Microbiologically Influenced Corrosion in Industrial Systems
Introduction
Microbiologically influenced corrosion (MIC) is the degradation of materials, usually metal, due to the activity of microorganisms. It is also known as biocorrosion and this form of corrosion mostly takes place underneath biofilms, which consist of colonies of microorganisms attached to a surface[1]. MIC applies not only to metals but also to non-metallic surfaces such as concrete and can result in severe industry losses[1].
Mechanism
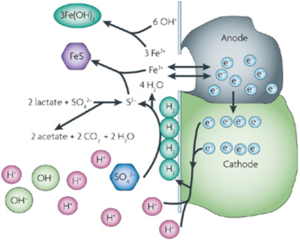
Corrosion, the oxidation of metal, requires the simultaneous reduction of another chemical. MIC usually takes place in stagnant water, and either dissolved oxygen or hydrogen is reduced in order for the process to proceed[1]. The area under the biofilm acts as the anode and the metal surface just outside the biofilm will become the cathode, where reduction takes place[2]. Metal dissolution will then occur under the biofilm. The electron flow from the metal anode to the terminal electron acceptor is facilitated by a series of both biotic and abiotic reactions through the biofilm[2]. This pathway of electron flow can occur because of electrochemical gradients that make it kinetically favorable[2].
Environment
The environment plays an important role in biocorrosion as environmental conditions can be responsible for either accelerating or inhibiting the corrosion that can result from various species of microorganisms. Generally, bacteria associated with MIC are found in seawater or stagnant water, and can function at a pH of 4 – 9 and at temperatures between 10 - 50oC[3]. Sulfate-reducing bacteria that cause corrosion damage to offshore steel structures are anaerobic while sulfur oxidizing bacteria responsible for biogenic sulfide corrosion require oxygen[2].
Microbial by-products
Microbes produce metabolic by-products that lead to changes in the properties of the metal–solution surface[4]. This affects the electrochemical behavior of the metal by adjusting environment conditions so that corrosion is favored[1]. One example would be the by-product iron(II) sulfide from sulfate-reducing bacteria, which has a strong cathodic effect and accelerates the rate of corrosion of iron[4]. Thus, the process of MIC is affected by the impact corrosion products have on the local environment[4].
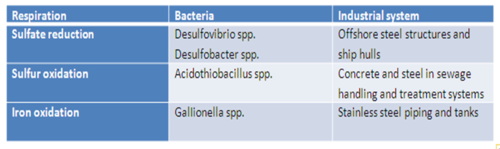
Associated Bacteria
The main types of bacteria associated with MIC of metals are sulfate-reducing bacteria, sulfur-oxidizing bacteria and iron-reducing bacteria[5].
Sulfate-Reducing Bacteria
Sulfate-reducing bacteria (SRB) such as Desulfovibrio desulfuricans have long been associated with the corrosion of metal exposed to sulfate-containing water, and have been the focus of much biocorrosion research because of the problem they pose to the offshore oil industry[5]. Under anaerobic conditions, SRB use sulfate as a terminal electron acceptor. A layer of molecular hydrogen is present on the metal surface, due to the reduction of hydrogen ions present in the water and the oxidation of the metal[5]. Sulfate-reducing bacteria oxidize the hydrogen via dissimilatory sulfate reduction, producing sulfide as a waste product. Sulfide reacts with metal ions present to produce metal sulfides, which accelerates the oxidation of the metal. It can also form hydrogen sulfide, which is used by sulfur-oxidizing bacteria, further contributing to corrosion[5].
Sulfur-Oxidizing Bacteria
Hydrogen sulfide can be oxidized by sulfur-oxidizing bacteria to form sulfuric acid, which corrodes steel and concrete in wastewater environments. Hydrogen sulfide gas can diffuse above anaerobic wastewater, and dissolve into water droplets on metal surfaces above the sewage[7]. With oxygen present, sulfur-oxidizing bacteria such as Acidithiobacillus thiooxidans can then metabolize the hydrogen sulfide gas to sulfuric acid, which has a destructive effect on exposed concrete and steel surfaces[7].
