Blastodinium
Classification
Higher Order Taxa
Domain: Eukaryota; Phylum: Dinoflagellata; Class: Blastodiniphyceae; Order: Blastodiniales; Family: Blastodinidae; Genus: Blastodinium
Genus
Blastodinium
Description and Significance
This genus contains cryptic species, which are species that are almost identical in physical features, with differing genetic material. All species in this genus have very similar morphological features, only differing slightly in their genomes. These species are obligate parasites, in which they must live inside a host in order to complete their life cycles. This genus will inhabit the gut of marine copepods, a group of small crustaceans typically 1-2mm in length. These parasites can grow to be a few hundred μm in length while inside the host.
These parasites are found in habits occupied by their copepods hosts-across the globe. However, these parasites are found in greater abundances in low nutrient, oligotrophic marine waters closer to the equator. The parasites found in this habitat have a greater chance of possessing chloroplasts, however, those found in colder waters near the poles of the planet are often found having no chloroplasts.
Life Cycle
The complete life cycle of species in the genus Blastodinium is unknown due to lack of experimental research. The life cycle stages during and after parasitism are known, however. This stage of the life cycle consists of the dinospore of the organism being ingested by the host and it becoming lodged in the copepod's gut. This dinospore grows into a trophocyte while inside the host. A trophocyte is just a name to describe a cell which supplies the organism with nutrients. The trophocyte then divides into a secondary trophocyte and a gonocyte. The seconary trophocyte can divide into succeeding trophocytes and gonocytes, while the gonocyte will form sporocytes. Through dispersal mechanisms, the sporocytes release sex cells from the host, allowing more copepods to become hosts after the ingestion of spores(1). Figure 1 below shows a fully grown parasite with each of these different stages labelled.
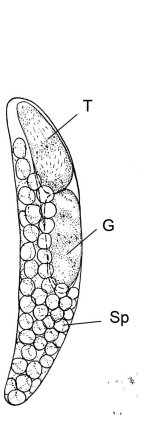
Cell Structure and Metabolism
As mentioned in the Life Cycle section, the entire life cycle of these parasites has not been studied, making it difficult to understand the cellular structure of each stage. However, studies on the trophocyte stage have been completed. These studies show that this stage possesses multiple sporocytes, each containing chloroplasts. The trophocyte also has a golgi region, which is responsible for the majority of the parasite's internal metabolic activity. Cross-sections of the tropocyte have also shown the possession of a nucleus of the trophocyte, as well as the individual sporocytes. The nuclei are different between these two stages of the parasites life, however. The nucleus of the sporocytic cells is dinokaryotic-possessing no histones and having permanently condensed chromosomes. Both the sporocytes and trophocytes are each surrounded by cuticle layer, which is a waxy-substance providing protection for the parasite.
These organisms are considered photoheterotrophic. This classification is debated, however, due to the difference in metabolisms between those parasites having multiple functioning chloroplasts and those which do not. Those with greater numbers of chloroplasts would have a reduced reliance for receiving energy from their host, so they would be considered photoautotrophic. This mixotrophic metabolism, in which organisms use carbon dioxide (autotrophy) or organic carbon (heterotrophy) for energy, is what fuels this genus' complexity(5). Based on this mixotrophic theory, the parasites that cannot satisfy their energy needs solely from photosynthesis would obtain some energy from their hosts, thus being considered photoheterotrophic.
Ecology and Pathogenesis
The parasitic species in the genus Blastodinium have the ability to reverse the sex of male copepods and cause female copepods to become sterile. It is hypothesized that the size of the parasite inside the host can cause the loss of physiological reproductive function in females due to the pressure on its organs. The sex reversal mechanism that occurs in male copepods remains inconclusive due to lack of research on the subject. These effects on the male and female copepods would have a direct impact on copepod populations. The copepod population would be reduced due to the inability to mate between copepods. This would impact the higher trophic levels. This is just one ecological impact these parasites have. This phenomenon is very similar to what occurs between the genus Wolbachia and their anthropod hosts. The sex reversal mechanism occurs in these populations as well, causing the sex ratios to change as more and more males to undergo feminization(4).
Another impact is the effect on the biogeochemical cycle. A study completed by the Norwegian Institute of Marine Research measured the fecal pellet reproduction rate of infected copepods and uninfected copepods(3). The results showed the rates of uninfected copepods were much higher when producing fecal pellets. This has a negative effect on the biogeochemical cycle because it causes reduced organic material to sink to the bottom of the ocean, where many organisms feed on this material on the ocean floor(3).
These two previous ecological impacts are due to the pathology of these parasites. However, the fact that Blastodinium do not directly harm their hosts causes some to speculate these species to act as symbionts under certain circumstances. The organisms that exhibit parasitism would often be the ones which cannot perform photosynthesis (the organisms in colder waters). This is due to the organism having to rely on consuming much of its host's energy via food. The photosynthetic Blastodinium are hypothesized to provide their host with certain nutrients, thus being considered symbionts. However, many of these theories have never been tested experimentally and only have been developed from natural observations.
References
1.Chatton É. (1920). Les Péridiniens parasites: morphologie, reproduction, ethologie. Arch. Zool. Exp. Gen. 59; 3, 116-117, 121
2. Coats D. W., Bachvaroff T., Handy S. M., Kim S. Y., Gárate-Lizárraga I., Delwiche C. F. (2008). Prevalence and phylogeny of parasitic dinoflagellates (genus Blastodinium) infecting copepods in the Gulf of California. CICIMAR Oceánides 23, 67–77
3. D. M. Fields, J. A. Runge, C. Thompson, S. D. Shema, R. M. Bjelland, C. M. F. Durif, A. B. Skiftesvik, H. I. Browman; Infection of the planktonic copepod Calanus finmarchicus by the parasitic dinoflagellate, Blastodinium spp: effects on grazing, respiration, fecundity and fecal pellet production, Journal of Plankton Research, Volume 37, Issue 1, 1 January 2015, Pages 211–220
4. Hurst G., Jiggins F. M., Graf von Der Schulenburg J. H., Bertrand D., et al. (1999). "Male killing Wolbachia in two species of insects". Proceedings of the Royal Society B. 266 (1420): 735–740. doi:10.1098/rspb.1999.069
5.Jones RI (1994) Mixotrophy in planktonic protists as a spectrum of nutritional strategies. Mar Microb Food Webs 8(1):87–96
6. Skovgaard A., Salomonsen X. M. (2009). Blastodinium galatheanum sp. nov. (Dinophyceae) a parasite of the planktonic copepod Acartia negligens (Crustacea, Calanoida) in the central Atlantic Ocean. Eur. J Phyco
Author
Page authored by Collin Hoskins and April Hendon, students of Prof. Jay Lennon at Indiana University.