Nematocida parisii
By: Amanda He
Introduction to Microsporidia
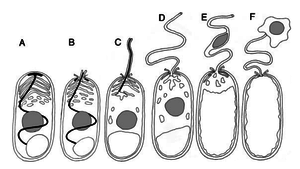
A) Shows a dormant spore containing a polar filament (black), nucleus (gray), polaroplast and posterior vacuole.
B) The posterior vacuole swells with water and ruptures the anchoring disk, allowing the polar filament to emerge through through spore cell wall.
C) Polar filament continues to to outward and evert.
D) Polar filament is fully everted and becomes a polar tube. Sporoplasm is squeezed into the polar tube.
E) Sporoplasm is moving through the polar tube.
F) Entire sporoplasm emerges from the polar tube while bound to the new membrane
Image source: http://www.annualreviews.org/doi/pdf/10.1146/annurev.micro.56.012302.160854 [1]
Microsporidia is a phylum of eukaryotic, intracellular, spore-forming parasites [1]. Fungi are currently considered the closest relative to this phylum, but it is still being debated whether or not they should be categorized as Fungi [1,2, 3]. Interest in microsporidia research began from the diagnosis of an intestinal infection called microsporidiosis. Microsporidiosis, as indicated by the name, is caused by microsporidia, which induce severe, persistent diarrhea in humans. The disease is found in immunocompromised individuals such as people with HIV and in individuals who live in locations without access to clean water [1,6]. Since microsporidia are intracellular pathogens, they are able to avoid detection by immune receptors.
Aside from humans, microsporidia are able to use a huge range of vertebrates, invertebrates, and protists as hosts [6]. They are found most prevalently in arthropods and fish [1]. The life cycles of microsporidia vary within each species, but all species have at least one intracellular and extracellular spore stage [3]. Microsporidian spores range in size from 1 μm to 40 μm and vary in shape from spherical to rod-shaped to crescent-shaped [1]. The majority of the species have an ovoid spore. Microsporidia are able to survive outside of their hosts as spores [6]. Microsporidian spores are characterized by the thick chitinous cell wall surrounding their cell membrane and the presence of a polar filament inside the spore [3]. Polar filaments are tubes located within the spore, attached to the apex of the spore by an anchoring disk [1]. These polar filaments are an essential component to allow for spore germination and infection of the host cell.
Spore germination in microsporidian spores occurs in several phases: activation, increase in intrasporal osmotic pressure, eversion of the polar tube, and passage of sporoplasm through the polar tube [2]. Microsporidia have to be stimulated by an external environmental trigger such as change in pH, presence of ions, or ultraviolet radiation exposure. Since microsporidia live in a wide range of hosts, each species requires a different activation stimulation. In response to the activation, all microsporidia respond by increasing flow of water into spore, increasing intrasporal osmotic pressure and swelling the polaroplast [2]. The pressure build-up within the spore becomes the driving force of germination as it forces the eversion of the polar tube [1]. In eversion, the discharging polar filament breaks through the thinnest portion of the spore cell wall (Figure 1B), where it serves as a polar tube (Figure 1D). The polar tube is used as a bridge to siphon the sporoplasm out of the spore and into its host cell [2]. However, it has yet to be determined how exactly the polar tube or sporoplasm interacts with the host cell’s membrane. It is a predicted possibility that the polar tube launches and pierces the cell membrane of a nearby host cell and injects the sporoplasm, allowing it to avoid all extracellular defenses [1,2,7].
Once the sporoplasm of the microsporidia spore is in the host cell, it replicates into a meront, which is a cell consisting of organelles and sporoplasm enclosed by a cell membrane, but no cell wall [6]. Meronts differentiate and develops into the spore form. Once the meronts complete development, they will begin producing more spores, which exit the host cell and repeat the cycle of infection [5].
