Comparing the Efficacy of Antibiotics Vs. Fecal Transplant in the Treatment of Clostridium Difficile Infection (CDI)
By Yara Awwad
Introduction
Clostridium difficile infection (CDI) has become a major source of morbidity and mortality at hospitals in recent years. [2] According to the CDC, around half a million Americans suffer from Clostridium difficile infection each year. Studies show that CDI became more severe, prevalent, and difficult to treat. [3] The infection is caused by the toxins produced by the bacteria Clostridium difficile and it spreads through spores. [4]
Clostridium difficile bacteria (shown in figure 1) are found around the environment is soil, food, air, water, and feces. Some people have C. diff in their large intestine in an inactive, non-infectious form. The bacteria also produce spores that ensure their survival under extreme conditions. [5] If these spores are formed in the colon, they can survive antibiotics and could turn into a pathogenic active form.
Depending on the severity of the Clostridium difficile infection, it can be treated using antibiotics that stop the bacteria from growing. However, some strains are becoming more resistant to antibiotics which is making CDIs harder to treat. If the infection is severe, surgery is usually performed to remove the diseased portion of the colon. Around 20% of the people with C.diff get the infection again.[6] In recurring cases, antibiotic therapy is conducted, but the effectiveness of this treatment has been declining. Recent research suggests that an alternative strategy for treating recurring CDI is through a fecal microbiota transplant (FMT), which has shown to have promising results.[7]
Bacteria & Pathogenesis
Clostridium difficile is a gram-positive bacillus bacteria that is part of the normal intestinal microbiota of 1-3% of adults and 15-20% of infants. [8] The clostridium genus consists of relatively large, rod shaped, and motile bacteria in the Firmicutes phylum. The genus has more than 100 known species including harmful pathogens such as Clostridium tetani, C. butyricum, and Clostridium sordellii, which produce some of the most potent toxins in human’s history. [5]
Clostridia are anaerobic and spore-forming bacteria that is commonly found in oxygen-deficient soil. They produce spores under stress which makes them highly resistant to physical and chemical influences. C. difficile spores germinate in the colon and form vegetative cells that initiate CDI. Due to its anaerobic nature, C. difficile is unable to survive aerobically in the vegitative form. Therefore, during the course of the infection, C. difficile induces the sporulation pathway that produces more dormant spores, which remain stable for long times. The spores are responsible for the persistence and recurrence of the infection in patients. They are also the source of horizontal transmission that occurs between patients. [9]
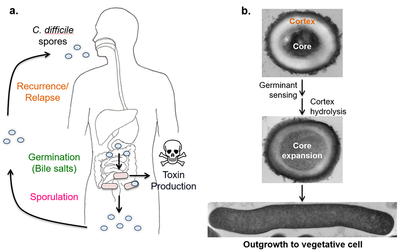
In the presence of cholic acid derivatives, the germination of spores plays a critical role in the pathogenesis of CDI.[11] Germination of spores is a biophysical process that results in the loss of spore specific properties. This includes the transitioning from the metabolically dormant spore form to the active-growing, toxin forming vegitative form. In C. difficile, this process is initiated by cholate derivatives, bile acids, and amino acid signals from the host cells. Germination is activated when the germinants bind to Ger-type receptors that are found inside or on the surface of the inner spore membrane.[12] As shown in figure 2, the signals trigger cortex hydrolysis by cortex lytic enzymes which are found in the spore coat region. The spore cortex is a thick layer of modified peptidoglycan that surrounds the cell well and helps prevent osmolysis. The coat and the cortex degenerations release Ca-DPA and result in the full core rehydration and the initiation of the outgrowth of a new vegetative cell. [13]
Through the infection cycle (figure 2), pathogenic strains of C. difficile grow and produce two major toxins, Toxin A (TcdA) and toxin B (TcdB). Certain C. diff strains are also able to produce a binary toxin called C. diff transferase (CDT), which is not as prevalent or severe as the other two toxins. Only a few strains can produce CDT in the absence of toxins A and B.These toxins are part of the large clostridial glucosylating toxin (LCGT) family. TcdA and TcdB act on the colon epithelium and immune cells, inducing a cascade of complex reactions that result in the secretion of fluids, inflammations, and tissue damage, all of which are important marking features of CDI. [14] Toxins A and B are produced in the Pathogenicity loci (PaLoc) by the genes tcdA and tcdB respectively. The expression of these genes is regulated by several environmental conditions including the availability of nutrients and temperature. PaLoc can be transferred horizontally to non-pathogenic strains converting them into pathogenic strains. [15]
