Hopanoid lipid
By Haofan Li
Introduction
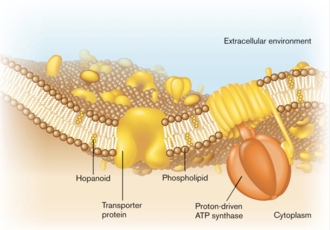
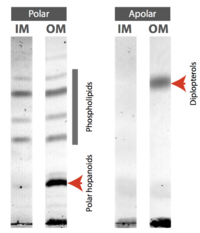
As major components of the selectively permeable bacterial membranes, lipids, though with relatively monotonic structures compared to membrane proteins, play significant roles in biochemical activities and stress tolerance.[3] Hopanoid lipids are well-studied modern lipid models. They are widely found in a large scale of organisms, such as bacteria, plants, and some lichens, but no hopanoid lipid is found in archaea. Among bacteria, both Gram-negative and Gram-positive bacteria contain hopanoid lipids, which indicates the critical roles they play in bacterial growth and reproduction (Figure 1).[4] Hopanoid lipids are commonly localized in the bacterial outer membranes. Methylobacterium extorquens, one Gram-negative bacterium, mostly produces three types of hopanoid lipids, which include diplopterols (Figure 3), 2-methyl-diplopterols, and bacteriohopanepolyols (polar hopanoid lipids).[5] According to the TLC plates, all those hopanoid lipids are only found on the bacterial outer membranes (Figure 2).[2]
Hopanoid lipids are pentacyclic lipids, each of which consists of four six-carbon rings and one five-carbon ring at one end (Figure 4). With the similar structure as the four-ring eukaryotic sterols, hopanoid lipids connect rings via sharing one carbon-carbon single bond between two neighboring ring structures, eventually forming flat, hydrophobic, and stable chemical structures.[6] Hopanoid lipids also contain different hydrophobic and hydrophilic side chains, like R groups in amino acids. As a result, the various side chains increase diversity and expand functions of hopanoid lipids.[7]
The hopanoid lipids play crucial roles in the microbiome and the microbial interaction with animals and plants. From previous studies, the hopanoid lipids, as one of the essential components in biomembranes, contributes substantially to bacterial membrane permeability and fluidity, stress resistance, nitrogen fixation and bacterial associations with plants, etc.[7] The study of hopanoid lipid, as one of the new and frontier research, remains lots of unknown and interesting topics to study. Although we understand many broad overviews of hopanoid lipids, the detailed mechanisms remain uncertain. Overall, future studies are needed to unveil more interesting facts about hopanoid lipids.[3]
Hopanoid biosynthesis
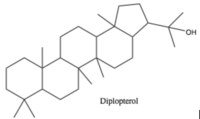
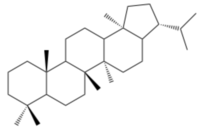
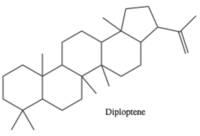
Hopanoid lipids, with a basic hopane skeleton structure (Figure 4), contain four cyclohexanes and one cyclopentane, all of which connecting each other by sharing one carbon-carbon single bond. Thus, those carbons rings, with all-chair conformations, form planar, hydrophobic, and stable structures with thirty carbon atoms.[7] Based on the basic skeleton structure, the hopanoid lipids are further modified by the methylation of the squalene precursor. The most common simple hopanoid compounds in the bacterial membranes are diplopterols (Figure 3) and diploptenes (Figure 5). Among 40 widely-found elongated hopanoids in the bacterial membranes, the most common hopanoids are aminobacterialhexanetriols and bacteriohopanetetrols.[3][7]
Around ten percents of bacteria can synthesize hopanoid lipids, such as Cyanobacteria and Bacilli.[3] The biosynthesis of hopanoid lipids is generally divided into five different stages. Stage one begins the synthesis of hopanoids through either the mevalonate pathway or the mevalonate-independent pathway. In the mevalonate pathway, bacteria utilize acetyl-CoA, which is produced during bacterial respiration, to synthesize the dimethylallyl pyrophosphate and isopentenyl pyrophosphate.[9] Both products from this mevalonate pathway are used as five-carbon building blocks. The mevalonate-independent pathway utilizes the glyceraldehyde-3-phosphate and two carbon compounds to produce dimethylallyl pyrophosphate through a series of complicated reactions, such as pyruvate decarboxylation, phosphorylation, and dehydration. The mevalonate-independent pathway, a biosynthesis pathway commonly found in plant plastid organelles, is also found in some wide-known pathogens, such as Mycobacterium tuberculosis.[10] This alternative pathway indicates a potential antibiotic target for bacterial inhibition. Fosmidomycin, isolated from the secondary metabolism of Streptomyces, a genus of Gram-positive soil bacteria, inhibits the activity of 1-deoxy-D-xylulose 5-phosphate (DXP) reductoisomerase, one essential enzyme utilized in the mevalonate-independent pathway. Additionally, this enzyme is not produced by the human body, and thus this potential new antibiotic candidate might destroy one of the crucial bacterial enzymes without negatively influencing the human body. [7][11][12]
