Ehrlichia: tick-borne pathogen in canines and humans
Introduction
By Emily Tysinger
Ehrlichia is an obligate intracellular bacterium of the family Anaplasmataceae- a classified α-proteobacteria. These Gram- negative coccobacilli, are best known for their etiological agency in the transmission of a group of tick-borne illnesses known as Ehrlichiosis (Ismail et al. 2011). All members of the genus Ehrlichia are pathogenic, causing mild to severe symptoms, specifically in humans and canines. This genus includes E.chaffeensis,E.ewingii, and E.canis (Nicholson et al. 2010). These bacteria are considered the agents responsible for the emergence of Human Monocytotropic Ehrlichiosis and canine Monocytotropic Ehrlichiosis. The Ehrlichial agents are hosted in vertebrate reservoirs and transmitted to both dogs and humans by “hard” tick vectors. Like many other tick-borne illnesses, the demographic distribution of Ehrlichiosis is greatest in regions with a high density of primary tick vector populations; namely the Lone Star Tick (Amblyomma americanum) and the Brown Dog Tick (Rhipicephalus sanguineus) (Nicholson et al 2010).
Globally, the two most common forms of disease causing Erlichia are Ehrlichia canis and Ehrlichia chaffeensis. The first case of Ehrlichiosis emerged from Africa in the mid 1930’s, reported by Donatien and Lestoquard. This Canine Monocytic Ehrlichiosis did not gain meaningful attention until the 1970s, when a significant number of cases were reported in German shepherds during the Vietnam War (Harrus et.al 2010). The disease is composed of 3 distinct phases described as acute, subclinical, and chronic. Symptoms are similar for all stages, however the chronic stage is exceedingly more severe than the acute and subclinical stages. Dogs entering the chronic phase of the disease must undergo intense and expensive treatment and in most cases have little hope in terms of survival. E.canis has been reported to infect all breeds of dog; however, the German shepherd seems to be most susceptible breed having the highest mortality rate compared to other dogs infected with the bacteria (Harrus et.al 2010). The tick vector responsible for transmission of Ehrlichiosis in canines is the brown dog tick. Until recently, E.canis was thought to only infect canines. However, in 2006 a study was done that isolated E.canis in human patients proving compatibility for human pathogenic infection in addition to E.chaffeensis and E.ewingii (Nicholson et.al 2010).
The primary agent for Human Monocytic Ehrlichiosis is E. chaffeensis, transmitted by the Lone Star tick. Ehrlichiosis in human patients was first detected in the United States in 1986, and has since emerged as one of the leading tick-borne illness, next to Rocky Mounted Spotted fever and Lyme’s Disease (Ismail et al 2010). E.ewingii has also emerged in recent years as the causative agent of a more mild case of human ehrlichiosis. Most cases of E. ewingii since 1999 have been reported in patients with HIV or other infections that coincide with decreased immune response (Ismail et al. 2011). Most occurrences of infection by E.canis and E.chaffeensis are predominantly reported in the south central and south eastern United States, as this is the natural ecological range of the known tick vectors.
Morphology and Replication
Ehrlichia are small, nonmotile, pleomorphic bacteria consisting of a thin, rippled outer membrane, separated from the cytoplasmic membrane by periplasmic space. According to Harrus et.al, each bacterial cell consists of one single chromosome, circular in shape, consisting of 1,315,030 nucleotides (2010). Using electron microscopy, both coccoid and coccobacillary (or reticulate and dense-cord) morphologic cell forms have been identified. The coccoid, or Dense-cord cells, are the smaller of the two cell forms, measuring 0.4 µm to 0.6µm in diameter. In contrast, reticulate cells measure 0.4 to 0.6µm by 0.7 to 1.9 µm. In comparison to reticulate cells, which contain regularly dispersed nuceloid DNA fibrils and ribosomes within the cytoplasm, Dense-cord cells are reported to have an electron dense, centrally located chromatin and ribosomes (Ismail et. al 2010).
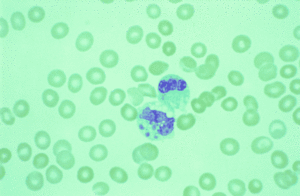
As a Gram-negative bacteria Ehrlichia are bound within an outer cell membrane. Interestingly, certain important pathogenic elements such as lipopolysaccharides and peptidoglycan are found lacking within the cell membrane (Ismail et al. 2010). While these important components are not present Ehrlichia cell walls do contain a significant amount of cholesterol. As reported by Ismail et.al, the presence of cholesterol comes from the host cell and may have importance in relation to ehrlichial survival and passage into the host cell (2010). In both canines and humans, Ehrlichia reside in the membrane-lined cytoplasmic vacuoles of infected Eukaryotic leukocytes in their mammalian hosts (1). Within the cytoplasmic vacuoles Ehrlichia form morulae (Fig 2). Morulae are formed by lose to condensed “mulberry” shaped microcolonies of bacteria, which color a dark blue when stained with Romanovsky-type stains (Paddock et.al 2003). Ehrlichia both resides and replicates within the vacuoles of host cells. E.canis and E.chafeensis replicate within the membrane-bound compartments of mononuclear phagocytes (Ismail et. al 2010).