Toxins enter the cells by endocytosis then they are translocated to the host cell cytosol. In the cytosol, TcdA and TcdB glucosylate several members of the Rho subfamily and result in the inactivation of Rho proteins. These proteins have several functions and they interact with the kinases and phospholipases in the host cell that are responsible for signal transduction pathways. Rho proteins regulate actin cytoskeleton, the cell cycle progression, and the phagocytosis and cytokine production. Therefore, the toxins can induce cytopathic effects causing the loss of cell-cell contact and increase the epithelial permeability. All of which lead to cell apoptosis. [15]
Toxin A results in the direct damage of the intestinal mucosa. This causes symptoms of CDI including pseudomembranous colitis, which is the swelling or inflammation of the colon due to the growth of C. difficile. During pseudomembranous colitis, the toxins are able to disrupt the tight junctions of epithelial barriers. This provides a way for neutrophils to accumulate in the intestines. In severe colitis cases, the toxins kill the tissues of the colon’s inner lining and cause the tissues to fall. [16]
Toxin B is an essential virulence factor, but it is not as important as toxin A. This was proven when mutant cells with inactive tcdA still produced the same levels of tcdB, while mutant cells with inactive tcdB produced tcdA 2 to 3 times more. [17]
Risk factors and transmission
Clostridium difficile can live in the human intestines without causing any diseases or illnesses. However, people begin to get sick when the spores are formed. In a healthy intestine with normal microbiota, the germination of C. difficile spores is prevented by other bacteria and the processing of cholate derivatives. Patients receiving several antibiotic treatments usually have most of their intestinal microflora disrupted or killed. This prevents the metabolism of cholates which in turn results in the germination and outgrowth of the C. difficile spores. [15] The lower competition in the intestine due to the absence of gut flora helps the spores to thrive and become more pathogenic. The antibiotics that most lead to CDI include penicillin, Fluoroquinolones, Cephalosporins, and Clindamycin.
The majority of CDI cases occur in people who had recent visits to a health facility. In these places, the most common method of infection spread mainly on hands from one person to another. Other risk factors associated with the development of CDI include advanced age, immunocompromised individuals, and renal diseases. One of the studies indicated that becoming infected with CDI is around 10 times greater in people who are 65 years or older. Women and people who previously had the infection are more likely to be infected. The risk continues to rise with each infection.
Spores are metabolically dormant which means that they are intrinsically resistant to antibiotics and attacks from the host's immune system. Therefore, spores from C. difficile are excreted in feces and spread in food and on surfaces when infected individuals do not wash their hands regularly and carefully. [6] Once spores are shed into the environment, they are also resistant to disinfectants that do not have bleach in them. [9] Therefore, the spores can be easily found on surfaces and equipment at hospitals, nursery homes, and households.
The best methods to prevent the transmission of CDI is by washing hands regularly. Soap and warm water are encouraged to maintain hygiene since most hand sanitizers do not effectively destroy the spores. Cleaning products containing bleach should be used to kill spores off of surfaces and objects. It is also very important to avoid the use of unnecessary antibiotics to maintain the healthy functioning of flora in the intestines and prevent the formation of spores.[6]
Symptoms
Clostridium difficile infection ranges from asymptomatic colonization and mild diarrhea to toxic and life-threatening megacolon and the inflammation of the lining of the abdominal. [9] The signs and symptoms of the infections take an average of 5 to 10 days to develop. The symptoms of the mild to moderate CDI include diarrhea around three times a day for at least two consecutive days and mild abdominal cramping and tenderness. [6]
Severe CDI, the colon becomes inflamed and sometimes forms patches of raw tissues that bleed or produce pus. The symptoms of the severe infection include blood or pus in the stool, and watery diarrhea 10 to 15 times a day, which leads to severe dehydration due to the lack/disruption of electrolytes in the body. This can cause blood pressure to drop to abnormally low levels which increases the heart rate. If dehydration occurs quickly and not enough liquid is supplemented, kidney function rapidly deteriorates which could result in kidney failure. Fever, nausea, appetite and weight loss, increased white blood cells count, and swollen abdomen are all common symptoms of CDI.