The mechanisms of both stage two and three are similar to the sterols synthesis. In stage two, bacteria, with the assist of isopentenyl pyrophosphate, connect two units of dimethylallyl pyrophosphate via head-to-tail condensation, forming one unit of farnesyl diphosphate. In stage three, two units of farnesyl diphosphate from stage two are reductively connected through head-to-head condensation with the assist of synthesizing squalene. Most of the final products of stage three show trans conformations with high stability.[7]
In stage four, the squalene proceeds the cyclization reaction to produce hopene. The organic reaction happening in stage four is one of the most complicated one-step organic biosyntheses. The squalene first folds into a proper orientation, therefore increasing the kinetics of this reaction and proceeding the polycyclic formation. This cyclization reaction includes the modification of nine stereocenters, the connection changes of thirteen covalent bonds, and the establishment of four cyclohexanes and one cyclopentane. In stage five, bacteria modify the products from stage four through side chain formation and core backbone structure modification. In the side chain formation, various enzymes, especially those coded by the hpn genes, further diversify the structures of hopanoid lipids through adding different types of side chains. Additionally, the backbone structure is modified via the methylation of the core structure. A few bacteria also conduct the desaturation of the five rings. However, less biochemical mechanisms are known for the stage five, and the details of this process need further studies.[3][7]
Bacterial membrane permeability and fluidity
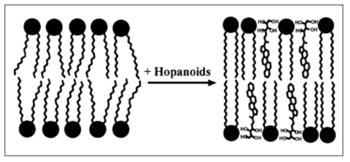
The hopanoid lipids, which have a major hydrophobic five-ring structure, are mostly hydrophobic and relatively stable. Thus, the majority of hopanoid lipids can travel and intercalate into the hydrophobic layer of double membranes and interact with the tails of hydrophobic lipids. In other words, the hydrophobic hopanoid lipids insert between the non-polar fatty acids, decreasing the extra space between the lipid hydrophobic tails. Therefore, due to the decreased movements of the lipid tails, hopanoid lipids increase the membrane thickness and decrease its fluidity (Figure 6).[7] Researchers used three different strains, which included the wild-type Methylobacterium extorquens (WT), hopanoid-depleted wild-type strain (WT-depleted), hopanoid-deficient shc mutation strain (ΔSHC).
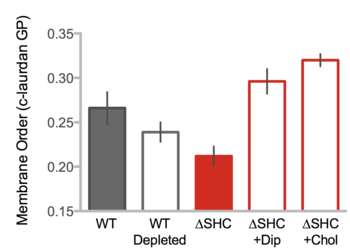
Researchers also added diplopterols and cholesterols into the growth media of shc mutant. The results showed that the wild-type strain and the mutant with the treatments of diplopterols and cholesterols showed higher ordered membranes compared to the membranes of the hopanoid-deleted wild-type and hopanoid-deficient mutation strains. Therefore, the hopanoid lipids increase the membrane order (Figure 7).[2]
Different side chains of hopanoid lipids might have different influences on the bacterial membrane properties. For example, 3-methyl-BHTs, with large hydrophilic side chains, experiences difficulties in inserting into the bacterial membranes. However, diploptenes with hydrophobic side chains relatively freely move between the lipid tails. Therefore, hopanoid lipids with unique functional groups show different distributions inside the bacterial membranes, the way that hopanoid lipids interact with environments, and their interactions with other hopanoid lipids. In the membrane dynamic simulation, the amphiphilic hopanoid lipids, with hydrophobic skeletons and hydrophilic side chains, show an upright orientation, which is similar to that of phospholipids (Figure 6). Due to the unique orientation of amphiphilic hopanoid lipids, they can interact with other lipid layers, thus condensing the overall structure and increasing the integration of the bacterial membranes. On the other hand, some hydrophobic hopanoid lipids with hydrophobic side chains show strong abilitys to transfer into the nonpolar part of the biomembranes, which increases the membrane thickness by limiting the movement of the hydrophobic tails.[3]
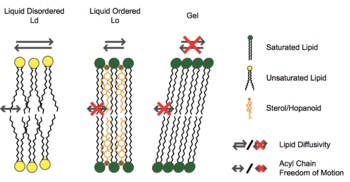
The functions of steroid alcohols in eukaryotes and hopanoid lipids in prokaryotes show similarities. Both of them contributes to the membrane stability, but they might contribute differently. In dynamic lipid simulation, researchers used cholesterols, one kind of steroid alcohols, and diplopterols, one type of hopanoid lipids, to analyze their influences on membranes. Both cholesterols and diplopterols prohibited the formation of gel phase during membrane simulation, and they also ordered the orientation of N-stearoyl-D-erythro-sphingosylphosphorylcholine, one artificial sphingolipid (Figure 8).