Transmission and Pathogenesis

Transmission
As a tick- borne illness the transmission of Ehrlichia to both humans and canines occurs through the bite of an infected tick. Ehrlichial infections by E.canis and E.chaffeensis are considered among the plethora of zoonotic diseases in existence today (Nicholson et.al 2010). Zoonotic diseases are characterized by the transmission of bacteria and infection from animal to human. The characteristic transmission of Ehrlichia is cyclic. Transmission cycles for Ehrlichia bacteria are extremely important as they are responsible for maintaining these bacterial pathogens within tick populations and ensure the continued transmission to humans and canines (Nicholson et. al 2010). The transmission of Ehrlichia requires primary mammalian reservoir hosts and arthropod vectors. For both species of Ehrlichia, E.canis and E. chaffeensis, there may be a large range of vertebrates that have the potential to serve as competent hosts and as sources of blood for tick vectors (Ganguly et.al 2008). The primary reservoir host for E.chaffeensis is known to be the white tailed deer (Odocoileus virginianus). These mammals are naturally infected with the bacteria and have come to be considered as the only vertebrates recognized as a complete and sufficient host for maintaining the transmission cycle of E.chafeensis. Next to the White- tailed deer, domestic dogs and domestic goats also act as important reservoir hosts for Ehrlichia. These three are important because of their regular exposure to ticks and their close proximity to human population (Paddock et. al 2003). Ticks have the primary role in the transmission process of Ehrlichia and act as vectors, carrying the pathogenic bacteria to and from their multiple hosts. In addition, tick vectors are the agents of transmission from vertebrate hosts to humans and canines. The central agent of transmission for E. chaffeensis is the Abmlyomma americanum or the lone star tick endemic to the Southern United States (Ganguly et.al 2008). E. canis however, is mainly transmitted by Rhipicephalus sanguineus, the brown dog tick and tends to have a more worldwide disease distribution (Shipov 2008).
The transmission cycle for both E.chaffeensis and E.canis begins with the dropping of eggs by an adult female tick. Once hatched these larvae are considered non-infected, as unfed eggs and larvae are presumed to be infection free. As transovarial transmission has yet to be demonstrated, larvae cannot contract the disease until they have obtained infected blood from a vertebrate reservoir (Ganguly et.al 2008). Once infected, these larvae maintain Ehrlichia up till the nymphal stage. Infected nymphs may either establish infection in another reservoir host, or pass Ehrlichia directly to a susceptible canine or human. From the nymphal stage, the ticks molt into infected adults after leaving a second host. At this stage infection can again be established either within a susceptible reservoir or human host (Paddock et.al 2003). The transmission cycle is reliant upon tick vectors and vertebrate hosts (Fig 3).
Pathogenesis
As Ehrlichia do not contain pili the process of entering the host and facilitating infection occurs once Ehrlichia bind to the host cell by way of its outer membrane (Paddock et. al. 2003). After the bacteria are enveloped within the host cell, Ehrlichia form membrane bound compartments (endosomes) that help to maintain their distinct cytoplasmic compartments (Paddock et al. 2003). Once transmitted, E. chaffeensis and E. canis target and infect the mononuclear phagocytic cells. Most generally infected are the monocytes, or blood cells, within the human or canine host. However, monocytes are not the only cells that are susceptible to infection. Further studies have reported ehrlicial infection within lumphocytes, promyelocytes, and metamyelocytes as well. It is assumed however, that because of their more frequent and higher rates of infection, mononuclear phagocytes are capable of maintaining the productive infection within their cells (Paddock et.al 2003). Typically an infected monocyte contains approximately 1 or 2 morulae (Ismail et al, 2008). Ehrlichia maintain and ensure their survival by multiplying within the host endosomes. Consequently, these pathogens are able to guarantee their survival because they possess the capability to reprogram the systems and mechanisms of defense employed by the host cell (Ismail et. al 2010). In human monocytic ehrlichiosis, E.chaffeensis acts as an inhibitor in the fusion of the phagolysosome and has the ability to suppress the signal transduction pathway of anti-ehrlicial activity (Ismail et.al 2010). In patients with severe and fatal HME that do not exhibit prior immune problems the intensity of the infection is thought to be the result of a deregulation of the immune system of the host may be the cause of multisystem organ failure and severe tissue damage according to Ismail et. al (2008). Additionally, studies have have illustrated the multi-sytemic nature of both Human and Canine monocytic ehrlichiosis resulting in multi-organ infection (Ganguly et.al 2008). In HME patients without prior immune difficulties, severe to fatal multi-organ infection has been linked to a decline in the immune response of the host, leading to severe tissue damage and in the most extreme cases, organ failure (Ismail et.al 2010). Infection of the splenic chords, periarteriolar sheaths, lymph nodes, liver, and bone marrow has been reported in human cases of ehrlichiosis, due impart to the sheer number of mononuclear phagocytic cells contained within these tissues (Paddock et.al 2003).
