Leptospira Species in the Environment
Introduction
By Toni Miller
There are various species of Leptospira, all belonging to the spirochete phylum. The Leptospira bacteria are characterized by their unique shape. These Gram-negative bacteria are long and helical in shape, similar to other spirochetes, but contain hooked ends. The hooked ends occur as the result of the two periplasmic flagella located at either pole of the cell. [Fig. 1] These are responsible for the rapid dissemination of the organism into the desired host. [6]Leptospires are obligate aerobes and grow best at temperatures ranging from 28-30°C. [7] One of the most common species of Leptospira is Leptospira interrogans.
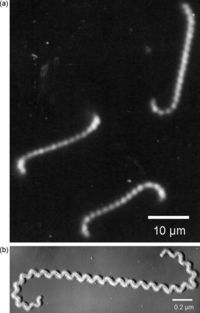
This species is the most predominant pathogen of the Leptospira phylum and results in a disease called leptospirosis. Leptospirosis is a zoonotic disease, predominant in the subtropical and tropical regions of the world. Most cases of leptospirosis in humans originate from a reservoir host, most commonly rodents, and are transmitted through water or contaminated soil. Specifically, L. interrogans colonizes the renal tubules of the host organism. [8] Leptospirosis in livestock can have detrimental effects on the agricultural industry, causing abortions, infertility and death. The effects of leptospirosis infection in humans can range from mild symptoms, such as a fever and a headache, to fatal problems causing death. These can include renal failure, jaundice, and pulmonary hemorrhage and are typically termed Weil's disease. [9],[10] Contrarily, L. biflexa is a non-pathogenic, saprophytic species that is incapable of infecting mammalian hosts. Although sharing many similarities in genetic and structural components, the L. biflexa lacks the genes necessary for virulence. The saprophytic species have been observed to have more overall genes however, serving as nutrient absorption. [11] With over 13 different species of pathogenic Leptospira and the recorded number of leptospirosis cases well over 1 million annually, Leptospira bacteria are ideal for research. Not only are there vast differences between these spirochetes and others, such as the bacteria responsible for Lyme Disease, but there are also many differences between each species of Leptospira. [12]
Genomic Difference Between Pathogenic and Saprophytic Species
Leptospira share many of the same features with other spirochetes yet have important differences that allow for the unique shape of the bacteria. Differences can be seen when comparing Leptospira to other spirochetes, as well as comparing the different species of Leptospira. The genomic and structural differences within species of the same genus, give rise to the different pathogenic and saprophytic traits seen in each of these species. Between the pathogenic and saprophytic species, there are around 2,052 common genes that make up the core of the genome. Research has shown that the functional categories of DNA and RNA metabolism, as well as protein processing/secretion and cell maintenance have been conserved across the varying species. However, the saprophytic species, L. biflexa, contains a third circular replicon that is absent in the pathogenic species. Most of the genes present in the replicon account for environmental sensing and nutrient acquisition. Because these species rely on the environment and not a host organism, it is imperative that the saprophytic species have these genes to code for the different mechanisms needed to adapt to the various environments. [13],[14] Differences can also be seen in the transmission methods of different pathogenic species. For example, L. interrogans can be transmitted from animal to animal/human though direct contact of infected urine or contaminated water. This transmission method requires the microbe to be in the environment for extended periods of time in between mammalian hosts. On the other hand, L. borgpetersenii, another pathogenic species, cannot survive for long periods of time in the environment and therefore has a transmission method that is much faster than that of L. interrogans. L. borgpetersenii can only be transmitted through direct contact because it is unlikely to survive in the environment. [15]
