Machine Learning Approach to Antibiotic Discovery
Summary
By Reed Crocker
Biology 238: Microbiology
Spring 2020
Antibiotics are essential for the modern practice of medicine. However, the rise of antibiotic-resistant bacteria presents new challenges to this paradigm in medical treatment. Discovery and development of novel antibiotics is not meeting the pace of microbial resistance. The arms race against pathogenic bacteria has prompted significant research into new modes of antibiotic discovery. One rapidly expanding sector of this research uses a high throughput, machine learning approach to predict candidate antibiotics in silico.
Traditional Discovery Methods
One of the first experimental platforms for antibiotic discovery is known as the Waksman Platform. First implemented by the Rutgers microbiologist Selman Waksman, the Waksman Platform involved the screening of soil-borne actinomycetes on a field of overlaid bacteria[1]. Actinomycetes are bacteria which commonly produce antibiotics in their soil environment. The development of a zone of inhibition on the plate was the primary way candidate antibiotic-producing microbes were discovered. This technique was successfully used to discover several important antibiotics including streptomycin. While this platform was fundamental to finding many of the world’s antibiotics, it has certain limitations. First, there is a finite number of soil antibiotics. Approaches to sample more diverse soils and bacterial environments have been mostly unsuccessful in producing new classes of antibiotic[2]. Second, the approach is not easily scaled up, meaning the process is slow and laborious. These reasons are some of many to prompt high throughput computational approaches to antibiotic discovery.
Machine Learning
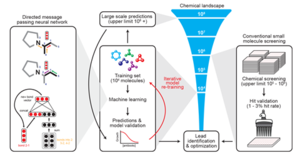
A paper published in February of 2020 revealed a novel platform for the computational discovery of antibiotics. This approach utilizes machine learning and enormous drug databases to train their algorithm to predict the effectiveness of potential antibiotics. This deep learning approach was successful in the identification of halicin, a novel antibiotic with broad spectrum activity. Halicine works to inhibit proton motive force and effectively inhibit growth of deadly pathogens like Clostridium difficile, Acinetobacter baumannii, and Mycobacterium tuberculosis. With halicin, the researchers validated the bactericidal function with physical in vitro experiments. However, the researchers also discovered 8 functionally distinct other potentially antibacterial compounds. This publication shows the new pace of antibiotic discovery that will become possible with improved computational and deep learning methodologies.
Advantages of Machine Learning in Antibiotic Discovery
New machine learning platforms as described above have overcome many of the challenges presented by the Waksman Platform and similar approaches. Compared to manually screening soil-derived microbes and their potential antibiotics, machine learning approaches have the potential to exponentially improve the rate of antibiotic discovery. This increase in speed is achieved by performing analyses in silico. With computers becoming more efficient every year, the discovery rate will likely get even faster. Improved learning algorithms and greater drug libraries will also be a key factor in improving discovery rate.
This approach will have lower cost, as it replaces physical tools and labor with a computational workflow. What before may have taken several microbiologists and years of time can be predicted with the deep learning model in only a few days.
This approach is also not limited by the origin of the candidate compounds. While the Waksman platform relied on finding unique soil-borne, antibiotic-producing actinomycetes, approaches like described in Stokes et al., 2020 analyze more structurally diverse compounds from various origins. The ZINC15 database used in this machine learning approach contains over 750 million compounds.
Conclusion
Deep learning application to antibiotic discovery has enormous potential to improve the field of antimicrobial therapeutics. This approach overcomes many of the challenges of traditional discovery platforms and sets an important precedent for the future of antibiotics. The improved efficiencies provided by this method and future iterations will aid in our arms race with antibiotic-resistant pathogens.
References
- ↑ [1]
- ↑ [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4692294/ Valiquette, 2015]
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.
