Microbial Biofuels: Current Production and Future Prospects
By Mark Michael Harden
Introduction
Fossil fuels like coal and oil have played a critical role in humanity’s recent history, providing a vast energy source which has fueled much of society’s development and industrialization. These fuels are still the primary source of energy for the world’s developed nations, and yet it is agreed that these traditional sources of energy cannot continue to power humanity’s growth into the future. The demand for oil production is at an all-time high, and will only increase as developing nations continue to grow. Furthermore, many experts predict that the rate of world oil production has already peaked, and that it will only decrease from now onwards as fewer and fewer oil reserves are discovered. This decreasing supply and rising demand will drive up the price of oil and other fossil fuels, and will eventually make them economically unsustainable [2]. The use of fossil fuels poses other problems as well, most notably that their consumption is environmentally unsustainable. Burning fossil fuels produces enormous quantities of the greenhouse gas carbon dioxide, which has a negative impact on the Earth’s environment by contributing to global warming [10, 14]. For all of these reasons, there is great incentive to pursue the development of renewable energy sources, particularly microbial biofuels.
Microbial metabolism is incredibly varied, and can both utilize and produce a wide variety of useful molecules. Many microbial systems are also well characterized and easy to manipulate genetically, and scientific advances will only make these systems easier to work with in the future [6]. Although there is no biofuel option currently available which solves all of the economic and environmental issues associated with fossil fuels, the potential for both “fine-tuning” biofuel-producing microbes, and genetically modifying species to be able to efficiently make use of otherwise useless materials and byproducts, makes microbial biofuels an appealing target for research [6].
Current Microbial Biofuels: Ethanol Produced by Fermentation
Microbial biofuel production is already in use, principally in the form of sugar fermentation by yeast to produce ethanol [4]. Although many microbes have been used in ethanol production, the yeast species Saccharomyces cerevisiae is primarily used in industry, using starch and sugars from plants as the starting material for the process [3]. The most common feedstocks (carbon source utilized by the microbes) are agricultural products which can easily be processed to create the simple sugars needed for fermentation. This is primarily corn in the United States, wheat in the European Union, and sugar cane in Brazil [4, 10].
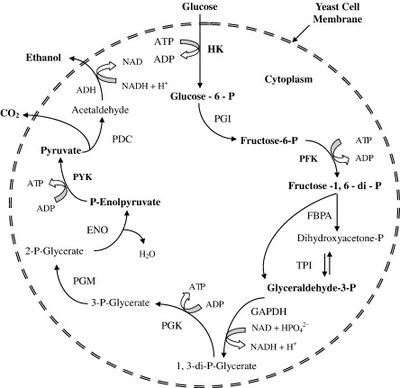
Image source: http://www.sciencedirect.com/science/article/pii/S0734975007001000 [3]
Ethanol fermentation by S. cerevisiae is primarily done via the standard glycolysis pathway (Figure 1) [3]. In the case of corn and other starch-containing plants, the simple sugars necessary are formed via the hydrolysis of starch to yield monosaccharide subunits, whereas the sugars in sugarcane are hydrolyzed only once and then go straight into the pathway [6]. In the process, a single molecule of glucose is oxidized to two molecules of pyruvate. Anaerobic conditions are required so that molecular oxygen is not available for use as an electron acceptor, and instead pyruvate must be used as the terminal electron acceptor. This fermentative process involves the decarboxylation of pyruvate to form carbon dioxide and acetaldehyde, and the subsequent reduction of acetaldehyde to produce ethanol [3].