[14] However, the cholesterols induced the immiscible formations of both liquid disordered and ordered configuration, while the diplopterols showed a separation of those two lipid formations. Thus, those two different configurations of lipids indicated the different functions of steroid alcohols and hopanoid lipids in the cell membrane. (Figure 9).[14]
The roles of either steroid alcohols or hopanoid lipids might be more different in vivo. In the artificial membranes, membranes with 2-methyl-diplopterols, one type of methylated hopanoid lipids, show higher thickness compared to membranes with cholesterols. Thus, the addition of 2-methyl-diplopterols in biomembrane might decrease the fluidity and increase the stability of membranes. However, the membrane thickness of Rhodopseudomonas palustris hpnP mutation strain, which has less production of 2-methyl-diplopterols, shows no significant differences compared with that of the wild-type strain. Therefore, the in vivo conditions are more complex compared to the lipid dynamic simulation.[14]
Stress tolerance
The abundance and diversity of hopanoid lipids positively influence bacterial resistance to environmental stress, such as the extreme pH, high pressure, non-optical temperature, and high concentration of antibiotic or other lethal chemical compounds.[3] With the help of hopanoid lipids, microbiomes develop different strategies to increase the fitness in non-optical environments. However, most of the detailed mechanisms of stress tolerance induced by the hopanoid lipids remain unclear and require further studies.[3]
Methylation of hopanoid lipids
Researchers found that under extreme conditions, the biosynthesis of hopanoid lipids increases in most bacterial strains.[3] For example, When Rhodopseudomonas palustris is placed under extreme environments, such as high temperature or low pH conditions, those stress induces the activation of the ecfG gene, which is a general stress regulator related gene for the alphaproteobacteria. Due to the expression of ecfG , the response factors and regulators upregulate the expression of hpnP, a gene that codes for hopanoid methylases. Therefore, the expression of hpnP increases the rate of hopanoid lipids methylation in bacteria. Methylation increases the hydrophobic characteristics of hopanoid lipids and further decreases the movements of phospholipids hydrophobic tails, perhaps increasing the bacterial membrane resistance to potential dangers.[16]
Membrane fluidity and permeability
The presence of hopanoid lipids decreases the fluidity and permeability of bacterial membranes. The hydrophobic part of the hopanoid lipids migrates into the bacterial membrane and moves between the phospholipid tails. Thus, hopanoid lipids fill the empty spaces and increase the membrane integrity. The hydrophilic side chains also form strong attractions with other molecules and environments. Overall, the biomembrane becomes more stable, and the leakage of lethal molecules decrease. Under the low pH or high antibiotic conditions, the protons and antibiotic compounds might experience difficulty across the bacterial membranes due to decreased membrane permeability. Therefore, bacteria show higher resistance towards the high concentration of protons, antibiotics, and other lethal molecules.[3]

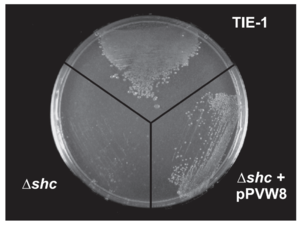
Different hopanoid lipids show different influences on membrane fluidity and permeability. Bile salts show no negative influence on the majority of the Gram-negative bacteria because these chemical compounds are not able to transfer across the outer membrane of the Gram-negative bacteria. Mutated bacteria, without the protection of hopanoid lipids, are vulnerable to the high bile salt conditions.[15] However, different mutations in the same bacteria species show different levels of bile salts sensitivities. shc gene codes for the production of most hopanoid lipids. The Rhodopseudomonas palustris with this mutation shows no bile salts tolerance (Figure 10).[15] The hpnH mutation causes the deficient production of diploptenes and diplopterols. Rhodopseudomonas palustris with this mutation shows resistance towards the bile salts but perform a slower growth rate compared to that of the wild-type strain. The hpnO mutant cannot produce aminobacteriohopanetriols, one essential bacterial hopanoid lipid. This mutant shows no difference in bacterial growth and culture density in the high bile salts concentration compared to the wild-type strain.[3][15]
Orientation and distribution of hopanoid lipids
