Mycobacterium leprae -- Leprosy


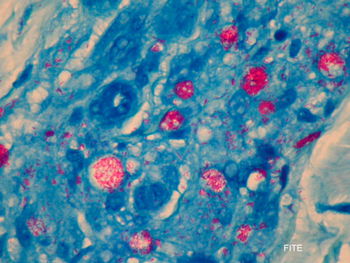

Etiology/Bacteriology
Taxonomy
| Domain = Bacteria | Phylum = Actinobacteria | Class = Actinobacteridae | Order = Actinomycetales | Suborder = Corynebacterineae | Family = Mycobacteriaceae | Genus = Mycobacterium | Species = M. leprae |
|
NCBI: Taxonomy Genome: Mycobacterium leprae |
Description
Mycobacterium leprae is a microaerophilic, acid-fast bacillus which causes leprosy. Since Mycobacterium leprae cannot easily be cultured in the lab, much is unknown about the infectious dose, incubation, and transmission of the disease [7]. The infection is thought to be spread through the skin and nasal mucosa. Humans and armadillos are the only known carriers of the disease, though there is some speculation about the possible role of insects in transmission [8]. Mycobacterium leprae colonizes the Schwann cells of the peripheral nervous system and can also live and grow within macrophages as a way to evade the host immune system [9]. The bacteria express many virulence factors that allow for invasion of the nerve cells and nutritional access [7]. The symptoms of leprosy are associated with the degree of host immune system response to the infection, so the disease is classified according to these varied responses. Polar tuberculoid (TT) leprosy results from the immune system trying to kill the pathogen. This is in contrast to lepromatous (LL) which is marked by no host resistance and thus direct killing of self-cells through the immune response. Borderline categories fall between the two polar forms of the disease. Reactions can then be divided into two types based on clinical manifestations. [10]. Biopsy from a lesion revealing acid-fast bacilli is necessary for diagnosis [7]. The standard treatment for leprosy is a Multi-drug Treatment (MDT) that consists of steroids and antibiotics [8]. Through prevention steps and treatment, the World Health Organization is attempting to eradicate the disease, and has so far succeeded in decreasing the total number of cases. However, the number of new cases each year remains the same despite some positive results from the M. bovis BCG vaccine. Due to the intracellular lifestyle of the bacteria, Mycobacterium leprae infection is associated with symptoms that are the result of the immune system attacking host cells. The bacteria elicit an immune response on nerve cells and other immune cells, causing inflammation and neuropathy [7].
Pathogenesis
Transmission
Although much about the transmission of Mycobacterium leprae is unknown, prolonged contact with an infected person increases an individual's chance of becoming infected. Armadillos can harbor the bacteria, but are not seen as a threat to human contraction of the disease. In addition, insects could be possible carriers of Mycobacterium leprae but this is unclear In humans, the bacteria is thought to be passed through skin and nasal mucosa [8]. One study has demonstrated that large numbers of the bacteria can be found on the skin of infected persons, providing a possible means of transmission [11]. Mycobacterium leprae could also be passed through nasal mucosa like the closely related Mycobacterium tuberculosis [9].
Infectious dose, incubation, and colonization
Mycobacterium leprae is not easily cultured in the lab, which can hinder studies of infectious dose and incubation, however some sources provide estimates for these categories [7]. With a doubling time of 14 days [12], Mycobacterium leprae has the longest doubling time of any studied bacteria [9]. The World Health Organization states that Mycobacterium leprae has an incubation period of an average 5 years. Humans and armadillos are currently the only known reservoirs of the bacteria, with infected humans accounting for up to 7 billion organisms per gram of tissue [8]. Mycobacterium leprae mostly lives in the extremities and facial region within macrophages and Schwann cells of the peripheral nervous system [9].
Epidemiology
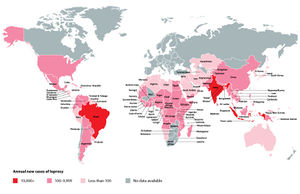
Mycobacterium leprae is thought to have originated in East Africa and spread across the globe through human migratory trends, reaching the Western world within the last 500 years [13]. In 2012, the World Health Organization recorded a prevalence of approximately 180,000 cases [8]. Through eradication efforts, the total number of cases worldwide has decreased, yet the number of new cases each year has remained consistent [7]. Mortality is difficult to measure with leprosy, as the infection is not the immediate cause of death in many cases [8].
