Idiomarina loihiensis
A Microbial Biorealm page on the genus Idiomarina loihiensis
Classification
Higher order taxa
Bacteria; Proteobacteria; Gammaproteobacteria; Alteromonadales; Idiomarinaceae; Idiomarina
Species
|
NCBI: Taxonomy |
Idiomarina loihiensis
Description and significance
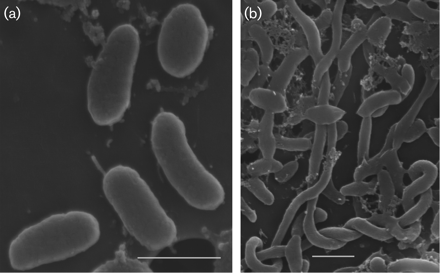
Idiomarina loihiensis is a rod shaped, Gram-negative aerobic cell driven by a single polar or subpolar flagellum. It was first throughly studied during a survey of bacterial diversity along deep sea hydrothermal vents located 35 kilometers off the coast of Hawaii. The volcanically active Lo'ihi Seamount is the home of Idiomarina loihiensis and several other active bacteria (2). The harsh conditions of the Lo'ihi Seamount, including high pressure and temperature (vents at 163º C), led to the early belief that these areas would be dominated by Archaea. However, further studies have indicated that it is Bacteria, instead that have dominated these vent microbial populations (2). Idiomarina loihiensis was one of the first bacterias cultivated and studied from these vents.
The bacteria was first collected in several dives during 1999 into Pele's Pit in the Lo'ihi Seamount. They were cultivated in marine agar or broth to be isolated and incubated at 30º C. The cells were determined to typically be 0.35µm wide and 0.7-1.8µm in length with optimum growth temperatures between 4-46º C. Comparison of the 16S rRNA genes with other bacteria revealed its similarity to I. abyssalis. However, the cultivated Idiomarina loihiensis were not similar to any other bacteria along the hydrothermal vents of the Lo'ihi Seamount. The strain cultivated, L2-TR, demonstrated the highest optimum growth salinity (7.5-10.0%) and growth temperatures relative to other species within its genus (2).
Genome structure
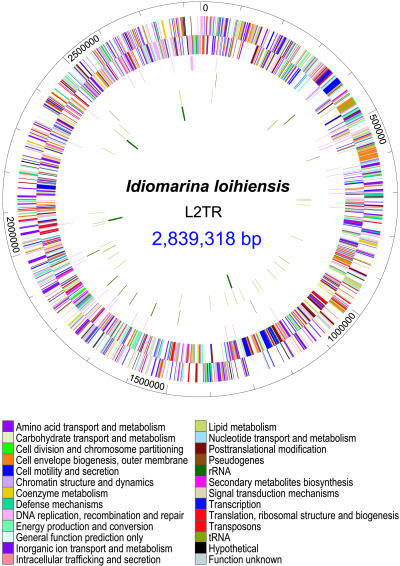
The genome of Idiomarina loihiensis is a circular chromosome comprised of 2,839,318 base pairs. It was fully sequenced through bacterial artificial chromosome (BAC) clone and whole-genome shotgun libraries (WGSL) and determined to have a G+C content of 47.04%. The genome contains 4 rRNA operons , encodes 2,640 proteins, and 56 tRNAs genes. Based on comparisons to other γ-proteobacteria, I. loihiensis's genome revealed a distinct excess of amino acid transport and degradation enzymes rather than of sugar transport systems and sugar metabolism (3). This indicates that I. loihiensis gains its energy and carbon sources through amino acid catabolism and not sugar fermentation. I. loihiensis has a cluster of 32 genes that specifically encode for viscous exopolysaccharides (EPS) to create necessary biofilms for their growth and survival within the hydrothermal environment (3).
Cell structure and metabolism
Based on its genome sequence, I. loihiensis has an excess of amino acid transport and degradation enzymes, revealing its gain of carbon and energy primarily through the use of amino acid catabolism and not sugar fermentation (3). Although animo acid catabolism is its its major source of energy; I. loihiensis lacks the enzymes required to synthesize Ile, Leu, Met, Thr, and Val. However of those five enzymes, only Thr and Val are required and are gained through hydrolysis of outside proteins. The abundance of heavy metals in the hydrothermal environment have allowed for an increase in peptidase production in I. loihiensis. While amino acid transport and degradation enzymes are abundant in I. loihiensis, the enzymes required for sugar metabolism are almost non existent (8). I. loihiensis does possess any forms of sugar transferase systems or transporters.