The orientation and the distribution of hopanoid lipids contribute to the bacterial heat resistance. Bacteria, with the help of high concentration of hopanoid lipids, obtain heat protections and keep the homeostasis of their cytoplasm. As the environmental temperature increases, the hopanoid lipids tend to move between two leaflets. In the lipid dynamic simulation at room temperature (298 K), most of the diplopterol molecules gather between the head and the tail of phospholipids. Thus, at milder temperature, the distribution of hopanoid lipids shows a large separation. However, as the temperature increases, the diplopterol molecules migrate to the empty spaces between the tails of two leaflets, largely increasing the thickness of the bacterial membranes and creating substantial protections for thermophiles (Figure 11).[17]
Cooperation with bacterial membrane proteins
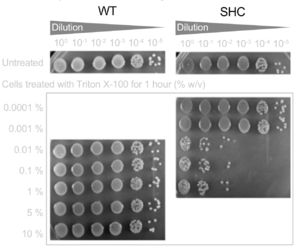
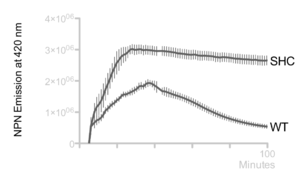
Hopanoid lipids might cooperate with outer membrane proteins.[3] The Methylobacterium extorquens mutant strain, with a non-functional hopanoid lipid production, shows an increased detergent sensitivity (Figure 12). Additionally, this strain perhaps cannot pump out the antibiotics due to its non-functional efflux. Researchers studied the accumulation of 1-N-phenylnaphthylamine (NPN), one type of lipophilic dye, in Methylobacterium extorquens. The wild-type strain constantly pumped out NPN, and therefore the intracellular concentration of PNP kept decreasing. However, the intracellular concentration of shc mutant did not change a lot, indicating that perhaps the mutant had deficient efflux (Figure 13).[2] Thus, the hopanoid lipids might contribute and assist the functions of membrane proteins.[3] The collaboration of the hopanoid lipids and membrane proteins enhances bacterial resistance and helps bacteria to maintain the homeostasis between the cytoplasm and extracellular environment. Additionally, bacterial membranes, one of the most significant energy production places, require the presence of effective hopanoid lipids to achieve energy production and storage. Nostoc punctiforme, with a hopanoid lipid mutation, show a decrease in energy storage compared to that of the wild-type strain.[3]
The function of hopanoids is more significant in the stress-resistance specified cells. The filaments of Nostoc punctiforme, one kind of filamentous cyanobacteria, are constituted by the vegetative cells. That specified cells conduct photosynthesis under the environment with abundant nutrients. Therefore, they fix carbon dioxide, showing a high growth and reproduction rate. In these photosynthetic cells, the hopanoid concentration has negligible influence on the stress response. However, under the environment with poor nutrient concentration, Nostoc punctiforme develop the akinete cells through differentiation, which contain higher resistance toward the cold and dry conditions. The akinete cells keep their cellular structure and bacterial activity. They also stay in low energy consumption until they encounter an environment with rich resources. In the akinete cells, a high concentration of hopanoid lipids is detected in bacterial membranes. Previous studies found that the mutant without functional hopanoid lipid showed less resistance to the extracellular resistance. Similarly, Streptomyces coelicolor, one Gram-positive soil bacteria species, forms vegetative cells under nutrient-rich environment. These vegetative cells have a low hopanoid lipid concentration. However, at the end of their life cycle, they produce and accumulate hopanoid lipids, forming spore via sporulation, which can assist their resistance under the extreme conditions.[3]
Nitrogen fixation
Various bacteria, with the ability to produce hopanoids, can fix nitrogen. Bacteria, such as Beijerinckia, Frankia, Anabaena, Burkholderia, etc., show a positive correlation between the hopanoid production and nitrogen fixation.[3] Most nitrogen-fixing bacteria live in close physical correlations with plants, such as alfalfa, soil beans, peas, etc. Both bacteria and plants can take advantage of this relationship. Bradyrhizobium spp. derive energy and other important chemical cofactors from their host plants. The plants also can protect bacteria from stress from the environments, such as competitions with other microbiomes, high proton concentration, etc. Equally, plants benefit from the nitrogen oxides produced by the nitrogen-fixing bacteria, and therefore plants show an enhanced growth rate and outcompete their competitors. There are also free-living nitrogen-fixing microbes, such as Anabaena spp., Frankia spp., etc. However, most of those bacteria do not fix nitrogen without coexisting with plants.