Virulence factors
Iron utilization
Although some genes from the closely related Mycobacterium tuberculosis have been deleted in Mycobacterium leprae, some iron utilization genes have been conserved to help the pathogen acquire nutrients for growth [9]. NRAMP proteins, which are coded by one particular conserved gene, allow transportation of iron into the macrophage for survival [14].
Waxy exterior
Bacteria of the Mycobacterium Genus are defined by their waxy exterior coat. In Mycobacterium leprae, the exterior allows for intake into the macrophage and into some dendritic cells, in which it can survive. The terminal mannose caps on the waxy mycobacterial ligand, lipoarabinomannan, of the pathogen are recognized by the PRR of the macrophage to allow for phagocytosis [15].
Macrophage invasion
Mycobacterium leprae survives and replicates in macrophages, dividing to approximately 100 organisms per cell [16]. The bacteria prevent phagosome and lysosome fusion to avoid degradation [17]. In the event that the bacteria are absorbed into the phagolysosome, Mycobacterium leprae has the ability to survive emission of reactive oxygen species [7].
Schwann cell invasion
The major target of Mycobacterium leprae is the Schwann cell [11]. The optimal temperature of the bacteria corresponds to the temperature in the peripheral nerves [18]. To access the cells, Mycobacterium leprae gets into the lymphatic system and the blood vessels [19]. Once in the area, Mycobacterium leprae binds to the Schwann cell via laminin-binding protein [9]. The bacteria are thought to then enter through the vascular epithelium into the cell [7]. The infection remains localized to the peripheral nervous system by rolling and binding to exposed Schwann cells [20].
Drug resistance
Mycobacterium leprae has many mechanisms of drug resistance to allow it to continue to survive despite antimicrobial presence [7].
Clinical features
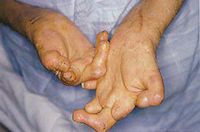
Classification
Leprosy presents with a variety of clinical features based primary on the degree to which the host immune system responds to the infection [7]. In general, the disease leprosy is a result of the immune response to Mycobcaterium leprae invasion of the Schwann cells, which leads to demyelination neuropathy [21]. Common results of the immune response are loss of sensation and disfiguration [9]. Chronic inflammation can lead to this paralysis, specifically along the periphery, and can also affect facial regions, contributing to factors such as blindness [7]. Classification of leprosy is based of a five part system which divides patients by immune response. There are two polar categories and three borderline types which fall between the polar classifications. One polar type is called tuberculoid (TT). This type is marked by a high level of cellular immune response, delayed hypersensitivity, and bacilli in biopsied tissue. The other extreme is called lepromatous (LL). In this type of leprosy, there is no host resistance. Nodular lesions will form all over body, and when biopsied, they reveal globi, or bundles of bacilli. The remaining three types are borderline categories, being closer to one polar type or the other, or having marks of both polar types. These are borderline lepromatous (BL), mid-borderline (BB), and borderline tuberculoid (BT) [10].
Clinical manifestations can be divided into two major categories: Type 1 and Type 2 reactions.
Type 1
Borderline leprosy types (BL, BB, and BT) are included in this classification. The symptoms include lesions that are red and hardened, fluid accumulation in the periphery, sensory loss, and neuropathy. Symptoms develop gradually and may last for several weeks [7].
Type 2
Erythema nodosum leprosum, or Type 2 reactions, affect multibacillary patients (LL and BL). In contrast to Type 1 reactions, symptoms develop suddenly, subside after a couple weeks, and may reoccur many times over a period of months. Tender, red lesions erupt on the face, extremities, and trunk. These patients may develop inflammation of the eyes leading to blindness and other complications. General muscle inflammation, joint inflammation, and orchitis may occur [7].
Lucio Phenomenon
The Lucio Phenomenon is a rare form of symptoms that describes a rapid onset of inflammation of the blood vessel walls and anemia. This rarity occurs primarily in Mexican leprosy patients [22].
Diagnosis
Diagnosis is typically made upon recognition of acid-fast bacilli in a skin biopsy of a lesion [7]. As some patients have few lesions, scientists are looking for immunodiagnostic tests to explain neuropathy and other symptoms that may be unaccompanied by lesions [9]. For instance, tuberculoid leprosy (TT) often produces few lesions, so the disease can misdiagnosed. In lepromatous (LL) cases, biopsy should be made from a nerve cell to rule out alternative diagnoses which might show similar symptoms and bacilli in tissue [7].
