Enterovirus 71

Etiology/Bacteriology
Taxonomy
ssRNA positive-strand viruses, no DNA stage
| Order = Picornavirales
| Family = Picornaviridae
| Genus = Enterovirus
| species = Enterovirus A
{|NCBI: Taxonomy Genome: Genome|}
Description
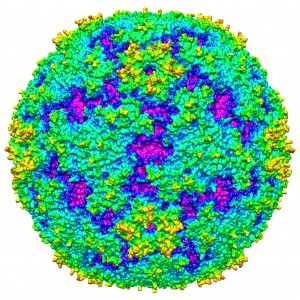
Enterovirus 71 is a single stranded RNA positive strand virus from the Picornaviridae family. Enteroviruses are divided into two classes, polioviruses and nonpolioviruses. Enterovirus 71 is considered a nonpoliovirus. Enterovirus 71 (EV71) is one of the common causes of hand-foot-and-mouth disease (HFMD). Although HFMD is the most common diagnosis from an EV71 infection, more severe complications can occur due to the infection, such as encephalitis, aseptic meningitis, and acute flaccid paralysis. This disease is transmitted via the fecal-oral route and can remain in fecal matter for up to 11 weeks. Colonization occurs in the intestinal tract and then can spread into the bloodstream and into organ systems. EV71 epidemics have occurred in the Asia-Pacific region with few outbreaks in Europe. Although complications can occur from this infection, the mortality rate has only reached 14% in the largest outbreak to date.
Pathogenesis
Transmission
Transmission of EV71 is through direct contact of discharge from the nose, throat, saliva, and blisters or stools of infected persons. The infection is most contagious during the first week of the illness[1], but fecal matter can continue to contain viral particles for up to 11 weeks.[2]
Infectious dose, incubation, and colonization
After direct contact with EV71, the virus must be able to replicate itself to reach an infectious dose. In this case, the TCID50 of EV71 is 9 log 7.[3] The incubation period is 3 to 10 days. EV71 can remain viable at room temperature for several days and can survive the acidity of the GI tract. Colonization is located in the GI tract or the respiratory tract. Initial replication begins in the intestinal tract. The virus then spreads into the bloodstream by the third day of infection. The virus then spreads to organ systems, causing a second viral infection episode on days 3-7. The infection can further progress to the CNS in more severe infections.[4]
Epidemiology
The first isolation of enterovirus 71 was in fecal matter of a baby with encephalitis in California in 1969. In 1973, EV71 was first recognized as causing epidemics of HFMD in Japan. Also in the 1970s, two large epidemics of EV71 occurred in Europe which were recognized from the virus causing HFMD or other symptoms. The first outbreak in Europe was in Bulgaria. EV71 was the causative agent in 77% of the children with non-specific febrile illness and neurological disease. Of the 451 children with the disease, approximately 10% died. In the second epidemic in Hungary, symptoms were aseptic meningitis and encephalitis. Few patients had HFMD. 3% of the 1550 cases of the disease died. Small sporadic outbreaks continued to break out in the 1980s in Hong Kong and Australia. By the late 1990s, large outbreaks occurred in the Asia-Pacific region. The death rate was less than 1 percent of the patients. The largest outbreak occurred in 1998 in Taiwan. This outbreak had 1.5 million infections, 405 severe cases, and 78 deaths. The latest outbreak was in China in 2008, when 490,000 infections were reported with 126 deaths. In many areas in the Asia-Pacific region, cyclical epidemics have occurred every 2 to 3 years. Fatality rates from these outbreaks have been as high as 14%, with the majority of these having complications such as encephalitis. Thus far, outbreaks of EV71 arise at a low level in the Africa, Europe, and the US. [5]
Virulence Factors
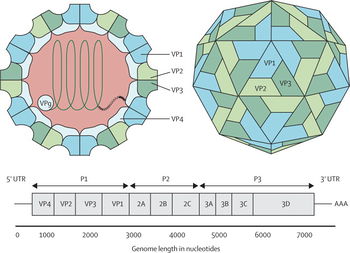
The virulence factors of EV71 are currently unknown. Attention to the capsid protein VP1 has been given for possible contributions to virulence. The residue 145 of the viral capsid protein VP1 determines whether the virus binds to the viral receptor, PSGL-1 in lymphocytes. Another viral receptor that is specific to EV71 is the scavenger receptor B2, which is also in lymphocytes. The residue VP1-145 controls the binding of the virus to the viral receptor, PSGL-1 (P-selectin glycoprotein ligand-1). In experiments with mice, it has been determined that major mutations in VP1 capsid protein can cause virulent EV71 strains.[6]
Clinical Features
Symptoms
EV71 infections can cause a wide range of symptoms and is most common in children under the age of 5. The most common feature is hand-foot-and-mouth disease (HFMD). HFMD usually occurs in children and is generally mild. It consists of a papulovesicular rash on the palms and soles as well as multiple oral ulcers (also known as herpangina). In children under the age of two, atypical rashes are frequently seen. Other symptoms that can occur from EV71 are upper respiratory infections, fever, gastroenteritis, bronchiolitis, and pneumonia. In a Tawainese outbreak of EV71 infections, many adults remained symptom free, however, more than 20% of them developed an upper respiratory infection.