Morphological Differences Between Species
Several differences can be seen in the structural representation of the Leptospira spirochetes compared to others. These differences can be seen in the DNA, cellular envelope, periplasmic filaments, spherical bodies, and flagellar motor. The DNA in Leptospira is not contained in the central nucleoid region like E. coli, but instead is distributed along the cell body, except for the terminal ends. Further analysis has shown that the DNA is arranged in filamentous bundles. Similar to Gram-negative bacteria, Leptospira contain a double membrane envelope, consisting of an inner membrane, outer membrane, and peptidoglycan layer. However, unlike other spirochetes such as T. pallidum and B. burgdorferi, the outer membrane contained a third density layer, or leaflet. This layer was made up primarily of lipopolysaccharide, or LPS. A major difference between L. interrogans and L. biflexa was the thickness of the third density layer of the outer membrane. The L. interrogans had a thicker third layer, suggesting the LPS is variable in each species and that the amount of LPS in the cell plays a role in the virulence [Fig. 2].[16],[17]
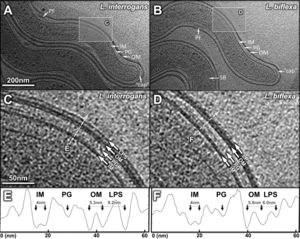
The periplasm of the cell contains flagella and novel filaments that give rise to the shape of the cell. The flagella filaments, which are located at the cell termini, are responsible for the hook shape of the cell's ends and for the overall motility of the cell. Different though is that these filaments are not responsible for the overall spiral shape of the cell. This shape is believed to be derived from the novel filaments. These filaments wrap around the cell, resulting in a spiral shape. This suggest that the novel filaments are responsible in determining the shape of the Leptospira. [18] The saprophytic and pathogenic species of Leptospira both contain spherical bodies. These spherical bodies are located in the cytoplasm and have been observed along the cell bodies of both L. biflexa and L. interrogans. [Fig. 3]
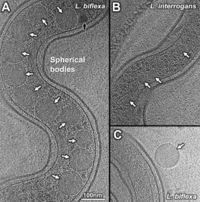
Two different spherical bodies were observed: a high density body and a low density body. Although primarily inside the body of the cell, they have previously been found outside of the cell as well. The bodies were considerably larger in the saprophytic (L. biflexa) species than in the pathogenic species (L. interrogans). The difference in size was on average 70 nm. Because of their lack of metabolic complexes and the similarities between these and the phosphate/carbon rich bodies seen in other species of bacteria, it is assumed that the role of these spherical bodies is simply nutrient storage. If this is the case, it would make sense that the larger bodies would be found in the saprophytic species because they are completely dependent on the environment. [19]
Comparing Leptospira to other spirochetes, it is easy to identify a major difference in motility: the number of flagella. While many other spirochetes contain multiple, Leptospira contain one flagella at each end of the cell. The primary differences in overall motility of the cell, come in the general size of the C ring, P ring and export apparatus. [Fig. 4]
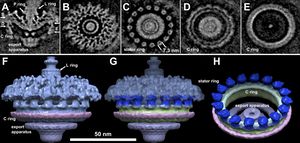
Compared to other spirochetes, the C ring and export apparatus are bigger and more complex. The P ring, which is responsible for a density layer on the distal rod, is missing in most other spirochetes. The extra density layers and bigger mechanisms allow for more powerful flagellar rotations, allowing the bacteria to move rapidly through their environment. [20] Without the endoflagella, the cell would not be able to inhabit the host's tissues as quickly or as effectively. This would result in faster diagnosis of illness and more time to respond to the illness. The leptospire flagella allow for the bacteria to invade and colonize at such a rapid rate, that treatment is often ineffective. The flagella are responsible for the amount of fatalities attributed to leptospirosis.