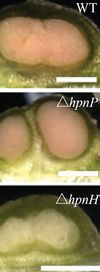
The legume-rhizobia root nodule symbiosis is among one of the most widely studied plants and bacteria associations. The production of hopanoid lipids influences the bacterial symbiosis with the host plants. The Bradyrhizobium spp., with shc mutation, produce a negligible amount of hopanoid lipids. Therefore, this bacteria strain is incapable of integrating with plants. Additionally, different types of hopanoid lipid contribute differently to the bacterial associations with plants. The Bradyrhizobium diazoefficiens with a hpnP mutation cannot produce the extended hopanoids. Their association with the Aeschynomene afraspera results in morphologically disorganized nodules. The Aeschynomene afraspera shows nitrogen starvation, indicating a decreased nitrogen-fixing ability of the hpnP mutant. However, the hpnH mutant, which have less production of 35 carbon hopanoids, cannot form a mature nodule (Figure 14).[18].
The presence of the hopanoid lipids might also contribute to the competition between bacteria to inhabit the hosts. The mutation strain shc, which produces no hopanoid lipids shows an inability in long-term colonization of a plant. Previous studies found that in the beginning, shc mutant grew and developed in host normally. However, after a few days, it started to degenerate and be recycled by its host. On the other hand, the high concentration of hopanoid lipids increases bacterial stress tolerances. For example, the Bradyrhizobium and Burkholderia, both of which contain a high concentration of hopanoid lipids, show resistance to high temperature and low pH conditions. Therefore, in the consideration of global warming and increasing soil acidity, both bacteria might outcompete other bacteria in soil and plants.
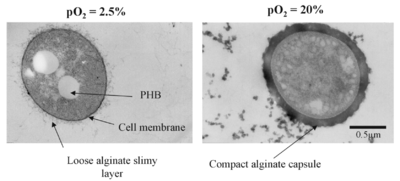
Most of the time, bacteria conduct nitrogen fixation in the low-oxygen concentration environment. Nitrogenase, the core enzyme in nitrogen fixation, is vulnerable in the presence of oxygen. Thus, most of the nitrogen-fixing bacteria exist under the anaerobic environment. Sometimes, nitrogen fixation can also be conducted at high oxygen concentration. Under this condition, bacteria need to develop more sophisticated mechanisms to protect the nitrogenase and achieve nitrogen fixation. The free-living nitrogen-fixing bacteria, Azotobacter vinelandii, develop various ways to fix nitrogen under high oxygen condition. For example, the Azotobacter vinelandii decrease its cellular surface area. Therefore, for each unit of cytoplasm, there are larger protections from the bacterial membranes. Also, under high oxygen condition, it also increased its oxygen consumption via keeping a high respiration rate, thus creating an optional anaerobic environment for nitrogen fixation. The Azotobacter vinelandii also synthesizes alginate to create an alginate capsule, a thick barrier which limits the oxygen fission and protects the nitrogenase from the high oxygen-concentrated surroundings. Under the lower oxygen concentrated environment, Azotobacter vinelandii develops a loose alginate capsule, while under the high concentration, it builds up a compact and thick alginate capsule (Figure 15). Additionally, when the bacteria interact with or inhabit plants, the host plants synthesize leghemoglobin molecules, one type of oxygen-transport metalloproteins. This hemoglobin scavenges oxygen from the root nodules of the leguminous plants, creating an optimal environment for nitrogenase activity[19].
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.