The use of fermented bio-ethanols as a fuel source offers some advantages over standard fossil fuels. Both corn ethanol and sugarcane ethanol require an input of fossil fuel energy in order to produce them because of the energy requirements of farming, processing, and transportation. However, several studies report that they yield more biofuel energy than fossil fuel energy that is invested in them, giving them a positive net energy balance (NEB), which means that they represent more efficient energy sources. The NEB for corn ethanol is relatively small, at approximately 25% extra energy output than the required fossil fuel energy investment [10]. This means that, while corn ethanol reduces the amount of necessary fossil fuel, it does not represent a total solution to the problem. Ethanol produced via sugarcane fermentation has a much higher reported NEB than corn ethanol, yielding almost eight times as much biofuel energy compared to the required input of fossil fuel energy. However, because sugarcane only grows in certain tropical climates, its fermentation also does not represent a viable solution for the worldwide problem [9].
Ethanol fermentation by yeast also helps to address the problem of greenhouse gas emissions, although it does not represent a perfect solution from an environmental perspective either. All biofuels with a positive NEB should theoretically emit less carbon dioxide, because the process of carbon fixation occurring within the growing plants should counterbalance the carbon dioxide emissions of both the invested fossil fuel energy and the combustion of ethanol [10]. However, in reality the nitrogen-rich fertilizer used to sustain the plants and the addition of extra plant matter into the soil supports communities of bacteria that produce nitrous oxide, a much more potent greenhouse gas than carbon dioxide. Considering this entire system, producing ethanol via corn fermentation emits approximately 88% of the greenhouse gas content of gasoline yielding the same amount of energy [15]. This mediocre improvement, coupled with the other environmental implications such as pesticides, make most current ethanol fermentation techniques of limited use, although they are nevertheless a positive alternative to fossil fuels.
Alternative Ethanol-Producing Microorganisms: Zymomonas mobilis and Escherichia coli
A major problem that plagues ethanol fermentation by S. cerevisiae is that the fermentation of sugars by glycolysis also produces ATP, which is then used in reactions of yeast cell growth and division. Although the production of growth-inducing ATP is a natural function of glycolysis, in this industrial setting the creation of undesired biomass is essentially an unwanted byproduct, and represents wasted carbon and energy resources which could have gone into increased ethanol production [12]. Although this problem can be somewhat resolved by immobilizing the yeast cells to retard cell growth, the consequence of slowing growth is that the intracellular ATP concentration quickly increases, and the accumulation of this product of glycolysis downregulates the entire glycolytic pathway, thereby decreasing ethanol production [3]. The use of yeast presents other problems as well, particularly the negative effects of environmental and intracellular stress which accumulate in the fermentation environment. The challenges faced in such an environment include high concentrations of ethanol, high sugar concentrations which increase osmostic pressure on the cells, temperatures as high as 38°C, high sodium ion concentrations, and others. These factors will often be a greater source of stress when acting together, making the conditions for S. cerevisiae even worse. The stress reduces the yeast’s overall viability, thereby inhibiting its ability to produce ethanol [12]. Overall, S. cerevisiae can make ethanol and carbon dioxide from glucose at an efficiency of about 90-93% of the theoretical yield [3].
Some of these efficiency problems could potentially be overcome by using different organisms to perform the fermentation reaction. One major contender is Zymomonas mobilis, an unusual anaerobic gram-negative bacterium. The Zymomonas genus of bacteria is unique, and potentially useful because members of the genus break down glucose through the Entner-Duodoroff (ED) pathway. The ED pathway differs from glycolysis by producing only one molecule of ATP instead of two, while still producing two molecules of pyruvate. The pyruvate can then be decarboxylated as usual to form acetaldehyde and carbon dioxide, at which point the acetaldehyde is reduced to produce ethanol. Because Z. mobilis only makes one molecule of ATP per glucose molecule, it grows and divides much more slowly than S. cerevisiae, and thus results in fewer resources wasted by creating unwanted biomass [12]. Furthermore, because it has a generally lower concentration of ATP, there is less product-inhibition of the ED pathway, and as a result flux through the pathway is increased overall [3]. Z. mobilis is also appealing because it produces ethanol more exclusively than S. cerevisiae, and is capable of withstanding certain stresses better, including a higher concentration of environmental ethanol. These combined differences allow Z. mobilis to convert glucose to ethanol at as much as 97% of the theoretical yield, and do so at a much faster rate than S. cerevisiae [3]. Unfortunately, the bacterium can only utilize a few simple mono- and disaccharides as a carbon source to produce ethanol, including glucose, fructose, and sucrose, and it will preferentially use glucose above all others [3, 12]. The specificity of Z. mobilis for a small handful of monopolysaccharieds means that using the bacterium requires additional processing of the feedstock, which is economically and energetically unfavorable compared to other microbial alternatives. Furthermore, it is more sensitive to certain kinds of environmental stress, and especially acetic acid, a common compound found in industrial fermentation environments [13].