Flagellar Motility
As previously mentioned, the bigger structures and thicker density levels involved in the flagellar complex allow for better movement of the leptospira, compared to other spirochetes. The direction of the flagellar rotation is regulated by chemotaxis, or movement toward or away from a chemical attractant. Due to the difference in ecologic and metabolic differences between the saprophytic and pathogenic species, the attractants and deterrents causing chemotaxis are different. The proteins in the genome of the saprophytic species have twice as many chemoreceptors. The presence of so many homologs allows the saprophytic species to adapt and thrive in so many different environments. Both species showed an attraction to sucrose and glucose, while little attraction was given to amino acids. Both palmitate (long-chain fatty acid) and hemin (source of iron) resulted in positive chemotaxis of L. interrogans but negative chemotaxis in L. biflexa. [21] The chemoreceptors involved in the motility of the cell also vary within the different species of Leptospira. The receptors are located approximately 500 to 1,000 nm away from the cell ends. However, the L. biflexa organisms have a chemoreceptor array consistently located at the cell end. Structurally, this array varied greatly from the others. This array had additional domains and was slightly longer than all the others. Although located near the flagella at the pole, the array was more readily visible in the L. biflexa than in the L. interrogans. By having a larger array, the L. biflexa is better equipped at detecting the various chemicals in the environment that it can use as attractants of deterrents.
Transmission and Infection of Pathogenic Leptospires
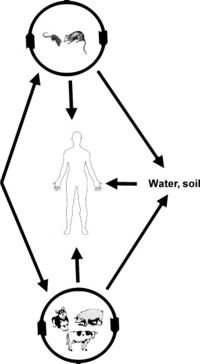
Leptospires can enter the human body through cuts or through mucous membranes. The leptospires are then able to circulate in the bloodstream for several days, branching out to various tissues in the body, such as the kidney and liver. Lesions will begin to appear due to unknown leptospiral toxins. As lesions accumulate on the tissue, necrosis and ischemia start to occur to the various organs. It is possible for tissue damage to be repaired once the antibodies have removed all the leptospires from the bloodstream. However, long lasting tissue damage can occur. This can lead to scarring on the organs, termed "white spots" when it is seen macroscopically. [22] Typically, Leptospirosis is found in dogs, cattle and swine who have been exposed to infected water or soil. [Fig. 5] The disease is characterized by several different symptoms: fever, renal insufficiency, pulmonary and reproductive failure. Transmission of the disease can occur to humans through mucous membranes or damaged skin that comes into contact with soil or water that has been contaminated by the infected organism. Transmission between animals occurs mainly through exposure to animal excrement that has entered the water supply. Due to the localization of the pathogenic bacteria in the renal tubules, most contamination is a result of an infected animal (i.e. cattle) discarding urine containing leptospira. Leptospires have also been found to be excreted in abortion products of some infected domestic animals. Many mammals become carriers of leptospires except for humans who mostly just suffer from acute infections. [23] The pathogenesis of leptospirosis can be identified by the stage-specific interactions between host and pathogen, mediated by the outer membrane proteins. Leptospires are capable of binding to various host ligands such as the following: fibronectin, fibrinogen, collagen, laminin and elastin. The outer membrane proteins involved in pathogenic bacteria are responsible for several things. These proteins can act as adhesins, as targets for antibodies, and as receptors for host molecules. Specifically, the proteins in Leptospira species are responsible for the adaptations occurring in response to the host's tissues or the change in environmental conditions that occur throughout the life cycle of the bacteria. Along with adapting to the environment, the different host ligand-binding proteins on the outer surface of the Leptospira can recognize host molecules, fight against the host's defense and ultimately instigate the invasion and colonization of the various tissues within the host. Knowing the ligand-binding properties of Leptospira is important for the development of antibiotics and vaccines. [24]
Symptoms of Leptospirosis and Co-infection