Microsporidia have mitochondria, which are used for their metabolism. However, they do not have electron transfer chains, oxidative phosphorylation, and the tricarboxylic acid (TCA) cycle [1]. Instead of these pathways, microsporidia break down glucose with glycolysis and followed by substrate-level phosphorylation. The resulting pyruvate from glycolysis is decarboxylated by either pyruvate dehydrogenase complex (PDHC) or the enzyme, pyruvate:ferredoxin oxidoreductase in anaerobic microsporidia [1]. Electrons are removed from pyruvate and transferred to ferredoxin, which reduces NAD+ to NADH. Finally the NADH donate their electrons to an organic terminal electron acceptor. The lack of TCA cycle induces a requirement for ATP, so the microsporidia also conduct fermentation to synthesize ATP and also have enzymes found in pentose-phosphate pathway (PPP) [1].
Nematocida parisii Cell Shape, Components, and Metabolism
Microsporidia have a vast range of spore shapes. For N. parisii, they have rod-shaped spores, but they are not always rod-shaped. N. parisii have two stages of meront types, early stage and late stage. Early stage meronts have irregularly shaped cell membranes, while late stage meronts are more developed and have a more regular shaped membrane, resembling a standard rod.
The N. parisii spore has a thick spore coat composed of an electron-dense, protein exospore and an endospore made of chitin and protein [8]. The endospore has the same thickness at all parts of the surface, except for the apex of the spore, where it is thinner. It is thinner because this is the area where the polar filament will be rupturing through when launching at an intestinal host cell [1]. N. parisii spore cells have a modification in the spore coat that allows it to adhere to the gastrointestinal tract, helping facilitate infection of host cells [8]. Like the standard microsporidia spore, the sporoplasm of N. parisii contains a polaroplast (a lamella of membranes), nucleus, and a polar filament wrapped from the nucleus with one end connected to the posterior vacuole and the other end connected to the anchoring disk at the apex of spore [1].
Image source: http://www.plospathogens.org/article/fetchObject.action?uri=info%3Adoi%2F10.1371%2Fjournal.ppat.1002227&representation=PDF [7]
However, N. parisii also has many differences from a standard microsporidia spore. In addition to its variation in cell membrane, N. parisii also vary in their cell size, polar tube number, nuclei number, etc. N. parisii cells have been reported to have general two different sizes, cells that are 2.18 ± 0.15 µm long and 0.8 ± 0.08 µm wide or cells that are 3.17 ± 0.22 µm long and 1.31 ± 0.15 µm wide [6]. The smaller spores only have one polar tube coil, while the larger spores can have up to five polar tube coils [6].
Like many other microsporidia species, N. parisii has multiple nuclei in its single cell. However, unlike other microsporidia, the nuclei are not dikaryotic, paired up nuclei. In all stages of meront development, N. parisii appears to be monokaryotic, unpaired nuclei. It is possible that the monokaryotic nuclei are used for a rare sexual cycle in N. parisii. In Cuomo, et al., 2012, the laboratory members hypothesize that the nuclei can fuse together and then reduce the number of chromosomes by undergoing either meiosis (found in conventional sexual cycle) or by chromosome loss (found in parasexual cycle) [5]. The sexual cycle in N. parisii would allow it to repair mutations or increase the number of mutations for virulent purposes.
Microsporidia are known for having a reduced metabolic capability. N. parisii genomes do not encode for components of the TCA cycle pathway or for oxidative phosphorylation pathway [5]. N. parisii encodes components of glycolysis and the PPP such as hexokinase. Hexokinase is an enzyme that catalyzes the conversion of glucose to glucose-6-phosphate [6]. Glucose-6-phosphate is able to be utilized in glycolysis and in pentose-phosphate pathway (PPP) [9]. N. parisii, like other microsporidia, have limited abilities when it comes to biosynthesis and degradation of molecules. N. parisii lack many of the biosynthetic capabilities that other organisms have, which force them to be extremely dependent on the host cell to provide it with nucleotides, amino acids, and lipids [5]. In order to provide the nucleotides, host cells have to be switched from a catabolic metabolism to an anabolic metabolism similar to the Warburg effect [5]. The Warburg effect is when cells produce energy rapidly through glycolysis to help meet the pathogen’s demands for nucleotides, amino acids, and lipids, first discovered in cancer cells [5]. N. parisii produces hexokinase secretion signals that promotes encourage this anabolic metabolism. From the host cell’s use of PPP, the meront is provided with lipid building blocks and nucleotides. The meront utilizes the host cell’s mitochondria to help it break down pyruvate with its TCA cycle to obtain amino acid building blocks and additional lipid building blocks [5].