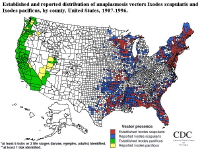
In comparison, pathological findings in canines have also described ehrlichial DNA present within the lymph, spleen, and kidneys (Waner et al 2013). Indicated by previous research, it may be determined that the transmission and infection of E.canis and E.chaffeensis can in fact be widespread, affecting the varying systems in the body of the infected animal (Waner et al 2013). In the most severe chronic stages of the disease the highest density of ehrlichiea are found within the bone marrow (Waner et. al 2013). In the United States, Ehrlichia transmission is most common in the southern region of the country- with the majority of cases reported form Southcentral and Southeastern states. Infection by E.canis and E. chaffeensis is most commonly reported in Texas, Tennessee, Oklahoma, Georgia, Arkansas, and North Carolina, among others (Fig 4) (Paddock et al 2003). As with Lymes disease, found most frequently in the Northeastern region of the United States, it is consistently confirmed that the highest incidence of Ehrlichiosis follows the same pattern of distribution as the responsible tick vectors and vertebrate reservoir hosts (Ismail et. al 2010). It has been noted that those areas with the highest density of Deer tick and Brown Dog tick have the highest reported cases of ehrlichiosis. Interestingly, reports of both human and canine monocytic ehrlichiosis fluctuate in relation to the season. As reported by Paddock et al., 70% of the verified cases of human monocytic ehrlichiosis occur between the months of May and July (2003). The seasonality and location of these reported cases corresponds very closely to the peak feeding times and endemic regions of the tick vectors.
Diagnosis and Treatment
There are multiple ways to use laboratory diagnosis to test for Ehrlichia infection in both canines and humans. The initial prognosis can be determined through the use of biochemical and hematological tests. As stated by Harrus et al. when viewed using light microscopy, blood smears revealed the morulae formed in the monocytes of canines, indicating infection by Ehrlichia canis (Harrus et al. 2010). In addition, PCR amplification, serological testing, and species isolation are also employed during the diagnostic process. Biochemical tests are used to effectively evaluate the concentration of protein levels in canines, as protein concentrations have been noted to increase during the acute phase (Harrus et al 2011). PCR is the most effective and thus the most commonly used assessment employed in laboratory diagnosis. It is most effective in the process of indicating ehrlichial infection, even when antibodies are at very low levels during the early stages of the disease due to its high sensitivity and specificity for ehrlichial illness (Ismail et al 2010). Serological tests are used to measure the reactivity of antibodies with E.chaffeensis or E. canis (Paddock et al 2003).
The disease may be hard to diagnose or detect as Ehrlicial illnesses present a wide array of symptoms and can manifest in three different phases (Paddock et al. 2003). All phases display a varied collection of symptoms, each specific to its severity. For example the acute phase is often characterized by a high fever, depression, lethargy, and anorexia (Harrus et al 2011). Blood smears and hemological indicators are frequently used for the diagnosis at the acute stage. Often times, there will be a relative decrease in the number of platelets in the blood, in addition to reduced white blood cells and anemia (Harrus et al 2012). It has been noted by Shipov et al that left without the proper treatment or no treatment at canines will eventually progress to the subclinical stage. At this stage the animal rarely exhibits any symptoms or evident clinical signs besides the hematological abnormality of a reduced platelet count (2008). Many canines can remain in the subclinical stage for their entire lives. Often times they act as carriers, recover from the illness altogether, or move on to the more sever chronic stage (Shipov et al 2008). The progression of the disease to the chronic stage is still under investigation (Harrus et al 2010). However, the chronic stage can be diagnosed as having symptoms similar to the acute stage, yet far more severe (Harrus et.al 2010). The additional symptoms reported in canines suffering from the chronic phase also include pale mucous membranes, muscle weakness, profuse bleeding, and weight loss (Harrus et.al 2011).