In some rare cases, the colon will be unable to expel gas and stool causing it to become enlarged (megacolon). If the colon is left untreated, it could rupture and cause the bacteria to enter the abdominal cavity. Moreover, in more extreme cases, extensive damage to the lining of the intestine which could lead to the formation of a hole in the large intestine. This also results in bacteria to be spilled from the large intestine into the abdominal cavity. All the aforementioned cases could eventually lead to life-threatening consequences.[6]
Treatments
The first step to treating CDI is by stopping the administration of any other antibiotics that are not prescribed to treat this infection. The most common/standard way of treating this infection is by taking antibiotics that prevent C. difficile from growing. The antibiotics vancomycin and fidaxomicin are usually used for this purpose. Around 10 percent of the patients do not respond to the antibiotic treatment the first time and approximately 20% of treated patients are reinfected with CDI. [19] In cases of non-respondents or recurrence, treatments are usually conducted either by antibiotics or a fecal microbiota transplant.
Probiotics are also being extensively tested for the preventative nature of the infection. The role of probiotics in CDI is controversial. However, recent studies are providing emerging evidence for their role in the primary prevention of CDIs. [20]
Antibiotics
For the first recurrence, tapered and pulsed vancomycin are usually used if vancomycin was used for the first treatment. Tapered/pulsed vancomycin is used to target the spores. After germination, a prolonged course of the tapered/pulsed regimen is given to attack the vegetative form of the cells. An alternative antibiotic is fidaxomicin, which has a narrower range than vancomycin and thus causes minimal disruption in the gut flora. Ridinilazole is another antibiotic used in phase two treatment that targets clostridia while causing minimal damage to the flora. [21]
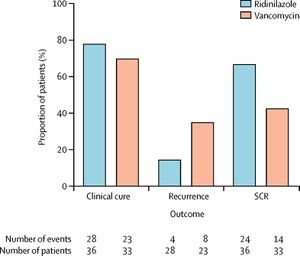
A study conducted by Vickers and colleagues in which patients who tested positive for CDI were split into two groups, a group that receives oral vancomycin and the other receives oral ridinilazole. They studied the sustained clinical response (SCR) which includes a cure at the end of the treatment and no recurrence after 30 days of infection clearance. As presented in figure 3, the results show that ridinilazole exhibited superiority over vancomycin in a sustained clinical response.[18] Therefore, studies show that ridinilazole and fidaxomicin are generally used in first treatments and they show better results in terms of a sustained clinical response when compared to other antibiotics, but their use is limited due to their high costs. These antibiotics are also used if the first treatment with vancomycin fails. However, the use of ridinilazole or fidaxomicin for recurrent CDI is still being studied.
