Mycoplasma
A Microbial Biorealm page on the genus Mycoplasma
Classification
Higher order taxa:
Bacteria; Firmicutes; Mollicutes; Mycoplasmatales; Mycoplasmataceae; Mycoplasma
Species:
Mycoplasma genitalium, M. pneumoniae, M. pulmonis
Description and Significance
The study of Mycoplasma has become important in the understanding of chronic diseases. As both an extracellular and intracellular pathogen, a better understanding of the virulence mechanisms of mycoplasma will provide fresh understanding of how to diagnose and combat this pathogen.
Genome Structure
There are currently 8 complete genome sequences, and 4 that are in progress. (click [Organism:exp here] for the NCBI genome list for Mycoplasma). Mycoplasma genitalium is thought to have the smallest genome of any self-replicating organism, measuring only 580,070 bp long with just 470 open reading frames. Its overall G+C content is 32%. The regions of lowest G+C content are around the origin of replication. The genome of Mycoplasma pneumoniae is much longer at 816 kbp, containing 209 open reading frames in the additional 236 kbp, giving it a total of 679 open-reading frames. Mycoplasma hyopneumoniae 232 is 892,758 bp long with G+C content of 28.6 mol%. It has 53 ORFs and is the largest family is the 34-member ABC transporter family. Mycoplasma mobile 163K is 777,079 bp long with 517 ORFs and has one of the lowest DNA G+C contents of 24.9% and most reduced set of tRNAs of any organism yet reported. Mycoplasma mycoides subsp. mycoides SC str. PG1 is 1,211,703 bp long with with the lowest G+C content (24 %) and the highest density of insertion sequences (13% of the genome size)of all sequenced bacterial genomes. This organism causes contagious bovine pleuropneumonia (CBPP), a severe respiratory disease in cattle that spreads very quickly, especially in developing countries. Mycoplasma penetrans HF-2 is 1,358,633 bp long with a average G + C content of 25.7%. This organism is known to infect people with HIV-1 infection, where it causes even more damage to the immune system. Mycoplasma pulmonis is 963,879 bp long with a G + C content of 26.6% that causes murine respiratory mycoplasmosis (MRM), and can cause genital infections. Mycoplasma gallisepticum R is 996,422 bp long with an overall G+C content of 31%, it causes chronic respiratory disease in chickens and other avian species.
The phylogeny of mollicutes is interesting as regards the degeneration of the genome. During their evolutionary history multiple reductions in genome size have occurred, the usual genetic code has been altered, and the overall rate of evolution uncharacteristically high. One suggested reason for the reduction in genome size is the evolution of mollicutes into strict parasites making much of their metabolic machinery obsolete. Mycoplasma also have unique use of the amino acid codon UGA, which they use as an additional codon for tryptophan, while other organisms use it as a stop codon.
In sequencing these genomes researchers hope to increase understanding of the pathophysiology of the pathogen, by identifying virulence factors. It will also be useful in the identification of protective antigens in order to develop a vaccine to combat the parasite. It will also help in forming the definition of a minimal cell, since the genomes of many Mycoplasma are stripped down to the bare bones.
Cell Structure and Metabolism
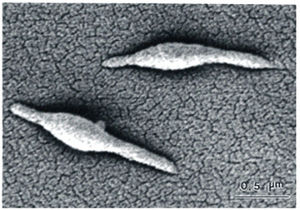
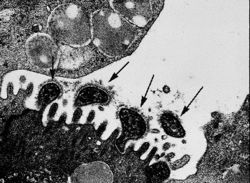
Mycoplasma are flask-shaped and are most likely descended from Gram-positive bacteria. Due to their seriously degraded genome they cannot perform many metabolic functions, such as cell wall production or synthesis of purines. As such stripped down organisms they are considered the perfect model of the minimalist cell. Meaning they are believed to contain the absolute minimum machinery necessary for survival and are considered the model organisms for the essential functions of all living cells. The Mycoplasma cell is built of a minimum set of organelles including a plasma membrane, ribosomes, and a highly coiled circular chromosome.
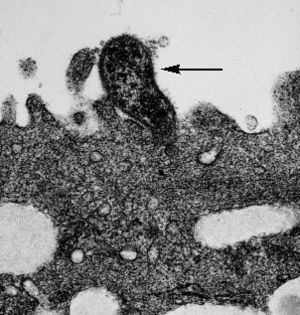
Because it is essential for Mycoplasma to be able to bind to their host cells they have developed special tip organelles for this purpose, with a significant percentage of genes in their genome devoted to this important function. Most species of Mycoplasma are extracellular pathogens, and use their tip organelle, which has a high concentration of adhesins, to attach to the eukaryotic cell. The organelle is bounded by the cell membrane and contains a central rod-shaped structure. Mycoplasma that act as intracellular pathogens, penetrate their host cell using their tip organelle. The lack of a rigid cell wall, may also help facilitate contact between mycoplasma and their host cell, by creating the possibility of fusion between the two membranes. This would enable the exchange of membrane and cytoplasmic components.
Ecology

There are no free-living Mycoplasma, they are strictly parasites. They parasitize a wide range of organism including humans, plants, animals, and insects. Mycoplasma grow very slowly, even under perfect conditions, with a generation time ranging up to nine hours in some species. They also have a very long lag phase, so it may take an entire week before colonies become visible on agar plates. Due to their degraded genome, and inability to perform basic functions, Mycoplasma rely on their host for much of their nutrition.
Many species of Mycoplasma are commensal, living innocuously with their host as part of the natural flora of the body, and having no detrimental effects. But when Mycoplasma act as pathogens they cause chronic but mild infections that rarely, if ever, kill their host. They are usually surface parasites, although several species, including the aptly named M. penetrans, are intracellular pathogens.
Pathology

Mycoplasma have been linked with several chronic diseases, including chronic fatigue syndrome, fibromyalgia syndrome, gulf war syndrome, and rheumatoid arthritis. There is a definite correlation between these diseases and mycoplasmal infections, although the connection remains obscure. In patients with chronic fatigue syndrome and fibromyalgia syndrome Mycoplasma sp. were found in 62.9% and 50% of patients respectively in a study done by Nasralla et al. They also found that more than 50% of patients with rheumatoid arthritis had mycoplasmal infections, and 36% of these patients had multiple infections by different species of Mycoplasma. In studies with Gulf War illness, about 50% of patients had mycoplasmal infections, leading to the rumor that the Gulf War was the first war in which biological weapons were used. Namely weapons with biologically engineered strains of Mycoplasma (this is still just a rumor).
The most common symptoms associated with mycoplasmal infections include night sweats, intermittent fevers, chronic fatigue, skin rashes, increased dermal sensitivity, joint and muscle pain, swelling and reduced mobility of joints, heart palpitations, pain and arrhythmia, stomach cramps and regurgitation, loss of vision, double vision, and the list can go on. Although there is a clear link between Mycoplasma infection and the above-mentioned diseases, it is unclear which is the precursor. Mycoplasma may cause these various diseases, or it may be an opportunistic pathogen that colonizes a host with a weak immune system, and causes a secondary infection.
References
Coyle, Michael. "Advanced Applied Microscopy for Nutritional Evaluation and Correction", Elbow Room Publishing, 2000.
Drexler, Hans G. & Cord C. Uphoff. 2002. Mycoplasma contamination of cell cultures: Incidence, sources, effects, detection, elimination, prevention. Cytotechnology, 39: 75-90.

