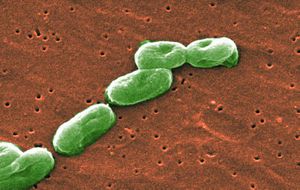
Burkholderia cepacia complex

Etiology/Bacteriology
Taxonomy
| Domain = Bacteria
| Phylum = Proteobacteria
| Class = Betaproteobacteria
| Order = Burkholderiales
| Family = Burkholderiaceae
| Genus = Burkholderia
| species = B. cepacia, B. multivorans, B. cenocepacia, B. vietnamiensis, B. stabilis, B. ambifaria, B. dolosa, B. anthina, B. pyrrocinia
Description
Burkholderia cepacia complex is a group of Gram-negative, non-spore forming bacilli composed of approximately 17 closely-related species which are grouped into nine genomovars. This is often referred to as the Burkholderia cepacia complex (BCC) [1][2]. It was originally recognized by W. H. Burkholder in the 1950s for the distinct stench it caused in onion bulbs, known as "sour skin" disease for its vinegar-like odor [1][2]. B. cepacia has emerged as a human respiratory opportunistic pathogen in individuals with weakened immune systems or chronic lung disease, espeically cystic fibrosis (CS) patients, within the past 30 years and has therefore recieved increased attention from the scientific community. BCC potentially causes abscesses and bacteremia, but this condition is highly uncommon in patients without CS [3]. Pulmonary colonization of B. cepacia can cause accelerated decline in lung funtions and cause “cepacia syndrome,” which is a progressive pneumonic illness that is fatal and essentially untreatable [4].
The organisms possess a remarkably large genetic make up with a total DNA content of more than 7 megabases and up to 3 megabase-sized replicons [1]. BCC was formerly classified as a Pseudomonas, but was transferred to the <Burkholderia genus in 1992 on the basis of phenotypic characteristics, 16S rRNA sequences, DNA-DNA homology values, and cellular lipid and fatty acid composition [2]. It poses little medical risk to healthy individuals, but weakened immune systems or chronic lung diseases, such as cystic fibrosis, may have increased susceptibility. BCC often causes infections in hospitalized patients as well [5]. Various traits distinguish BCC from other CF pathogens, such as its inherent resistance to many antibiotics, high transmissibility, and association with greater virulence. Because of these factors, management of B. cepacia complex patients is difficult [4].
Ecological Role
BCC bacteria exist throughout the environment. These bacteria can act as a powerful pesticide, capable of eliminating many soil-borne plant pathogens. In contrast, there is potential for the B. cepacia complex to act as a biofertiliser for rice that is cultivated in low fertile, low acidic soils [2]. Though initially discovered as a plant pathogen and capable of acting as a pesticide, these bacteria generally interact with plants in an ecologically beneficial manner. Additionally, their metabolic capacity and substantial genetic diversity allow them to degrade significant pollutants such as trichloroethylene [4]. Scientists are utilizing the diverse metabolic properties of BCC to create an assitive agent for bioremediation of contaminated environmental sites [4]. Currently, it is not possible to determine strains of B. cepacia that can be safely used in agriculture without potentially harming humans, which causes discourse between the scientific and agricultural communities [4].
Infection of cystic fibrosis patients
Pathogenesis
Transmission
Susceptible persons can acquire B. cepacia organisms through person-to-person contact, contact with contaminated surfaces, and exposure to it in the enviroment (i.e. soil and water) [6]. Transmission from contaminated medicines, nasal spray, mouthwash, and sublingual probes have been recorded also[6]. B. cepacia complex is highly transmissible between cystic fibrosis patients and can be transferred from one CF patient to another in hospitals, health clinics, and social environments [7].
Epidemiology
B. cepacia may colonize and cause respiratory tract infections in cystic fibrosis patients. It also may cause catheter-related infections in patients on hemodialysis or cancer patients through formation of biofilms. In patients treated with ceftazidime antibiotics and fluoroquinolones, there have been reports of B. cepacia nosocomial pneumonia. Soft-tissue, skin, surgical-wound, and genitourinary tract infections with B. cepaica have been reported [8]. In cystic fibrosis patients who contract cepacia syndrome, the pneumonic illness is fatal [2].
Virulence Factors
B. cepacia bacteria are able to produce a diverse collection of potential virulence factors, though not all of them have demonstrated a role in pathogenesis of human disease thus far [2]. A substantial amount of research has been undertaken to define the virulence factors expressed by B. cepacia bacteria because of its effects of cystic fibrosis patients [2].