Fecal Microbiota Transplant (FMT)
Fecal transplant, also known as stool transplant, is an emerging method for treating recurrent CDI. It consists of the infusion of fecal microbiota from a healthy donor into the infected patient. The altered healthy colon microbiota is the main cause of recurrent CDI and the restoration of the healthy flora in the colon is the principle of FMT. The most common way of delivering FMT is through colonoscopy, which is a safe method but it could cause mild bloating, gasses, and fever. [21]
In a study done by Cammarota and colleagues, they compared the effects of using vancomycin and FMT in the treatment of recurrent CDI. They did this by conducting a randomized controlled clinical trial. Patients were randomly assigned to one of the treatments (standard vancomycin or FMT). Follow Up with patients was conducted 10 weeks after the treatment. The cure of C. difficile infection was defined by the disappearance of diarrhea and two negative stool tests for C. difficile toxins. Recurrence after the treatment is defined by diarrhea at least three times for two consecutive days. Statistical results provided evidence that FMT had a significantly higher efficacy than vancomycin. Overall, 90% of the FMT treatment group was cured while 26% of the vancomycin group was treated. [23]
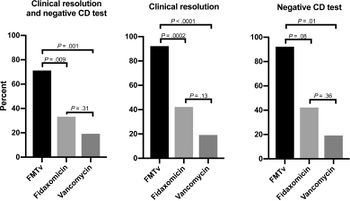
In another study conducted by Lodberg Hvas and colleagues, they found that FMT is superior to fidaxomicin in treating recurrent CDI as demonstrated in figure 4. A low hemoglobin level was used as a predictor for the failure of the fecal transplant. FMT delivered by colonoscopy after a short course of vancomycin has higher efficacy than fidaxomicin. In this study, they also found that the resolution rates for vancomycin and fidaxomicin are not statistically significantly different.[22]
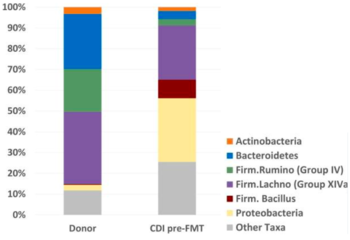
FMT has been shown to significantly impact the overall microbiota composition. This was demonstrated in a study done by Mintz et al where they characterized how FMT changes fecal microbiota in 12 patients with recurrent clostridium difficileinfection by analyzing 16S rRNA sequences. The results showed that several imbalances including the relative abundance of three strictly anaerobic groups (Bacteroidetes, Firmicutes Ruminococcaceae, and Firmicutes Lachnospiraceae) were less before the FMT treatment when compared to fecal samples of healthy donors. On the other hand, the relative abundance of Proteobacteria and Firmicutes Bacillus was higher in CDI patients before treatment in comparison with healthy donors. They also assessed the overall differences of microbiota composition (beta-diversity) using a fitted linear mixed model. Mintz and colleagues found that the beta-diversity differed significantly in the samples before the FMT treatment from the samples after the FMT treatment. Their results also showed that the alpha diversity of 3 months after FMT treatment increased from the alpha-diversity of pre-FMT. PCR analysis was used to examine the relative abundance of F. prausnitzii, a gram-positive and anaerobic bacteria that is a leading representative in the Firmicutes phylum and part of the Ruminococcaceae family. It is one of the most abundant and important commensal bacteria of the human gut microbiota. This bacteria is involved in the generation of butyrate, a critical nutrient for maintaining colonic mucosal homeostasis. Before the fecal transplant, patients had reduced relative abundance of F. prausnitzii compared to healthy donors as shown in figure 5 where Ruminococcaceae abundance significantly decreases in CDI patients. The results found that after the fecal transplant, CDI patients experienced increased abundance in F. prausnitzii. It is thought that the increase in the relative abundance specifically of F. prausnitzii taxa plays an important role in the prevention of recurrent CDI after fecal transplant, but this has not been verified experimentally yet. Therefore, this study has provided substantial evidence that FMT is an effective treatment for preventing recurrent CDI by restoring the normal functioning of gut microbiota.[24]