Treatment

The current standard treatment for Mycobacterium leprae infection is a Multi-drug Treatment (MDT) which consists of corticosteroids and antimicrobials. For paucibacillary leprosy, rifampicin and dapsone are used, while rifampicin, clofazimine, and dapsone are used in multibacillary leprosy. Rifampicin, ofloxacin, and minocycline can be combined in single lesion paucibacillary leprosy. Oral prednisolone can be used in secondary complications such as neuropathy and eye problems. Drops can be used to dilate the eye and stimulate relaxation to help the healing process [8]. However, scientists are discovering drug-resistant strains of Mycobacterium leprae, so precautions must be taken [7]. Minocycline or ofloxacin can be used in the event of a rifampicin allergy, resistance, or presence of a disease antagonistic to rifampicin. Interestingly, the disease can sometimes be self-limiting and cure itself independently of drug treatment [8].
Prevention
Vaccination
A proper vaccination for Mycobacterium leprae would provide protective immunity. One vaccine has shown positive results in protecting the inoculated person from infection. The M. bovis BCG vaccine proved effective in India [23] and Brazil [24].
Eradication attempts
Global efforts to eradicate leprosy has resulted in elimination of the disease from 119 of 122 countries thought to be highly affected [8]. With proper prevention measures, early detection, and treatment, numbers of new cases would decrease and those already infected could be cured [9].
Immune Response
Protective immunity and susceptibility
Science has discovered that over 95 percent of people could be immune to leprosy, due to protection and exposure [7]. However, some populations may be more susceptible to the disease based on genetic factors. The PARK2/PACRG gene could contribute to susceptibility to Mycobacterium leprae infection [25]. In addition, the Vitamin D receptor (VDR) gene is associated with both polar types of the disease [26]. Innate immunity can fail in an instance of susceptibility, so the adaptive immune response works to eradicate the infection through T cells [7].
Host defense
The symptoms associated with Mycobacterium leprae infection, such as lesions and neuropathy, are typically results of the host attacking its own nerve cells through an immune response. While the bacteria can trigger apoptosis in the cells, the body also contributes to cell death by mounting an immune response on the infected cells. Dendritic cells phagocytose the pathogen to stimulate the adaptive immune response through T cells [7]. In tuberculoid leprosy, CD4 T cells are most prevalent, whereas CD8 cytotoxic T cells are in higher numbers in lepromatous leprosy [28]. The T cells, specifically the cytotoxic cells, kill the infected Schwann cells, which results in sensory loss [27]. As well, infected Schwann cells and macrophages can be attacked by NK cells [29], [30]. Toll-like receptors (TLRs) also have an important function in the immune response, but specifically for distinguishing between polar types of the disease. Tissue biopsies showed that tuberculoid patients produced stronger TLRs on leukocytes than the lepromatous type, suggesting that TLRs significantly affect the ability of the immune system to recognize the bacteria to avoid killing host cells [31]. TLRs are also associated with the cytokine TNF-α which causes tissue damage in leprosy [32]. TNF-α is produced by macrophages, and is used to stimulate T cell mediated immunity, which causes many symptoms of the disease [7]. This suggests that tuberculoid leprosy (TT) may share similar symptoms to lepromatous leprosy (LL) despite varied immune responses due to the effects of cytokines. The immune system also can work to deter bacteria from adhering to the nerve cells using antibodies directed at the outer coat of Mycobacterium leprae cells [33].
Bacterial evasion
Many of the aforementioned virulence factors of Mycobacterium leprae are associated with the ability to evade the host immune response, such as living and surviving within macrophages through various mechanisms. The bacteria also suppress the immune system by directly affecting the dendritic cells. T cell mediated immune response can be halted by preventing the interaction of dendritic cells and T cells, which Mycobacterium leprae accomplishes by causing the dendritic cell to express PGL-1 on its surface. This protein appears to alter dendritic cell and T cell matching, and thus slows the immune attack on the bacteria [34]. The bacteria can also stop dendritic cell maturation by preventing IL-2 production, further disabling the immune defenses [35].