EV71 infections can also cause neurological and systemic complications such as aseptic meningitis, acute flaccid paralysis, and encephalitis (usually in the brainstem). These symptoms are usually successive to or in conjunction with HFMD. Only a small portion of infected children develop these complications, which can be severe and even fatal. Children can develop these symptoms in a matter of hours to days. [2]
Diagnosis
For diagnosis of EV71 infections, many different tests can be performed, depending on the manifestations of the disease. The most efficient approach for diagnosis is throat swabs and swabs from at least two vesicles or from the rectum for patients with no vesicles. Other approaches for diagnosis is the use of EV71-specific primers to perform PCR directly on samples. This allows for faster diagnosis than virus cultures, but this only tests for EV71. DNA microarray is a tool used as well, and it has been reported to be 90% accurate in diagnosis of EV71 infections, however it is an expensive tool. In cases that have CNS involvement, lumbar punctures are essential for diagnosis.[2]
Treatment
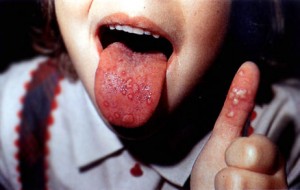
For mild infections of EV71 which typically include hand-foot-and-mouth disease, there is no particular treatment. Symptoms typically clear up within seven to 10 days from the day of infection. To relieve pain from mouth sores that develop, an oral anesthetic can be used. The treatment of the general pain associated with the disease can be treated with over the counter drugs such as ibuprofen and acetaminophen.[7]
Antiviral agents and intravenous immunoglobulin have both been used as an attempt to treat the EV71. The antiviral drug, Pleconaril, has been used in clinical trials of aseptic meningitis, however it’s not effective against EV71. Other capsid-function inhibitors have been studied, some of which have promising activities against EV71. Intravenous immunoglobulin has been used in the large outbreaks of EV71 in Asia. It was thought to neutralize the virus and have non-specific anti-inflammatory properties. When comparing those who received intravenous immunoglobulin to those who did not, recipients seemed to benefit from the treatment if it was given during early onset of the infection.[2]
Prevention
To prevent EV71 outbreaks, good hygiene and improved sanitation are important. Since transmission is through the fecal-oral route, it is important to frequently wash one’s hands, disinfect contaminated surfaces, and wash soiled clothing. EV71 is resistant to many cleaning products, thus it is important to use disinfectants that are chlorinated or iodized.[1]
A vaccine for EV71 is currently under research. Two research groups has made the inactivated alum-adjuvent EV71 vaccine, but it is currently in a phase III trial. This vaccine has thus far had high efficacy and sustained immunogenicity. Virus-like particle (VLP) vaccines have also been studied. The studies have shown that a VLP with yeast is a potential vaccine for EV71.[6]
Host Immune Response
The host immune response is similar to that of the polio virus, however there have been limited studies on the enterovirus 71 host immune response. Studies suggest the cell-mediated immune response plays a significant role in immunity against EV71. Induction of type I interferons is the earliest non-specific response to viral infections and is an innate defense mechanism to control EV71 infections, but these decreased later in the infection. [8] Studies have shown that there were increases in mRNAs encoding chemokines, proteins associated with protein degradation, complement proteins, and proapoptosis proteins. cDNA microarray expression revealed cells down regulated in several genes associated with host RNA synthesis.[9] Antibody production usually occurs within the first 7-10 days in the lymphatic system of the GI tract before reaching the CNS. This prevents the spread of the infection to further complications; however, complications do occur. [4]
References
[1] Information Sheet: Hand-Foot-Mouth Disease related to Enterovirus 71. Public Health Agency of Canada. http://www.phac-aspc.gc.ca/id-mi/ev71-eng.php
[2]Ooi,Mung How, et al. Clinical features, diagnosis, and management of enterovirus 71. Lancelot Neurol. 2010 Nov;9(11):1097-105. doi: 10.1016/S1474-4422(10)70209-X. http://www.ncbi.nlm.nih.gov/pubmed/20965438
[3]Liu CC1, Lian WC, Butler M, Wu SC. High immunogenic enterovirus 71 strain and its production using serum-free microcarrier Vero cell culture. Vaccine. 2007 Jan 2;25(1):19-24. Epub 2006 Aug 2. http://www.ncbi.nlm.nih.gov/pubmed/16919374
[4]Bennet, Nicholas John, et al. Pediatric Enteroviral Infections. Medscape. http://emedicine.medscape.com/article/963637-overview#a0104
[5]Solomon, T., et al. Virology, epidemiology, pathogenesis, and control of enterovirus 71.Lancelot Infectious Diseases.2010 Nov;10(11):778-90. doi: 10.1016/S1473-3099(10)70194-8. Epub 2010 Oct 18. http://www.ncbi.nlm.nih.gov/pubmed/20961813
[6]Wang SM1, Liu CC. Update of enterovirus 71 infection: epidemiology, pathogenesis and vaccine.Expert Rev Anti Infect There.2014 Apr;12(4):447-56. doi: 10.1586/14787210.2014.895666. Epub 2014 Mar 3. http://www.ncbi.nlm.nih.gov/pubmed/24579906
[7]Hand-foot-and-mouth disease. Mayo Clinic. http://www.mayoclinic.org/diseases-conditions/hand-foot-and-mouth-disease/basics/treatment/con-20032747
[8]Liu, ML, et al. Type I interferons protect mice against enterovirus 71 infection. J Gen Virol. 2005 Dec;86(Pt 12):3263-9. http://www.ncbi.nlm.nih.gov/pubmed/16298971
[9]Shih SR1, Stollar V, Lin JY, Chang SC, Chen GW, Li ML. Identification of genes involved in the host response to enterovirus 71 infection. J Neurovirol. 2004 Oct;10(5):293-304. http://www.ncbi.nlm.nih.gov/pubmed/15385252
Created by Regina Swenton, a student of Tyrrell Conway at the University of Oklahoma.