In extreme case of Leptospira infection, the host organism can be said to have Weil's disease. This disease is considered to be life-threatening and it is necessary for the host to undergo laboratory testing. This severe infection is typically caused by icteric serovars, often times Icterohaemorrhagiae. [25],[26] Again, although most commonly found in domestic animals such as dogs and cattle, the disease can be easily transmitted to the human owners. At the onset, the symptoms present as standard influenza symptoms, such as headache, chills, fever, and nausea. It is only when the symptoms become very severe that the organism is recognized to have Weil's disease. These symptoms include the following: aseptic meningitis, hemorrhages, cardiac arrhythmia, and psychosis and/or delirium. [27] A majority of leptospirosis cases are reported near the tropic and subtropic regions of the world than anywhere else. This is due to the reliance on natural water that has not been processed or purified as drinking water. These areas are also known to have warmer climates and with it, a higher number of mosquitoes. This plays an important role in the rare cases of co-infection of Dengue Fever and leptospirosis. Dengue fever is a virus transmitted by mosquitoes and coincidentally results in similar symptoms. Although the symptoms for Dengue Fever and leptospirosis are similar, the treatments are very different. It is near impossible for medical personnel to determine the status of the sick individual by looking just at their symptoms. It is necessary in these situations for medical testing to be done to determine whether the individual has Dengue Fever or leptospirosis. However, in areas where medical testing is limited, the individuals can simply be treated as being "co-infected" and both illnesses are treated simultaneously. [28] Leptospirosis can be very detrimental to agricultural industries, such as dairy farming, and pig production. If left unattended, the disease can destroy an entire herd of livestock easily. Once infected, the disease can easily be transmitted to other healthy hosts. The disease also has the potential to affect the reproductive system of the host. In both severe and acute cases of leptospirosis, intrauterine infection and fetal death have been reported. It is important to realize that if the reproductive system is infected, any abortion products expelled by the host will also be infected and can lead to the transmission of the disease if contact is made between these products and the water supply.[29],[30]
Treatment and Control of Leptospirosis
Once infected with leptospirosis, there are several options of treatment, depending on the severity of infection. In mild cases, antibiotics, such as penicillin, are given. This treatment however has a tendency to be problematic. Rapid death of spirochetes within the body, commonly from drug use, is associated with a life-threatening disease, known as Jarish-Herxheimer reaction. When the spirochetes die from the antibiotics, they release endotoxins into the body. These endotoxins are released at a faster rate than the body can remove them, resulting in the accumulation of these toxins within the body. This results in fever, chills, headache, hypertension, etc. [31] However, in extreme cases of Leptospira infection in which the host is going into renal failure, the individual can undergo dialysis to protect against fatality. Because the main method of transmission is between animal excrement and human, the prevention of treatment is associated with the actions of the human and their interaction with the infected animal. For example, protective clothing can be worn when dealing with sick cattle and measures can be taken to improve the water supply of the animals to decrease the risk of the animals consuming contaminated water. To account for infected rodents, there are rodenticides that can be used, however they are risky to other non-infected organisms in the surrounding environment. Looking specifically at domestic animals, vaccination is available. These vaccinations give short-term protection to specific serovars. Because of their short time frame, boosters are needed often to ensure the vaccine is still effective. Up until now, human vaccines have been limited, particularly in the United States. Like the animal vaccines, they are short lived and would require boosters and only protect against certain serovars. [32]
Geographical Distribution of Leptospires