The restoration of gut microbiota after a FMT is particularly important for the metabolism of primary bile acids to secondary bile acids in the colon. The primary bile acid is a potent germinate and is a key component of C. difficile growth media. On the other hand, secondary bile acid has been shown to inhibit the germination of C. difficile. In a study done by Weingarden and colleagues, they examined the influence of FMT on the metabolism mediated by the gut microflora in CDI patients. They also analyzed the composition of fecal microbiota in the patients before and after treatment and compared it with a healthy donor. They showed that the restoration of a normal microbiota composition is accompanied by the rapid normalization of fecal bile acid in recurrent CDI patients. Similar to results found in the Mintz et al study, they found that taxonomic bacterial composition of fecal samples from patients who received a FMT treatment showed a rapid change as they became more similar to the healthy donor after the procedure as shown in figure 6. The fecal samples before the transplant contained a high concentration of primary bile acids and salts, while secondary bile acids were almost undetectable. Contrary to this and similar to the donor’s sample, post-FMT samples contained mostly secondary bile acids. Therefore, similar to the aforementioned papers, this study showed that FMT results in the normalization of fecal bacterial communities and their metabolic composition. It also provided evidence that FMT is able to restore the normal metabolism of primary bile acids to secondary bile acids in patients with recurrent CDI. The correction of bile acid metabolism is thought to be one of the mechanisms by which FMT prevents recurrent CDI. [25]
FMT is, therefore, an effective method for treating recurrent CDI. However, according to the FDA, there is a risk of transmitting multi-drug resistant organisms (MDRO) through a fecal transplant, which could result in serious adverse outcomes. As of 2019 two immunocompromised individuals developed invasive bacterial infections due to the transmission of MDRO by FMT. Therefore, more research needs to be done to understand the exact mechanism by which FMT inhibits recurrent CDI and to possibly prevent any negative bad effects associated with this treatment method. [26]
Conclusion
Clostridium difficile infection is caused by the formation of resistant spores in the colon of the human intestine. There are several methods for preventing the spread of CDI including personal hygiene specifically washing hands, and limiting the use of antibiotics. CDI is posing a challenge to the health care systems across North America and Europe due to its recurring nature and its severe impact on the gut flora. Several studies have indicated that fecal microbiota transplant is the most recommended treatment for recurrent Clostridium difficile infection. Antibiotics had a significantly lower sustained clinical response when compared with FMT and some people do not respond to antibiotics treatment. The efficacy of FMT has been demonstrated in several studies that prove the effective restoration of the gut microflora post-FMT.
References
- ↑ Clostridioides difficile Infection. (2019, November 13). Retrieved from https://www.cdc.gov/hai/organisms/cdiff/cdiff_infect.html
- ↑ Liubakka, A., & Vaughn, B. P. (2016, July). Clostridium difficile Infection and Fecal Microbiota Transplant. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/27959316
- ↑ Nearly half a million Americans suffered from CDI in a single year. (2017, March 22). Retrieved from https://www.cdc.gov/media/releases/2015/p0225-clostridium-difficile.html
- ↑ Harvard Health Publishing. (n.d.). Clostridium difficile: An intestinal infection on the rise. Retrieved from https://www.health.harvard.edu/staying-healthy/clostridium-difficile-an-intestinal-infection-on-the-rise
- ↑ 5.0 5.1 Clostridium. (n.d.). Retrieved from https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/clostridium
- ↑ 6.0 6.1 6.2 6.3 6.4 C. difficile infection. (2020, January 4). Retrieved from https://www.mayoclinic.org/diseases-conditions/c-difficile/diagnosis-treatment/drc-20351697
- ↑ What is C. diff? (2020, March 27). Retrieved from https://www.cdc.gov/cdiff/what-is.html?CDC_AA_refVal=https://www.cdc.gov/hai/organisms/cdiff/cdiff-patient.html
- ↑ Goudarzi, M., Seyedjavadi, S. S., Goudarzi, H., Mehdizadeh Aghdam, E., & Nazeri, S. (2014). Clostridium difficile Infection: Epidemiology, Pathogenesis, Risk Factors, and Therapeutic Options. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4058799/
- ↑ 9.0 9.1 9.2 Paredes-Sabja, D., Shen, A., & Sorg, J. A. (2014, July). Clostridium difficile spore biology: sporulation, germination, and spore structural proteins. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4098856/#R11
