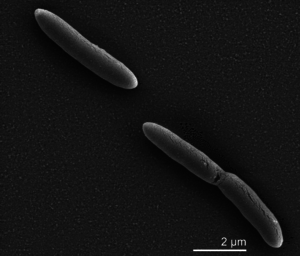
Desulfotomaculum
Classification
Higher Order Taxa:
Bacteria; Firmicutes; Clostridia; Clostridiales; Peptococcaceae
Species:
Desulfotomaculum acetoxidans, D. aeronauticum, D. alcoholivorax, D. alkaliphilum, D. antarticum, D. autralicum, D. carboxydivorans, D. geothermicum, D. gibsoniae, D. guttoideum, D. halophilum, D. hydrothermale, D. kuznetsovii, D. luciae, D. nigrificans, D. putei, D. ruminis, D. sapomandens, D. solfataricum, D. thermoacetoxidans, D. thermobenzoicum, D. thermocisternum, D. thermosapovorans, D. thermosubterraneum, D. varum
Description and Significance
Desulfotomaculum bacteria are sulfur reducing, obligate anaerobes. D. thermosubterraneum for instance was isolated from an underground mine in Japan and found to live alongside methanobacterium archaea. Similar relationships can be found for most species of this genus due to high temperature, fluctuations between SO4 and CH4 concentrations and varied pH levels. Many species have in fact been found in truly ancient places such as within iron deposits and meteorite water 5 kilometers underground, which is almost 2 kilometers deeper than most mines are even dug.
An experiment using D. nigrificans demonstrated just how useful sulfur reduces can be. Eucalyptus tereticornis bark was first used as a means to absorb the metal ions and raise the pH from 2.3 to 5.5. The bark and water together then served as a positive growth medium for D. nigrificans. E. tereticornis removed 41% sulfate from the water, but once paired with the bacteria, the sulfur levels became reduced to as much as 84% at a pH of 5.5. It has also been show that sulfur reducing bacteria in mine shafts and aquifers are large contributors to degradation of their respective walls.
Cell Structure and Metabolism
Most Desulfotmaclum stain negative, but under electron microscope are gram-positive due to a lipid layer. All species are obligate anaerobes and can be either bacillus or vibrio. Approximate size ranges from ~0.5 X 1-6 micrometers, and grow optimally in basic pH. Population growth is generally slower than aerobic bacteria because sulfur is the limiting reagent and bacteria must be in contact with it. Some species are slight thermophilic, at 53C, or mesophilic, at 37C. Their DNA G-C contents do not appear to correlate to optimal temperature conditions, some of the species were able to live at moderate thermophilic conditions with a G-C content of 43%. Cell membrane composition and chaperone proteins likely account for high temperature survivability. Almost all species are motile using peritrichous flagella.
Desulfotomaculum are very poor at metabolizing in the absence of sulfur compounds. Spores formed in poor conditions are spherical and can be located either central or lateral. Desulfotomaculum thermosubterraneum spores are capable of germination after intense heat for thirty minutes with minimal cell death. The most common electron donors are H2, acetate, formate, ethanol, lactate and pyruvate. All species use at least one of the following as their main electron acceptors: sulfite, sulfate and thiosulfate. While most sp. are unable to tolerate NaCl above 1.5%, D. varum found in bore well runoff thrives in it. As mentioned before, the high range of niches they inhabit can be exploited to fix problems where most life can’t survive.
Ecology
Desulfotomaculum are usually found living in underground water, inside sediment or rock, or within feces of animals (mainly pigs) where little oxygen has reached. Notably D. alkaliphilum is the only known obligate alkaliphile, only grows in pH between 8.5-11, and found within pig and cow manure.
D. varum and D. australicum have been noticed as the cause to deteriorating bore holes in the Great Artesian Basin of Australia. The process is not well known, but it is directly tied to greater access to and metabolism of sulfur like molecules. The bacteria are thought to have already been in the bore-hole locations, but were given greater access to sulfur with the water currents.