I. loihiensis is also unique in its fatty acid composition. It has double the normal percentages of saturated fatty acid in comparison to other Idiomarina species (3). It is also codes a complete set of enzymes that are required for fatty acid biosynthesis that may be significant in maintaining the cell membrane. Like many other deep-sea hydrothermal bacteria, I. loihiensis also produces exopolysaccharides that are used to create biofilms to help sustain their communities. These exopolysaccharides are highly viscous, which allows for its use in biotechnology (8).
Their deep-sea hydrothermal environment have allowed for several adaptations in I. loihiensis such as a flexible sensory transduction system and the ability to take in certain elements as well as the detoxification of heavy metals such as attaining nitrogen from both ammonia and nitrate. These adaptations are necessary due to the many fluctuations in the temperature, pressure, and oxygen within the environment (3).
I. loihiensis also uses multiple methods to gain energy; they encode a primary sodium pump to create a sodium ion gradient as well as an ATP synthethase specific for H so it does not have to rely primarily on the sodium ion cycle (3). These gradients are created with the use of multiple, variable Na/H antiporters within the cell.
Application to Biotechnology
Similar to other deep-sea hydrothermal microorganisms and those that live in extreme environments, Idiomarina loihiensis produces a viscous exopolysaccharide (EPS) that allows it to grow and survive under high pressure and temperature. Recently, EPS has been studied in its potential as food and nonfood thickeners due to its physical properties. Gram-negative cells, like I. loihiensis produce bacterial cellulose which have been used due to their high affinity to bind water (5). Because of its water binding capabilities, EPS has been used in a medical environment, such as in wound dressings for patients with tissue damage. Other species that produce EPS have also been used for texture in foods such as tofu, as well as emulsifiers and gelling agents. Some water-soluble forms of EPS have a high affinity towards metals; leading to its use in industrial sectors such as in oil and metal recovery from industrial waste (4).
Current Research
Using in silico analyses, further study was done to investigate I. loihiensis's unique, asymmetric chromosome. There is a distinct GC skew within the chromosome, indicative of a 600-kb chromosomal inversion (1). Comparison of the other related Gammaproteobacteria such as S. oneidensis and V. parahaemolyticus showed differences in chromosomal distances on specific segments. GC skew curves of I. loihiensis were asymmetrical and contained two inflexion points with an atypical curve between 0.7 and 1.3 Mb, indicating an inversion of regions in the chromosome. This may be caused by unique, uncommon configurations or perhaps misassembly of sequences (1).
Closer studies of Idiomarina loihiensis flagella demonstrate unique helical filaments. Typical flagella are categorized into Normal (left-handed) and Curly (right-handed). However I. loihiensis displayed left-handed Curly helical filaments requiring a modification of previously established Calladine's model for a filament lattice. Flagella studied under different pH environments and salt concentrations yield five different polymorphs; left-handed Curly and right-handed curly being most dominant. Two variants: left-handed Curly and left-handed semi-coiled were atypical from usual right-handedness in Salmonella flagella (6). This has led to the conclusion that there are at least two families of helical flagellar filaments.
N-acetylation of L-glutamate to carry out arginine biosynthesis in marine gamma proteobacteria use a combination of enzymes including N-acetylglutamate (NAGS) and ornithine acetyltransferase (OAT). However recent studies on two marine gamma proteobacteria Moritella abyssi and M. profunda have demonstrated a fusion designated argH(A). This fusion uses an enzyme in arginine biosynthesis and a complementary sequence to an argA mutant. Proteobacteria that possessed typical NAGS were compared against I. loihiensis and Pseudoalteromonas haloplanktis, both of which do not have typical NAGS and instead use the arg(A) sequence required for arginine biosynthesis. Because they do synthesize arginine yet lack the typical enzymes required for biosynthesis, is important in further studies of this new fusion argH(A) (7).
References
Edited by Beverly Lin, student of Rachel Larsen
Edited by KLB