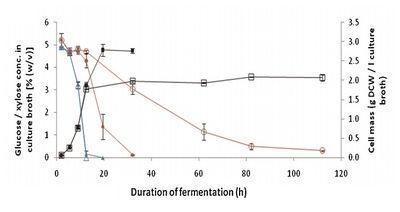
Image source: http://onlinelibrary.wiley.com/doi/10.1002/bit.23021/full [1]
Although these limitations have prevented the bacterium from being applied in industry, some researchers have attempted to compensate for them. For instance, the group of Deanda et al. used metabolic engineering to produce Z. mobilis strains capable of consuming five carbon sugars like arabinose and xylose. In one experiment during the 1990’s, they cloned the Escherichia coli araBAD operon and used it to construct a Z. mobilis strain capable of consuming arabinose. However, the strain still preferentially utilized simple six carbon sugars, and the arabinose flux was too low to be used effectively [7]. More recently, the group of Agrawal et al. has successfully cloned E. coli xylose metabolic genes into a strain of Z. mobilis, and then used an adaptive laboratory evolution technique to enhance its otherwise poor ability to ferment xylose. After an extended culture period in medium containing both glucose and xylose, followed by a period of culturing in medium containing only xylose, the researchers ultimately isolated a Z. mobilis strain that could utilize xylose much more effectively. A comparison between a strain isolated partway through the adaptation process (A1) and the final adapted strain (A3) can be seen in Figure 2. When cultured in media containing both glucose and xylose, the more ancestral strain A1 grew quickly while there was still glucose present in the medium, but hardly grew at all once the only sugar left in solution was xylose. On the other hand, the adapted strain A3 could continue to grow in the absence of glucose, quickly consuming all of the xylose in the solution as well [7]. With the continued development of Z. mobilis strains, either by adaption or metabolic engineering, Z. mobilis might eventually be used to greatly increase bioethanol production and efficiency.
E. coli is another potentially useful microbe for ethanol production. This is because E. coli is well known for its ability to consume a wide variety of sugars (unlike Z. mobilis), and the well-known bacterium is already very well established as an industrial tool. E. coli also has the advantage of being the ideal candidate bacterium for experimentation with metabolic engineering because it has been so extensively studied as a model organism [6, 12]. Indeed, the late 1980s and 1990s saw an extensive amount of research into establishing an E. coli strain that would be optimized for ethanol production. However, E. coli is also much more sensitive to environmental conditions like pH and ethanol concentration, which has successfully prevented it from being used for this process so far [12].
Alternative Feedstocks
Lignocellulose
The production of bioethanol typically only utilizes the starchy or sugary components of the agricultural crop, wasting much of the energy locked away in the plant’s biomass. The unused parts of the plants, like stalks, leaves, and wood, are composed of lignocellulosic materials, and are much more difficult for microbes to break down. If these materials could be exploited, they would represent a massive new energy resource for biofuel production [5]. Lignocellulose is made up of different combinations of cellulose, hemicelluloses, and lignin, which together make up a plant’s cell walls and provide structural support for the organism. Cellulose is a homopolysaccharide of glucose connected by β(1,4) linkages between monomers. The unusual β-linkage (instead of the α-linkages found in most other biological systems) is much more resistant to microbial degredation. Hemicellulose is a heteropolysaccharide, which exists in great variety and can be comprised of many different sugars, including xylose, arabinose, mannose, and galactose. Because hemicellulose composition is so diverse, many microbes would not be able to utilize such a wide range of sugars. The final component of lignocelluloses, lignin, is a not a polysaccharide, but rather is a complex network of phenyl-propane units. Lignin is interwoven through the plant’s cellulose, providing structural support and further protecting it from microbes [11].