References
1 University of Oklahoma Education Abroad.
2 Bhuchar, S., Chan, C.S., Mir, M.R., Subrt, P., Tschen, J.A., Collins, W.E., and Sylvia Hsu. "Clinical pearl for the diagnosis of Hansen disease." Dermatology Online Journal 15 (12):7. 2009.
3 "About us." The Leprosy Mission International.
4 "What is leprosy?" The Leprosy Mission International.
5 "Biology at Davidson." Davidson College.
6 "Photo Gallery." St. Luke's Leprosarium.
7 Scollard, D.M., Adams, L.B., Gillis, T.P., Krahenbuhl, J.L., Truman, R.W., and D.L. Williams. "The Continuing Challenges of Leprosy." Clinical Microbiology Reviews 19 (2). 2006.
8 "Leprosy Today." World Health Organization. 2013.
9 Cole, S.T., Eiglmeier, K., Parkhill, J., James, K.D., Thomson, N.R., Wheeler, P.R., Honoré, N., Garnier, T., Churcher, C., Harris, D., Mungall, K., Basham, D., Brown, D., Chillingworth, T., Connor, R., Davies, R.M., Devlin, K., Duthoy, S., Feltwell, T., Fraser, A., Hamlin, N., Holroyd, S., Hornsby, T., Jagels, K., Lacroix, C., Maclean, J., Moule, S., Murphy, L., Oliver, K., Quail, M.A., Rajandream, M.-A., Rutherford, K.M., Rutter, S., Seeger, K., Simon, S., Simmonds, M., Skelton, J., Squares, R., Squares, S., Stevens, K., Taylor, K., Whitehead, S., Woodward, J.R., and B.G. Barrell. "Massive gene decay in the leprosy bacillus." Nature. 409. 1007-1011. 2001, February 22.
10 Ridley, D. S., and W. H. Jopling. 1966. Classification of leprosy according to immunity—a five-group system. International Journal of Leprosy. 34. 255-273.
11 Job, C. K. "Nerve damage in leprosy." Int. J. Lepr. Other Mycobact. Dis. 57. 532-539. 1989.
12 Shepard, C. C. 1960. "The experimental disease that follows the injection of human leprosy bacilli into footpads of mice." J. Exp. Med. 112. 445-454.
13 Monot, M., Honore, N., Garnier, T., Araoz, R., Coppee, J.Y., Lacroix, C., Sow, S., Spencer, J.S., Truman, R.W., Williams, D.L., Gelber, R., Virmond, M., Flageul,B., Cho, S.N., Ji, B., Paniz-Mondolfi, A., Convit, J., Young, S., Fine, P.E., Rasolofo, V., Brennan, P.J., and S. T. Cole. 2005. "On the origin of leprosy." Science. 308. 1040-1042.
14 Gruenheid, S., and P. Gros. "Genetic susceptibility to intracellular infections: Nramp1, macrophage function and divalent cations transport." Curr. Opin. Microbiol. 3:43-48. 2000.
15 Brennan, P. J., and V. D. Vissa. "Genomic evidence for the retention of the essential mycobacterial cell wall in the otherwise defective Mycobacterium leprae." Lepr. Rev. 72. 415-428. 2001.
16 Hagge, D., Robinson, S.O., Scollard, D., McCormick, G., and D. L. Williams. "A new model for studying the effects of Mycobacterium leprae on Schwann cell and neuron interactions." J. Infect. Dis. 186. 1283-1296. 2002.
17 Sibley, L.D., Franzblau, S.G., and J. L. Krahenbuhl. "Intracellular fate of Mycobacterium leprae in normal and activated mouse macrophages." Infect. Immun. 55. 680-685. 1987.
18 Truman, R. W., and J. L. Krahenbuhl. "Viable M. leprae as a research reagent." Int. J. Lepr. Other Mycobact. Dis. 69. 1-12. 2001.
19 Scollard, D. M. "Endothelial cells and the pathogenesis of lepromatous neuritis: insights from the armadillo model." Microbes Infect. 2. 1835-1843. 2000.
20 Khanolkar, V. R. "Pathology of leprosy." In R. G. Cochrane and T. F. Davey (ed.), Leprosy in theory and practice. 125 -151. 1964.
21 Swift, T. R. "Peripheral nerve involvement in leprosy: quantitative histologic aspects." Acta Neuropathol. 29. 1-8. 1974.