Lipopolysaccharide (LPS), composed of O-antigen, core oligosaccharide, and lipid A, is a common virulence factor in Gram-negative bacteria. However, the LPS of BCC is distinct because the core oligosaccharide contains less phosphate and 3-deoxy-D-manno-oct-2-ulosonic acid than most other Gram-negatives. Additionally, 4-amino-4-deoxyarabinose moieties are attached to the phosphate residues in the lipid A backbone, which have been implicated in resistance to some antibiotic effects. Compared to the LPS of the other major CF pathogen, P. aeruginosa, the LPS of BCC is 4-5 times more endotoxic and induces heightened activity of neutrophil burst and induction of interleukin-8 (IL-8) from epithelial cells [2].
Lipase has been shown to effect the invasion of lung epithelial cells in BCC bacteria, but little is specifically known as to how [9].
Metalloproteases, specifically ZmpB, play a crucial role in the virulence of BCC bacteria in lung tissue. ZmpB is proteolytically active against a great number of proteins in the lung's extracellular matrix, including type IV collagen and fibronectin. It has also shown its ability to destory members of the immune system [9].
BCC bacteria express one of two flagellin types, whicha are distinguished by RFLP patterns of the fliC gene and and subunit size. These flagella have been shown to contribute to lung epithelial cell invasion through in vitro experimentation [2].
Because of P. aeruginosa's ability to grow in biofilms, it is thought that other CF pathogens may also be able to form biofilms. BCC has grown biofilms in vitro which show increased resistance to ciproflaxin and ceftazidime when compared with their planktonic counterparts. Biofilms may be highly advantageous for BCC bacteria, especially in the environment of a CF lung, because the bacteria may be better protected from antibiotics and host-defence mediators [4].
Clinical Features
Burkholderia cepacia complex infections are difficult to recognize because of their similarity to all other lung infections. These symptoms are often already occuring in cystic fibrosis patients, which causes even more difficulty in detecting the infection. Common symptoms include fever, cough, shortness of breath, congestion, and wheezing [10].
Diagnosis
Currently, the sole method of diagnosis for B. cepacia complex infection is the culturing of the patient's sputum. A culture will indicate which strains of Burkholderia, if any, are present within the patient [10].
Treatment
Clinical indicators of BCC infection vary. Cepacia syndrome is almost universally fatal, but aggressive treatment has yielded some success in specific cases [4]. BCC is often resistant to many common antibiotics, and treatment decisions are typically made on a case-by-case basis [5]. Because it is resistant to many antibiotics, effective therapies and treatments are not straightforward. Therefore, management efforts are primarily aimed at prevention of infection [6].
Prevention
Hand hygiene and other infection control procedures can reduce the risk of transmission. CF patients should not share hospital rooms with eachother and should limit contact in outpatient clinics, such as CF summer camps [5]
Host Immune Response
Little is known about the specific immune responses of those infected with BCC pathogens, but it is evident that persons with weakened immune systems, especially chronic lung issues, are especially susceptible to the pathogens. Its high transmissibilty creates even more vulnerability for patients with weakened immune systems and reinforces that cystic fibrosis patients should be kept separate from one another to prevent further infection [5].
References
1 Miller, S.C.M., LiPuma, J.J., Parke, J.L. “Culture-Based and Non-Growth-Dependent Detection of the Burkholderia cepacia Complex in Soil Environments.” Applied and Environmental Microbiology.
2 Mahenthiralingam, E., Urban, T.A., Goldberg, J.B. “The multifarious, multireplicon Burkholderia cepacia complex.” Nature.
3 Hua, C.N.C., Tokeshi, J. “Emergence of Burkholderia cepacia in Honolulu: A Case of Nursing Home-acquired B. cepacia sepsis. Hawai’i Journal of Medicine and Public Health.
4 Jones, A.M., Dodd, M.E., Webb, A.K. “Burkholderia cepacia: current clinical issues, environmental controversies and ethical dilemmas.” European Respiratory Journal.
5 Chu, K.K., Davidson, D.J., Halsey, K., Chung, J.W., Speert, D.P. “Differential Persistence among Genomovars of the Burkholderia cepacia Complex ina Murine Model of Pulmonary Infection.” American Society for Microbiology.
6 “Burkholderia cepacia in Healthcare Settings.” Centers for Disease Control and Prevention.
7 Gilchrist, F.J., Webb, A.K., Bright-Thomas, R.J., Jones, A.M. “Successful treatment of cepacia syndrome with a combination of intravenous cyclosporin, antibiotics and oral corticosteroids.” European Cystic Fibrosis Soceity.
8 Cunha, B.A. “Burkholderia.” Medscape.
9 Coenye, T., Vandamme, P., Govan, J.R.W., LiPuma, J.J. “Taxonomy and Identification of the Burkholderia cepacia Complex.” Journal of Clinical Microbiology.
10 Elborn, J.S. “Practical Management of Cystic Fibrosis.” Chronic Respiratory Disease.
Created by Paige Whitson
Student of Dr. Tyrrell Conway, University of Oklahoma