Despite the potential gains of using lignocellulose for feedstock, the challenges of degrading lignin and cellulose require creative solutions which often involve expensive pretreatment procedures. For instance, digesting the lignin in the plant material is inefficient enough that consuming it is impractical, so it must be separated from the carbohydrates. But even after separation from lignin, the remaining cellulose and hemicellulose is still resistant to digestion with hydrolysis enzymes, and must undergo further processing prior to any enzymatic digestion. Even then, because this digest contains a wide variety of different types of monosaccharides, the new challenge of obtaining a microbe (or a collection of microbes) which can consume all of these products presents itself. Substantial research has been done on the genetic engineering of metabolic pathways, and it is now possible to introduce entire pathways to “design” specialized microbes capable of both breaking down and synthesizing a wide range of compounds [6].
Unfortunately, all of these pretreatment steps make utilizing lignocellulose extremely expensive. However, a possible solution to this problem is to consolidate the enzymatic hydrolysis step with microbial processing, by engineering microorganisms capable of exporting the enzymes necessary to break down cellulose and hemicellulose. The research group of Bokinsky et al. has recently made progress in this area. The group intended to obtain an engineered strain of E. coli which could synthesize the enzymes necessary for cellulose and hemicellulose digestion, and then synthesize several different biofuels. They used combinations of specially designed plasmids containing the relevant metabolic enzymes from different microbes, placed under the control of ordinary E. coli promoters. The group was successful, and obtained strains which expressed and exported cellulose- and hemicellulose-digesting enzymes, which could both break down these plant biomass components and then use them to produce three different useful biofuels: pinene, butanol, and fatty acid ethyl esters. The plant material only needed to be pretreated with an ionic liquid, thereby making this a potentially useful and economical alternative [5].
Fatty Acids and Glycerol
Microbes are incredibly diverse with respect to their ability to utilize different substrates, and this diversity provides the opportunity to use different compounds for making biofuels besides the standard carbohydrates. In particular, fatty acids provide one appealing alternative to sugars. Because they are so much more reduced than simple carbohydrates, fatty acids contain much more carbon and energy with which to produce greater biofuel yields. Furthermore, because fatty acids are broken down directly into acetyl-CoA and do not have to go through the intermediate of pyruvate, they never have to be carboxylated, and thus unlike glycolysis, every carbon molecule from the substrate could theoretically be incorporated into the product [6].
Glycerol could also be a useful feedstock, as it is readily produced as a byproduct of many industrial processes, and could be harvested from microbes like those of the algae genus Dunaliella, which produce it in abundance. The low cost and availability would be an important asset: its economic viability would place it in stark contrast to agricultural feedstocks like corn, the use of which in biofuel production has unfortunately driven up food prices. Furthermore, glycerol is highly reduced, and thus would provide more energy and carbon to produce biofuel with [6].
Alternative Biofuels
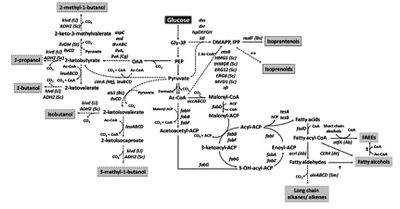
Image source: http://link.springer.com/article/10.1007/s00253-010-2446-1 [6]
Other Fermentation Products
Ethanol fermentation was the first to be put to industrial application, but the field of synthetic biology has opened up many new possibilities. Because ethanol is not compatible with most modern engines when used alone (it must be mixed with another fuel), a lot of research has been done to clone entire metabolic pathways, introduce them to relevant organisms for biofuel production, and optimize them for industrial purposes. Two possibilities are isopropanol and 1-butanol, which would be advantageous because they are compatible with most modern engines. Several species of the Clostridium genus are known to naturally produce these compounds via fermentation reactions, and there has been some success with introducing these pathways to E. coli. Production of these larger, more highly reduced alcohols requires additional acetyl-CoA and NADH, so knocking out enzymes which compete with these substrates can improve the organism’s production efficiency [6].