Iron-Oxidizing Bacteria
Iron-oxidizing bacteria such as Gallionella ferruginea oxidize soluble ferrous iron into an insoluble precipitate of ferric iron in the presence of oxygen[8]. They deposit iron oxides or hydroxides at a rate hundreds of times higher than the abiotic process, which creates conditions that are favorable for localized corrosion[9]. In the case of stainless steel, the deposits of iron hydroxides can overcome the passivation of stainless steel and initiate biocorrosion[9]. Iron-oxidizing bacteria initiate corrosion of stainless steel at an extremely high rate and MIC can penetrate a 3 mm thick stainless steel pipe in a month[10].
Industrial Impact
Microbiologically influenced corrosion usually occurs in aqueous environments such as water treatment and sewage plants, but is also common in other sectors of the economy, from oil and gas production systems to underground pipelines and chemical processing plants[11]. Corrosion costs the United States over 200 billion dollars per year and microbial corrosion often goes undetected in many industrial systems, resulting in significant waste and increased operating costs, which can lead to severe economic losses[12].
Treatment
Complete drainage and dry storage is usually an effective preventative measure against MIC[3]. For systems in use, regular mechanical cleaning, if possible, can be done to prevent the corrosive effects of biofilms[3]. Chemical treatments, mainly biocides, are also commonly used to kill the bacteria that cause corrosion[1]. However, biocides can pose a risk to human health or cause long-term damage to the environment and so other methods are being researched for dealing with MIC[1]. For example, the injection of nitrate compounds to stimulate nitrate-reducing activity is used to counteract the corrosive effects of sulfate metabolism in the petrochemical industry[6].
References
(1) Corrosion Technology Laboratories. Microbial Corrosion. Kennedy Space Center. Available at corrosion.ksc.nasa.gov/microbial.htm. Accessed October 30, 2013.
(2) Beech IB, Sunner JA, Hiraoka K. Microbe-surface interactions in biofouling and biocorrosion processes. International microbiology. 2005;8:157.
(3) Corrosion Consulting Services. Microbiologically Influenced Corrosion. WebCorr Corrosion Consulting Services. Available at corrosionclinic.com/types_of_corrosion/microbiologically_influenced_biological_microbial_corrosion.htm. Accessed October 30, 2013.
(4) Videla HA, Herrera LK. Microbiologically influenced corrosion: looking to the future. International microbiology. 2005;8:169.
(5) Beech IB, Sunner J. Biocorrosion: towards understanding interactions between biofilms and metals. Current Opinion in Biotechnology. 2004;15:181-186.
(6) Muyzer G, Stams AJM. The ecology and biotechnology of sulphate-reducing bacteria. Nature Reviews Microbiology. 2008;6:441-454.
(7) Hardy JA, Bown JL. The Corrosion of Mild Steel by Biogenic Sulfide Films Exposed to Air. Corrosion. 1984;40:650.
(8) Hallbeck L, Pedersen K. Culture parameters regulating stalk formation and growth rate of Gallionella ferruginea. Journal of General Microbiology. 1990;136:1675.
(9) Starosvetsky D, Starosvetsky J, Pokroy B, Hilel T, Armon R. Electrochemical behaviour of stainless steels in media containing iron-oxidizing bacteria (IOB) by corrosion process modeling. Corrosion Science. 2008;50:540-547.
(10) Chamritski I, Laycock N, Webster B. Corrosion by Iron Oxidizing Bacteria: Model Development and Field Studies. 2001.
(11) Lee AK, Newman DK. Microbial iron respiration: impacts on corrosion processes. Applied Microbiology and Biotechnology. 2003;62:134-139.
(12) Gadd GM. Metals, minerals and microbes: geomicrobiology and bioremediation. Microbiology. 2010;156:609-643.