In Humans
In humans, typically within one or two weeks after receiving a bite from an infected tick, the infected individual will experience those symptoms associated with the acute stage of the illness. Symptoms for HME include weakness, low-back pain, gastrointestinal symptoms, rash and almost always are characterized by a mild to high fever (Ganguly et. al 2008). Due to the nature of the infection Doctors suggest that treatment should begin within 3 to 4 days after the first outbreak of symptoms (Ganguly et al 2008). As a multisytemic disease other forms of infection have been reported to occur namely cough, pharyngitis, lymphadenopathy, diarrhea, vomiting, and at times shifts in the mental status of the individual (Ganguly et al 2008). Ehrlichiosis does not often cause fatalities in the human or canine population. However, when fatalities do arise from ehrlichial infection they occur once the patient has entered into the chronic stage of this illness (Paddock et.al 2003). Interestingly, these fatalities are usually seen in individuals with severely compromised immune systems, like HIV (Paddock et. al 2003).
In Canines
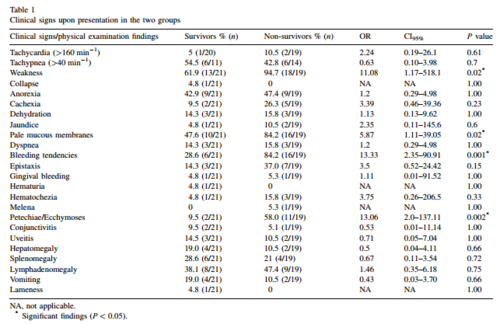
Blood counts and hematological tests are crucial to the diagnosis of Canine monocytic ehrlichiosis. Low blood counts or thrombocytopenia are often critical signs of ehrlichial infection. In a study conducted by Shipov et. al red blood cell count and hemoglobin were used as the diagnostic indicators for canine ehrlichiosis (2008). Within the study dogs were assigned to two categories: Survival(s)and No survival(NS. Within each group the prognostic indicators and treatment protocols were observed and compared. Most commonly the dogs under examination exhibited symptoms such as weakness pale mucous membranes, fever, and bleeding tendencies (Figure 5). Other clinical findings included anemia, leucopenia, and thrombocytopenia (low blood cell count) (Shipov et. al 2008). From these indicators it was determined that the NS group often exhibited a wider range of symptoms at a greater degree of severity (fig. 4) The acute stage was diagnosed in those dogs that showed symptoms including high fever, depression, lethargy, anorexia. During the treatment phase of this study it was noted that the three phases of the disease had differential responses to the therapy and can carry differential prognosis ( Shipov et al 2008). Canines who have entered into the chronic stage of the illness usually have little hope for survival as the treatment regime is extremely extensive and costly (Shipov et al 2008).
Treatment for both humans infected with E.chaffeensis and canines infected with E. canis is relatively similar. For most ehrlichial infections the antibiotic Doxycycline is considered the best method of treatment (Nicholson et. al 2010). It is crucial however that treatment is started right away in order to ensure that the illness does not move past the acute stage. Today, the only clear method of prevention is the education of the public- making individuals aware of the transmission and symptoms of the infection (Nicholson et. al 2010). Individuals living in areas with high density tick populations have been advised to wear light clothes while outside and perform frequent tick checks (Ismail et al 2010). However, it has been predicted that tick-borne pathogens like ehrlichiois will continue to spread at an even more rapid rate than previously seen in coming years (Nicholson et. al 2010).
References
1. Ismail, Nahed, Karen C. Bloch, and Jere W. McBride. "Human Ehrlichiosis and Anaplasmosis." Clinics in Laboratory Medicine 30.1 (2010): 261-92. Print. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2882064/
2. Ganguly,S, Mukhopadhayay, S.K. “Tick-borne Ehrlichiosis infection in human beings”. Vector Borne Diseases 45. (2008): 273-280. http://www.mrcindia.org/journal/issues/454273.pdf
3. Paddock, C., Childs, E. “Ehrlichia chaffeensis: a Prototypical Emerging Pathogen.” Clinical Microbiology Reviews Jan. 2003, p. 37–64 http://cmr.asm.org/content/16/1/37.full.pdf+html
4. Shipov, A. Klement, E., Reuveni- Tagar, L., Harrus. S. “Prognostic indicators for canine monocytic ehrlichiosis.” Veterinary parasitology 153. (2008): 131-138 http://www.sciencedirect.com/science/article/pii/S0304401708000319
5.Nicholson, W., Allen, K., McQuiston, E. “The increasing recognition of rickettsial pathogens in dogs and people.” Trends in Parasitology. 2010 http://www.sciencedirect.com/science/article/pii/S147149221000019X
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2013, Kenyon College.