Non-Fermentation Products
The same advances in synthetic biology also allow researchers to “cut and paste” enzymes, and make an organism like E. coli produce a new product through a pathway which does not occur naturally. Extensive research has yielded a myriad of working non-fermentative metabolic pathways, allowing simple E. coli to produce many different types of alcohols, isoprenoids, fatty-acid alcohols, and much more (Figure 3). None of these alternative pathways have been applied to industry yet, and will require more research to optimize them and make them viable alternatives [6].
Improving Microbial Biofuel Production: Recent Developments
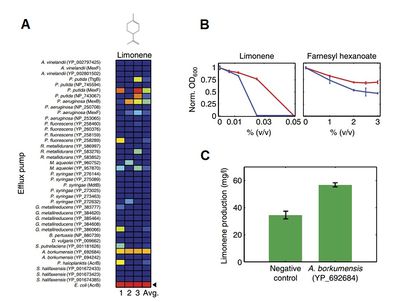
A) A competition assay was performed by co-culturing many strains of E. coli, each containing a unique efflux pump-encoding plasmid. One culture was maintained per biofuel investigated, each containing a significant amount of the relevant biofuel (here limonene). Comparative strain concentrations were measured after 24 (1), 48 (2), and 72 (3) hours. Dark red represents high relative concentration of the strain, dark blue represents low relative concentration of the strain.
B) The efflux pump AcrB confers resistance to the biofuel limonene. Strain indicated with an arrow from (A) and a strain containing only the plasmid vector were grown in separate cultures for 24 hours in the presence of varying concentrations of limonene.
C) The efflux pump increases limonene production in a biofuel-producing strain. The production strain A. borkumensis was transformed with the plasmid coding for the efflux pump AcrB, or an empty plasmid vector, and then grown under limonene production conditions. Media was analyzed for limonene concentration at the end of the production period.
Image source: http://www.nature.com/msb/journal/v7/n1/full/msb201121.html [8]
Using Efflux Pumps to Improve Biofuel Production
A common problem of biofuel production is that the biofuel itself is frequently toxic to the cell. This is the case with ethanol, the most common biofuel, and others. Furthermore, the accumulation of the biofuel within the organism itself often downregulates the pathway that produces it, decreasing production efficiency. The research group of Dunlop et al. sought to address this problem by engineering E. coli, introducing many different efflux pumps from different species of microbes to investigate whether or not the pumps would improve the organism’s ability to survive biofuel stress, and determine whether it would improve biofuel production [8].
In order to accomplish this the researchers constructed a database containing members of the hydrophobe/amphiphile efflux (HAE1) family of bacterial protein pumps, which contains all efflux pumps known to confer solvent resistance in gram negative bacteria. The team then conducted a bioinformatic analysis, searching for the proteins with similar sequences of their substrate-binding regions to the substrate-binding region of TtgB, a well-studied efflux pump known to be able to export solvents out of the cytoplasm. They cloned the operon of all identified efflux pumps into plasmid vectors, and transformed E. coli with one of each of these plasmids. The authors conducted a competition assay in order to identify useful efflux pumps. They did so by growing all E. coli strains together in a single culture, which also contained a significant amount of some biofuel, presuming that E. coli expressing efflux pumps that conferred resistance to that biofuel would come to dominate the population. After maintaining and rediluting that culture for four days, they quantified the relative concentrations of the plasmids in each culture, and found that certain efflux pumps can confer resistance to biofuels, and that (at least in the case of limonene production) the pump also improves the efficiency of biofuel production in a limonene production strain (Figure 4) [8].
References
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2013, Kenyon College.
