indicates viability. The use of propidium iodide and the vivid staining of V. fischeri cells served as an indicator that the heat inactivated or azide treated V. fischeri cells were not viable. When non symbiotic bacterial species are incubated with heat inactivated or azide treated unviable V. fischeri cells, the non symbiotic bacteria aggregate as they normally would without the presence of viable V. fischeri cells. The non symbiotic bacterial species are the dominant bacterial cells present in those aggregations. Therefore, V. fischeri cells need to be viable in order to form tight aggregations in competition with other non symbiotic bacterial strains for space.3
Motility
Motility is the best understood characteristic of V. fischeri that is important for symbiosis. Symbiont cells that are incapable of motility cannot colonize the organ by navigating through the duct.4 When nonmotile mutants were tested for the ability to colonize the squid light organ, none were capable of initiating a symbiotic relationship. Whether or not a flagellum is also required for adherence to host tissue remains to be studied. During initiation of the colonization process, bacteria are not only swept along by the host's epithelial ciliary movement, but also propel themselves into the crypt spaces of the light organ.5
Lux Genes
In a study carried out in 2000, researchers found that mutant V. fischeri strains that are defective in light production cannot persist in the organ and are eliminated from the organ if they are not producing luminescence.4 Specifically, luxA, luxI, and luxR mutants of V. fischeri expressed less than 0.1% of the luminescence of their lux+ parent strain. LuxA encodes an enzyme that catalyzes the light-producing reaction and the luxI and luxR genes are associated with quorum sensing and induction of the lux operon. Luminescence mutants of V. fischeri also fail to elicit cytoplasmic swelling in the epithelial cells that line the light organ. Wild-type strains of V. fischeri were also found to outcompete strains that are defective in light production.6
The Role of Quorum Sensing
Autoinducers Control Bioluminescence
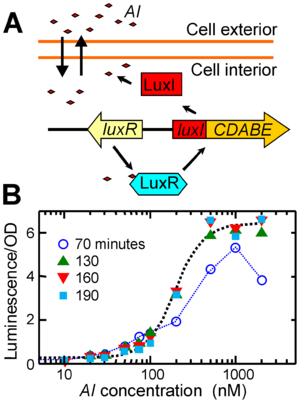
Quorum sensing was first discovered in V. fischeri cells as the mechanism that induces luminescence. At low cell densities of V. fischeri cells, the lux genes that synthesize luciferase are switched off and the bacterial cells do not produce luminescence. When V. fischeri cells colonize E. scolopes, the cell density increases and the lux genes are strongly induced. V. fischeri may attain 109-1010 cells/cm.3. The quorum sensing pathway usually includes three autoinducer synthases and three corresponding autoinducer pathways. When the concentration of the autoinducer 3OC6HSL reaches a high level, it forms a complex with LuxR that activates transcription of the lux operon, as shown in Figure 3 part (A). This leads to the synthesis of luciferase, a key enzyme that produces bioluminescence.
Furthermore, an additional study as shown in Figure 3 part (B) found that different cells emit light with different intensities even when the population of cells is saturated with a high concentration of autoinducer. Individual cells emitted very little light during a ten minute exposure of autoinducer at 1000 nM. The shape of the growth curve for luminescence versus autoinducer was established two to three hours after the autoinducer was initially introduced. In the absence of autoinducer, light emission from the cells is barely detectable. However, studies have found that there is significant cell-to-cell variability in the overall level of emission and in the onset time for a measurable response in bioluminescence. These studies are in response to a defined chemical signal, the autoinducer 3OC6HSL and short term fluctuations in brightness were present. In contrast, the addition of autoinducer increases the average brightness and the differences in the brightness of individual cells. Overall, the luminescence response of V. fischeri cells is regulated by quorum sensing, but fluctuations in this response is common. 9
.
Presence of Two Quorum-Sensing Systems
V. fischeri cells possess two quorum-sensing systems: the ain quorum sensing system and the lux quorum sensing system. The ain quorum sensing is required for normal initiation of colonization while the lux system is not fully active during the initial stages of colonization of E. scolopes. 10 The effect of the ain system on luminescence is mostly apparent at moderate bacterial concentrations occurring in culture while the lux system is responsible for inducing luminescence at high cell densities. The ain quorum sensing is required for appropriate expression of the lux quorum sensing system and inactivation of the ain quorum sensing system produces an initiation defect from the loss of regulation of early colonization factors and an absence of a functional lux quorum sensing system. Once V. fischeri has colonized the crypt regions of the squid with a high concentration of cells, the ain quorum sensing system is initially induced. The lux quorum sensing system will then induce expression of genes encoding luminescence. 11
Effect on Motility
Proper motility of V. fischeri has been shown to be crucial for proper initiation in the colonization of the squid. Nonmotile mutants are impaired significantly in establishing the colonization of the light organ. At later stages of colonization, V. fischeri cells lose their flagella, and this implies that flagellation is not required for the proper maintenance of symbiosis. In one study, hyper motile ainS and motility deficient mutants displayed a delay in initiation of colonization. The altered motility behavior of these mutants is enough to explain their inability to colonize the squid properly but further research should focus on the possibility that AinS regulates other initiation factors. 11
Further Thoughts
The squid-vibrio symbiotic relationship uses ciliary mucus currents to harvest symbionts, but further research should focus on the mechanisms underlying this process. Uncertainty remains on how bacteria induce the formation of this ciliary-mucus structure and how the squid responds to its symbionts on the molecular level using signal transduction pathways. It is also unclear whether or not V. fischeri cells are enriched within the aggregates or how symbionts direct their migration from aggregations to their target tissue.2 V. fischeri cells may also alter the chemistry of the mucus matrix and create a biofilm that favors their persistence in their host. Other studies have shown that bacteria often participate in the formation and maturation of a biofilm that promotes microbial growth and persistence.3
Conclusion
The relationship between the Hawaiian bobtail squid Euprymna scolopes and the Gram-negative bacterium Vibrio fischeri has been studied for more than fifteen years as a model for the establishment of a horizontally transmitted symbiosis. One of the main challenges of a horizontally transmitted symbiosis is the acquisition of a microbial symbiont by the host from the environment. Tissues that the host maintains as a juvenile animal allow it to harvest and maintain specific symbionts. Colonization of the host occurs in a series of steps but the symbiont is an active partner in this process. The ability of V. fischeri to colonize the squid's tissue is in large part due to its viability and motility. Viable and motile V. fischeri are the only bacteria that can navigate through the pores and into the ducts of E. scolopes. Viable bacteria also form dense aggregations while motile bacteria can outcompete other bacteria for space. Bioluminescent V. fischeri can colonize the host's crypt epithelium for a long period of time. The presence of two quorum sensing systems in V. fischeri regulates the normal initiation steps of colonization and bioluminescence.
References
Edited by Giselle Garcia student of Suzanne Kern in BIOL168L (Microbiology) in The Keck Science Department of the Claremont Colleges Spring 2015.