Black Sea
Introduction

The Black Sea is an inland body of water with a surface area of roughly 422,000 km2 and a maximum depth of 2,210 metres [1]. Located in southeastern Europe, it is bound by the borders of Russia, Ukraine, Georgia, Turkey, Bulgaria and Romania [1]. It is connected to the Atlantic Ocean through the Marmara, Aegean and Mediterranean Seas via the Bosphorus and Dardanelles Straits. The Black Sea is classified as a meromictic body of water. The McGraw Hill Science Encyclopedia [2] defines a meromictic lake as having two distinct layers, the top mixolimnion and the lower monimolimnion, that do not mix. The top layer of the Black Sea is approximately 50 to 100 metres deep and is exposed to oxygen in the atmosphere; the bottom layer is 2000 metres deep and has no dissolved oxygen [17]. What makes the Black Sea unique is that it is not a lake, but a large basin in which two distinct layers exist, forming the largest anoxic body of water on Earth [3]. The sea formed thousands of years ago after glaciers from the ice age retreated and left a large freshwater lake in which saltwater from the Mediterranean Sea flowed in. The saltwater sank to the bottom of the lake and led to stratification based on density differences [3]. This separation between the oxic and anoxic zones allows for various microorganisms to inhabit the different environments [3]. Aerobic organisms can be found in upper layer, whereas anaerobic organisms are found at greater depths below the surface. The interface between the oxic and anoxic zones, referred to as the suboxic, holds a high density of microbes, with aerobic and anaerobic bacteria living within metres of each other [4]. The Black Sea serves as a model ecosystem for studying microbial life, as it functions as a redox gradient in which microorganisms respire through various redox reactions [5].
Chemical and Physical Characteristics
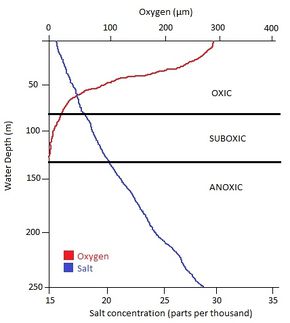
The physical environment of the Black Sea contributes to the non-mixing layers of water. The temperature at the surface varies during the seasons, falling as low as -0.5⁰C during the winter and rising as high as 26⁰C during the summer [1]. At depths of 50 -100 metres the temperature ranges from 6 - 8⁰C and at depths over 400 metres the temperature does not fluctuate during the seasons, remaining around 8⁰C [1]. The rapidly changing temperatures at the surface of the sea helps prevent the mixolimnion from mixing with the monimolimnion and being exposed to the atmosphere [6].
Salinity plays a role in the density gradient that results from the dissolved salt. The surface waters hold approximately 17 parts per thousand of salt. At depths below 400 metres, the salt concentration reaches over 30 parts per thousand [1]. This further separates the lower mixolimnion from the upper monimolimnion waters with similar temperature ranges [6].
The dissolved oxygen content is what differentiates the mixolimnion from the monimolimnion layer. The upper mixolimnion holds over 200 µM of dissolved oxygen, while the interface suboxic zone has 75 µM of dissolved oxygen as a result of aerobic respiration being performed by microorganisms above [7]. Below 130 metres, no oxygen is present [7]. These oxic and anoxic zones allow for a redox gradient to form. A redox gradient shows depletion of terminal electron acceptors in order of decreasing redox potential for the respiratory processes occurring. Oxygen, the best electron acceptor, is preferentially used first and is depleted by aerobic organisms found in the mixolimnion [8]. This affects the microbial activity and rates of organic matter decomposition at greater depths [8]. Redox processes are normally found in sediments of centimetre intervals, but the Black Sea’s chemical processes occur over hundreds of metres [3].
Key Microorganisms
Oxic Zone
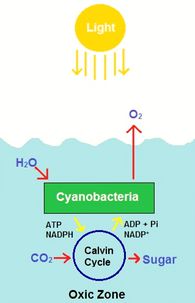
Schubotz et al. used membrane lipid analysis to determine the presence of various aerobic bacteria in the Black Sea [5]. The oxic zone includes various cyanobacteria, methanotrophs, and nitrifying bacteria. Some specific classes include Actinobacteria, Gammaproteobacteria and Alphaproteobacteria [5][9][10].