The high presence of leptospirosis in tropic and subtropic regions, brings questions as to why there is not a greater distribution of the disease. Spirochetes are unique in that serovars are capable of living outside the host for periods of time. Across species, the amount of time an organism can spend outside the host is not conserved, as was seen between L. interrogans and L. borgpetersenii. Without the death of these bacteria once they leave their mammalian host, the risk of infection increases. The survival of these Leptospira bacteria outside the host are dependent on humid and warm conditions (Hence why they are primarily located in tropical regions). Thinking globally, increased rainfall and global warming throughout the world are two important factors triggering the growth of Leptospira in the environment and consequently, increased infection and Leptospirosis. Environmental changes can lead to migration of Leptospira hosts, both humans and animals. Environmental changes and the changes in socio-economic standing of human populations also lead to a greater risk of infection. For example, many people decide to travel to these tropical and subtropical destinations as a tourist activity. Any interaction with contaminated water is an opportunity for these bacteria to invade a host and be carried back to the host's homeland. These interactions can cause for an increase in the distribution of the Leptospira bacteria. [33]
Conclusion
Leptospira species are highly present in the environment. There are several pathogenic species, such as L. interrogans, but also many saprophytic species, like L.biflexa. Many differences can be seen between the pathogenic and saprophytic species, as well as between Leptospira species and other spirochetes. The most apparent difference between Leptospira and other spirochetes, is the presence of a hook shape at either end of the bacteria. This is caused by endoflagella. Between the saprophytic and pathogenic species, many differences can be seen in both morphology and in their respective genomes. Morphologically, differences arise in the location of the DNA, the cellular envelope, the periplasmic filaments, presence of spherical bodies, and size and complexity of the flagellar motor. The most apparent difference between the two types of Leptospira bacteria is the density of LPS, or lipopolysaccharide, in the cell. Compared to L. biflexa, L. interrogans had considerably more LPS, suggesting the LPS plays a role in virulence. Genomically, the saprophytic species were found to contain a third replicon, containing genes coding for environmental sensing and nutrient acquisition. L. interrogans, along with other pathogenic species, are responsible for the zoonotic disease called Leptospirosis, that occurs primarily in tropic and subtropic regions. After typically colonizing in the renal tubules of the host, transmission occurs primarily through contamination via urine. While mild cases of infection results in average influenza symptoms, severe cases can cause aseptic meningitis, hemorrhages, and even death. Leptospirosis is very detrimental to agricultural industries, as contamination can occur easily. Although vaccines exist, they are short lived and therefore most infection is cured through antibiotics.
References
1. Pinne, M.; Matsunaga, J.; Haake, D.A. Leptospiral Outer Membrane Protein Microarray, a Novel Approach to Identification of Host Ligand-Binding Proteins. Journal of Bacteriology. 2012, 194, 6074-6087. [34]
2. Raddi, G.; Morado, D.R.; Yan, J.; Haake, D.A.; Yang, X.F.; Liu, J. Three-Dimensional Structures of Pathogenic and Saprophytic Leptospira Species Revealed by Cryo-Electron Tomography. Journal of Bacteriology. 2012, 194, 1299-1306. [35]
3. Adler, B.; Lo, M.; Seemann, T.; Murray, G.L. Pathogenesis of Leptospirosis: The Influence of Genomics. Veterinary Microbiology, 2011, 153, 73-81. [36]
4. Adler, B.; de la Pena Moctezuma, A. Leptospira and Leptospirosis. Veterinary Microbiology, 2012, 140, 287-296. [37]
5. Abdoel, T. Rapid Test for the Serodiagnosis of acute canine leptospirosis. Veterinary Microbiology, 2011, 150, 211-213. [38]
6. Slamti, L.; de Pedro, M.A.; Guichet, E.; Picardeau, M. Deciphering Morphological Determinants of the Helix-Shaped Leptospira. Journal of Bacteriology, 2011, 193, 6266-6275. [39]
7. Eribo, B.; Mingmongkolchai, S.; Yan, T.; Dubs, P.; Nelson, K. Leptospire Genomic Diversity Revealed by Microarray-Based Comparative Genomic Hybridization. Applied and Environmental Microbiology, 2012, 78, 3045-3050. [40]
8. Lambert, A.; Takahasi, N.; Charon, N.; Picardeau, M. Chemotactic Behavior of Pathogenic and Nonpathogenic Leptospira Species. Applied and Environmental Biology, 2012, 78, 8467-8469. [41]
9. Hartskeerl, R.A.; Collares-Pereira, M.; Ellis, R.A. Emergence, control and re-emerging leptospirosis; dynamics of infection in the changing world. Clinical Microbiology and Infection, 2011, 17, 494-501. [42]
10. Sharma, K.K.; Latha, P.; Kalawat, U. Coinfection of leptospirosis and dengue fever at a tertiary care center in South India. Scholar's Research Journal, 2012, 2, 12-16. [43]
11. Jarisch-Herxheimer reaction. Wikipedia. Accessed 4/20/14. [44]