- ↑ Graduate School of Biomedical Science. (2019, March 31). Retrieved from https://gsbs.tufts.edu/facultyResearch/faculty/shen-aimee/research
- ↑ Zhu, D., Sorg, J. A., & Sun, X. (2018, February 8). Clostridioides difficile Biology: Sporulation, Germination, and Corresponding Therapies for C. difficile Infection. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5809512/
- ↑ Shrestha, R., Cochran, A. M., & Sorg, J. A. (n.d.). The requirement for co-germinants during Clostridium difficile spore germination is influenced by mutations in yabG and cspA. Retrieved from https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.100768/
- ↑ Kochan, T. J., Foley, M. H., Shoshiev, M. S., Somers, M. J., Carlson, P. E., & Hanna, P. C. (2018, July 25). Updates to Clostridium difficile Spore Germination. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6060349/
- ↑ Chandrasekaran, R., & Lacy, D. B. (2017, November 1). The role of toxins in Clostridium difficile infection. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5812492/
- ↑ 15.0 15.1 15.2 Di Bella, S., Ascenzi, P., Siarakas, S., Petrosillo, N., & di Masi, A. (2016, May 3). Clostridium difficile Toxins A and B: Insights into Pathogenic Properties and Extraintestinal Effects. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4885049/
- ↑ Lee, D. (2016, October 11). C. diff (C. difficile Colitis): Symptoms, Treatment & Causes. Retrieved from https://www.medicinenet.com/clostridium_difficile_colitis/article.htm#clostridium_difficile_c_difficile_facts
- ↑ Voth, D. E., & Ballard, J. D. (2005, April). Clostridium difficile toxins: mechanism of action and role in disease. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1082799/
- ↑ 18.0 18.1 Vickers, R. J., Tillotson, G. S., Nathan, R., Hazan, S., Pullman, J., Lucasti, C., … CoDIFy study group. (2017, July). Efficacy and safety of ridinilazole compared with vancomycin for the treatment of Clostridium difficile infection: a phase 2, randomised, double-blind, active-controlled, non-inferiority study. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/28461207
- ↑ Clostridium Difficile Infection | C. difficile. (2020, April 13). Retrieved from https://medlineplus.gov/clostridiumdifficileinfections.html
- ↑ Goldstein, E. J. C., Johnson, S. J., Maziade, P.-J., Evans, C. T., Sniffen, J. C., Millette, M., & McFarland, L. V. (2016, December 14). Probiotics and prevention of CDI. Retrieved from https://www.sciencedirect.com/science/article/pii/S1075996416301664
- ↑ 21.0 21.1 Singh, T., Bedi, P., Bumrah, K., Singh, J., Rai, M., & Seelam, S. (2019, July). Updates in Treatment of Recurrent CDI. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6575119/
- ↑ 22.0 22.1 Hvas, C. L., Jørgensen, S. M. D., Jørgensen, S. P., Storgaard, M., Lemming, L., Hansen, M. M., … Dahlerup, J. F. (2019, January 2). Fecal Microbiota Transplantation Is Superior to Fidaxomicin for Treatment of Recurrent Clostridium difficile Infection. Retrieved from https://www.sciencedirect.com/science/article/pii/S0016508518354349
- ↑ Cammarota, G., Masucci, L., Ianiro, G., Bibbò, S., Dinoi, G., Costamagna, G., … Gasbarrini, A. (2015, March 1). Randomized clinical trial: faecal microbiota transplantation by colonoscopy vs. vancomycin for the treatment of recurrent Clostridium difficile infection. Retrieved from https://onlinelibrary.wiley.com/doi/full/10.1111/apt.13144
- ↑ 24.0 24.1 first, Mintz, M., Khair, S., Grewal, S., Lacomb, J. F., Park, J., Channer, B., … Li, E. (2018). Longitudinal microbiome analysis of single donor fecal microbiota transplantation in patients with recurrent Clostridium difficile infection and/or ulcerative colitis. Plos One, 13(1). doi: 10.1371/journal.pone.0190997
- ↑ second, Weingarden, A. R., Chen, C., Bobr, A., Yao, D., Lu, Y., Nelson, V. M., … Khoruts, A. (2014). Microbiota transplantation restores normal fecal bile acid composition in recurrent Clostridium difficile infection. American Journal of Physiology-Gastrointestinal and Liver Physiology, 306(4). doi: 10.1152/ajpgi.00282.2013
- ↑ FDA, Commissioner, O. of the. (n.d.). FDA warns about potential risks related to investigational use of FMT. Retrieved from https://www.fda.gov/news-events/fda-brief/fda-brief-fda-warns-about-potential-risk-serious-infections-caused-multi-drug-resistant-organisms
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.