Suboxic Zone
This interface holds both aerobic and anaerobic microbes. These include cyanobacteria, denitrifying bacteria, methanotrophs, methanogens, iron-reducing bacteria, manganese-reducing bacteria, anammox bacteria, and purple sulfur bacteria [5][7][9][10]. Specific bacteria include Planctomycetes [9], Deltaproteobacteria [11] and the genera of Arcobacter and Pelobacter [7].
Anoxic Zone
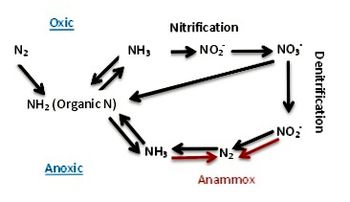
All these organisms respire by anaerobic processes and cannot tolerate oxygen. Microbes include sulfate-reducing bacteria (upper part of anoxic zone), purple and green sulfur bacteria, fermenting bacteria, methanotrophs, methanogens, manganese-reducing bacteria, anammox bacteria and various Archaea [5][7][9][10][11].
Microbial Processes
Photosynthesis
Cyanobacteria in the oxic zone derive their energy through the process of photosynthesis [12]. Photosynthesis involves energy from photons used to fix carbon dioxide into sugars and oxygen [12]. The oxygen and sugars produced can be used by aerobic bacteria to respire (use oxygen as a terminal electron acceptor), and in return, these aerobic bacteria provide cyanobacteria with carbon dioxide from the oxidation of sugars.
Denitrification
Denitrification is performed by nitrate and nitrite reducing bacteria found in the suboxic zone [13]. Denitrification uses nitrate (NO3-) or nitrite (NO2-) as terminal electron acceptors, and converts them into the gaseous products nitric oxide (NO), nitrous oxide (N2O) or dinitrogen (N2), oxidizing organic matter in the process [13]. Nitrate is used after oxygen is depleted, indicating it is the best electron acceptor remaining. This explains why the majority of denitrifying bacteria are located just below the oxic zone [13]. Denitrification can have greater effects on the environment, including production of greenhouse gases (N2O), and the degradation of dead organic matter in the sea.
Anammox
The anammox process primarily occurs in the lower suboxic zone and the anoxic zone of the Black Sea [3][9]. The process involves the reduction of nitrite and oxidation of ammonium into dinitrogen: NH4+ + NO2− → N2 + 2H2O. It is performed by the Planctomycetes [14] and is used to release nitrogen into the atmosphere.
Methanogenesis
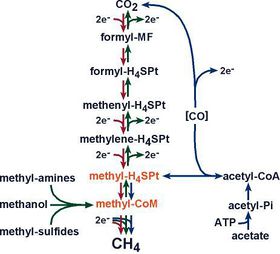
A high concentration of carbon dioxide is found in the deepest parts of The Black Sea due to gas seeps in the seafloor and is used by methanogens that respire anaerobically and produce methane. Carbon dioxide has the lowest redox potential of the electron acceptors (NO3-, O2, Fe3+, SO42-) in the Black Sea and thus is limited to microbes at the bottom of the sea [15].
Methanotropy
The Black Sea has over 3000 gas seeps, releasing methane (CH4) from deep-sea mud volcanoes [16]. Methanotrophs can oxidize methane aerobically and anaerobically [11], yet the majority are found in the anoxic zone where the concentration of methane is the highest [15]. Aerobic methanotrophs oxidize methane using oxygen as the electron acceptor producing formaldehyde [10]. Anaerobic methanotrophs are sulfate-reducing bacteria [17]. Oxygen and nitrate are depleted at the anoxic zone, thus sulfate is the next best terminal electron acceptor that microbes use to respire and oxidize methane into other organic compounds [11].
References
[1] "Black Sea". Encyclopedia Britannica. Encyclopedia Britannica Inc., 2012. Web. <http://www.britannica.com/EBchecked/topic/68234/Black-Sea/33213/Hydrology>.
[2] "Meromictic lake." McGraw-Hill Encyclopedia of Science and Technology. The McGraw-Hill Companies, Inc., 2005.