22 Donner, R. S., and J. A. Shively. "The 'Lucio phenomenon' in diffuse leprosy." Ann. Intern. Med. 67. 831-836. 1967.
23 Zodpey, S.P., Ambadekar, N.N., and A. Thakur. "Effectiveness of bacillus Calmette Guerin (BCG) vaccination in the prevention of leprosy: a population-based case-control study in Yavatmal District, India." Public Health 119. 209-216. 2005.
24 Cunha, S.S., Rodrigues, L.C., Pedrosa, V., Dourado, I.M., Barreto, M.L., and S. M. Pereira. "Neonatal BCG protection against leprosy: a study in Manaus, Brazilian Amazon." Lepr. Rev. 75. 357-366. 2004.
25 Mira, M.T., Alcais, A., Nguyen, V.T., Moraes, M.O., Di Flumeri, C., Vu, H.T., Mai, C.P., Nguyen, T.H., Nguyen, N.B., Pham, X.K., Sarno, E.N., Alter, A., Montpetit, A., Moraes, M.E., Moraes, J.R., Dore, C., Gallant, C.J., Lepage, P., Verner, A., Van De Vosse, E., Hudson, T.J., Abel, L., and E. Schurr. "Susceptibility to leprosy is associated with PARK2 and PACRG." Nature 427. 636-640. 2004.
26 Roy, S., Frodsham, A., Saha, B., Hazra, S.K., Mascie-Taylor, C.G.N., and A. V. S. Hill. "Association of vitamin D receptor genotype with leprosy type." J. Infect. Dis. 179. 187-191. 1999.
27 Spierings, E., De Boer, T., Zulianello, L., and T. H. Ottenhoff. "Novel mechanisms in the immunopathogenesis of leprosy nerve damage: the role of Schwann cells, T cells and Mycobacterium leprae." Immunol. Cell Biol. 78. 349-355. 2000.
28 Modlin, R.L., Melancon-Kaplan, J., Young, S.M.M., Pirmez, C., Kino, H., Convit, J., Rea, T.H., and B.R. Bloom. "Learning from lesions: patterns of tissue inflammation in leprosy." Proc. Natl. Acad. Sci. USA 85. 1213-1217. 1988.
29 Steinhoff, U., Wand-Wurttenberger, A. Bremerich, A., and S.H.E. Kaufmann. "Mycobacterium leprae renders Schwann cells and mononuclear phagocytes susceptible or resistant to killer cells." Infect. Immun. 59. 684-688. 1991.
30 Chiplunkar, S.V., Deshmukh, M.V., Samson, P.D., Butlin, R., Bhatki, W.S., Chulawalla, R.G., Deo, M.G., and S.G. Gangal. "Natural killer-cell-mediated and antibody-dependent cellular cytotoxicity in leprosy." Int. J. Lepr. Other Mycobact. Dis. 58. 334-341. 1990.
31 Bochud, P.Y., Hawn, T.R., and A. Aderem. "Cutting edge: a Toll-like receptor 2 polymorphism that is associated with lepromatous leprosy is unable to mediate mycobacterial signaling." J. Immunol. 170. 3451-3454. 2003.
32 Underhill, D.M., Ozinsku, A., Smith, K.D., and A. Aderem. "Toll-like receptor-2 mediates mycobacteria-induced proinflammatory signals in macrophages." Proc. Natl. Acad. Sci. USA 96:14459-14463. 1999.
33 Choudhury, A., Mistry, N.F., and N.H. Antia. "Blocking of Mycobacterium leprae adherence to dissociated Schwann cells by anti-mycobacterial antibodies." Scand. J. Immunol. 30. 505-509. 1989.
34 Hashimoto, K., Maeda, Y., Kimura, H., Suzuki, K., Masuda, A., Matsuoka, M., and M. Makino. "Mycobacterium leprae infection in monocyte-derived dendritic cells and its influence on antigen-presenting function." Infect. Immun. 70. 5167-5176. 2002.
35 Nigou, J., Zelle-Rieser, C., Gilleron, M., Thurnher, M., and G. Puzo. "Mannosylated lipoarabinomannans inhibit IL-12 production by human dendritic cells: evidence for a negative signal delivered through the mannose receptor." J. Immunol. 166. 7477-8485. 2001.
Created by Gracen Conway, student of Tyrrell Conway at the University of Oklahoma.