- ↑ Slonczewski, J., and Foster J. W.. Microbiology: An Evolving Science. New York
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Saenz J. P., Grosser D., Bradley A. S., Lagny T. J., Lavrynenko O., Broda M., and Simons K.. 2015. Hopanoids as functional analogues of cholesterol in bacterial membranes. PNAS. Sep. 22, 2015, vol. 112, no. 38, 11971-11976
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 Belin B. J., Busset N., Giraud E., Molinaro A., Silipo A., and Newman D. K.. 2018. Hopanoid lipids: from membranes to plant-bacteria interactions. Nature Reviews, Microbiology 2018.
- ↑ Ourisson G. and Rohmer M. 1992. Hopanoids. 2. Bio-hopanoids: A Novel Class of Bacterial Lipids. Acc. Chem. Res. 1992, 25, 403-408
- ↑ 5.0 5.1 Diploptene, Sigma Aldrich, Accessed May 11th, 2018
- ↑ Saenz J. P., Grosser D., Bradley A. S., Lagny T. J., Lavrynenko O., Broda M., and Simons K.. 2015. Hopanoids as functional analogues of cholesterol in bacterial membranes. PNAS, September 22, 2015, vol. 112, no, 38, 11971-11976
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 Kannenberg E. L., and Poralla K.. 1999. Hopanoid Biosynthesis and Function in Bacteria. Naturwissenschaften 86, 168–176 (1999). Springer-Verlag 1999.
- ↑ Hopane Reference Standards for use as Petrochemical Biomarkers in Oil Field Remediation and Spill Analysis, Amann N., Sigma Aldrich, Accessed May 11th, 2018
- ↑ Betts J. C., Lukey P. T., Robb L. C., McAdam R. A., and Duncan K.. 2002. Evaluation of a nutrient starvation model of Mycobacterium tuberculosis persistence by gene and protein expression profiling. Molecular Microbiology (2002) 43(3), 717–731.
- ↑ Lichtenthaler H. K. 1999. The 1-deoxy-D-xylulose-5-phosphate pathway of isoprenoid biosynthesis in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999. 50:47–65.
- ↑ Rohmer M. 1999. The discovery of a mevalonate-independent pathway for isoprenoid biosynthesis in bacteria, algae, and higher plants. Nat. Prod. Rep., 1999, 16, 565–574.
- ↑ Hale I. O'Neill P. M., Berry N. G., Odom A., and Sharma R.. 2012. The MEP pathway and the development of inhibitors as potential anti-infective agents. Med. Chem. Commun., 2012, 3, 418–433.
- ↑ Diploptene, Sigma Aldrich, Accessed May 11th, 2018
- ↑ 14.0 14.1 14.2 14.3 14.4 Saenz J. P., Sezgin E., Schwille P., and Simons K.. 2012. Functional convergence of hopanoids and sterols in membrane ordering. PNAS. August 28, 2012, vol. 109, no. 35, 14236-14240.
- ↑ 15.0 15.1 15.2 15.3 Welander P. V., Hunter R. C., Zhang L., Sessions A. L., Summons R. E., and Newman D. K.. 2009. Hopanoids Play a Role in Membrane Integrity and pH Homeostasis in Rhodopseudomonas palustris TIE-1. JOURNAL OF BACTERIOLOGY, Oct. 2009, Vol. 191, No. 19, p. 6145–6156.
- ↑ Welander P. V., Doughty D. M., Wu C. H., Mehay S., Summons R. E., and Newman D. K.. 2011. Identification and characterization of Rhodopseudomonas palustris TIE-1 hopanoid biosynthesis mutants. Geobiology (2012), 10, 163–177.
- ↑ 17.0 17.1 Caron B., Mark A. E., and Poger D.. 2014. Some Like It Hot: The Effect of Sterols and Hopanoids on Lipid Ordering at High Temperature. J. Phys. Chem. Lett. 2014, 5, 3953−3957.
- ↑ 18.0 18.1 18.2 Kulkarni G., Busset N., Molinaro A., Gargani D., Chaintreuil C., Silipo A., Giraud E., and Newman D. K.. 2015. Specific Hopanoid Classes Differentially Affect Free-Living and Symbiotic States of Bradyrhizobium diazoefficiens. mBio 6(5):e01251-15. doi:10.1128/mBio.01251-15.
- ↑ Sabra W., Zeng A. P., Lunsdorf H., and Deckwer W. R.. 2000. Effect of Oxygen on Formation and Structure of Azotobacter vinelandii Alginate and Its Role in Protecting Nitrogenase. APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Sept. 2000, Vol. 66, No. 9, p. 4037–4044