[3] Staley, T. “Probing nitrogen metabolism in the redox gradient of the Black Sea.” PNAS, 2007, DOI:10.1073/PNAS.0702088104
[4] Neretin L.N, Abed, R.M., Schippers, A., Schubert, J., Kohls, K. and Kuypers, M.M. “Inorganic carbon fixation by sulfate-reducing bacteria in the Black Sea water column.” Environmental Microbiology, 2007, DOI:10.1111/j.1462-2920.2007.01413.x
[5] Schubotz F., Wakeham, S.G., Lipp, J.S., Fredricks, H.F. and Hinrichs, K. “Detection of microbial biomass by intact polar membrane lipid analysis in the water column and surface sediments of the Black Sea.” Environmental Microbiology, 2009, DOI:10.1111/j.1462-2920.2009.01999.x
[6] Stewart K.M., Walker, K.F. and Likens, G.E. “Meromictic Lakes.” In: Encyclopedia of Inland Waters. volume 2, pp. 589-602 Oxford: Elsevier.
[7] Thamdrup B., Rossello-Mora, R. and Amann, R. “Microbial Manganese and Sulfate Reduction in Black Sea Shelf Sediments.” Applied and Environmental Microbiology, 2000, DOI:10.1128/AEM.66.7.2888-2897.2000
[8] Yakushev E.V. “Chemical Structure of Pelagic Redox Interfaces: Observation and Modeling.” Handbook of Environmental Chemistry, 2012, DOI:10.1007/698_2012_167
[9] Kirkpatrick J., Oakley, B., Fuchsman, C., Srinivasan, S., Staley, J. and Murray, J.W. “Diversity and Distribution of Planctomycetes and Related Bacteria in the Suboxic Zone of the Black Sea.” Applied and Environmental Microbiology, 2006, DOI:10.1128/AEM.72.4.3079–3083.2006
[10] Schubert C.J, Coolen, M., Neretin, L.V., Schippers, A., Abbas, B., Durisch-Kaiser, E., Wehrli, B., Hopmans, E., Damsté, J.S., Wakeham, S. and Kuypers, M.M. “Aerobic and anaerobic methanotrophs in the Black Sea water column.” Environmental Microbiology, 2006, DOI:10.1111/J.1462-2920.2006.01079.X
[11] Leloup J., Loy, A., Knab, N.J., Borowski, C., Wagner, M. and Jørgensen, B.B. “Diversity and abundance of sulfate-reducing microorganisms in the sulfate and methane zones of a marine sediment, Black Sea.” Environmental Microbiology, 2007, DOI:10.1111/J.1462-2920.2006.01122.X
[12] Bryant D.A., Frigaard, N.U . "Prokaryotic photosynthesis and phototrophy illuminated". Trends Microbiology, 2006, DOI:10.1016/J.TIM.2006.09.001
[13] Jensen M.M., Kuypers, M.M., Lavik, G. and Thamdrup, B. “Rates and regulation of anaerobic ammonium oxidation and denitrification in the Black Sea.” ASLO, 2008, DOI: 10.4319/lo.2008.53.1.0023
[14] Kuypers M.M., Sliekers, A.O., Lavik, G., Schmid, M., Jørgensen, B.B., Kuenen, J.G., Damste, J.S., Strouss, M. and Jetten, M.S. “Anaerobic ammonium oxidation by anammox bacteria in the Black Sea.” Nature, 2003, DOI:10.1038/nature01472
[15] Treude T., Orphan, V., Knittel, K., Gieseke, A., House, C.H. and Boetius, A. “Consumption of Methane and CO2 by Methanotrophic Microbial Mats from Gas Seeps of the Anoxic Black Sea.” Applied and Environmental Microbiology, 2007, DOI: 10.1128/AEM.02685-06
[16] Egorov V.N., Artemov, Y.G., Gulin, S.B. and Polikarpov, G.G. “Methane seeps in the Black Sea: discovery,quantification and environmental assessment.” J.Black Sea/ Mediterranean Environment, 2011
[17] Ince B.K., Usenti, I., Eyigor, A., Oz, N.A., Kolukirik, M. and Ince, O. “Analysis of Methanogenic Archaeal and Sulphate Reducing Bacterial Populations in Deep Sediments of the Black Sea.” Geomicrobiology Journal, 2006, DOI:10.1080/0149045060076072
[18] Algeo, T.J. and Tribovillard, N. “Environmental analysis of paleoceanographic systems based on molybdenum-uranium covariation.” Chemical Geology, 2009, DOI:10.1016/j.chemgeo.2009.09.001