Background on Interactions between C. elegans and Nematocida parisii
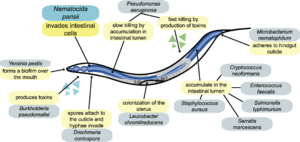
Image source: http://www.plosbiology.org/article/fetchObject.action?uri=info%3Adoi%2F10.1371%2Fjournal.pbio.1000005&representation=PDF [3]
Caenorhabditis elegans are model organisms for many different fields of biology, ranging from behavioral to neurobiological research. Over time, C. elegans has become a more and more popular host for studying pathogenesis as a result of the discovery of intracellular pathogens. In Franconville, France, a community outside of Paris, a wild-caught C. elegans isolated from a compost bin was discovered to have the presence of microbial rod-shaped cells in its intestinal cells [4]. The rod-shaped cells were Nematocida parisii, an intracellular microsporidium that invade intestinal cells of C. elegans (Figure 2). While discovered in France, the N. parisii can be found in different Caenorhabditis nematodes all over the world [6]. N. parisii became of interest in the research community because it was the first pathogen isolated directly from a wild-caught C. elegans and it was the first pathogen that invaded and lived in C. elegans intestinal cells [6].
The C. elegans and N. parisii relationship allowed researchers to have an opportunity to study the specific mechanisms of microsporidia pathogenicity and other intracellular microbes that thrive in the intestines of other organisms [6]. There are many reasons as to why N. parisii is such a unique pathogen. For one matter, C. elegans is still able to survive and reproduce successfully with high cellular infestation by N. parisii, which means that there is a significant coevolution between the two [4,6,7]. In fact, even if all of its intestinal cells are filled with N. parisii spore cells, C. elegans still moves and appears normal initially, despite the lethality of the infestation [6]. Gradually over time, as the volume of the microsporidian spores increase, C. elegans begins to have sluggish movements [5]. Although it is known that N. parisii causes a lethal infestation to its host's intestinal tissues, it is still unclear as to how the N. parisii impacts C. elegans and how its pathegenesis functions as it is isolated to only the intestinal cells and has evolved to cause minimal impact on its host [3,6].
Infection and Spread of N. parisii in C. elegans
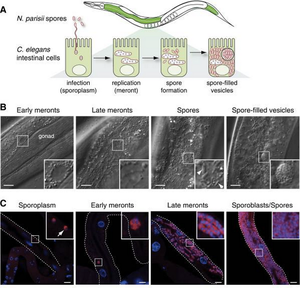
A) Visual of N. parisii infection of C. elegans intestinal cells (green) from sporoplasm invasion to meront formation to replication and formation of spores.
B) DIC images of dissected animals showed various stages of infection. Arrows point out large spores.
C) FISH (red) and DAPI (blue) staining of N. parisii at various stages of infection.
Image source: http://genome.cshlp.org/content/22/12/2478.full.pdf+html [3]
N. parisii can only be transmitted from an infected host to an uninfected host, which is horizontal transfer [3]. However, they show little to no evidence for any sort of vertical transfer, transmission from parent to offspring [3,6]. Infected C. elegans parents can yield healthy uninfected offspring if the surface of the eggs are sterilized or if they are removed from the presence of infected parents immediately following hatching [3,6]. Observation of N. parisii have only shown intestinal cells to be colonized. They are able to enter the intestinal region of C. elegans through either oral consumption or rectal entrance. It infects C. elegans intestinal cells during its transmissible spore form, outlined in Figure 3A [7]. Using a light microscope, researchers are able to identify infection by the formation of gut granules in the intestine of C. elegans [5,6]. It takes one to two days for distinct gut granule formation [6].
Following the identical spore germination outlined in microsporidia, N. parisii spores evert their polar filament to form a polar tube. The polar tube breaks through the cell wall and cell membrane of the spore and launches itself through the cell membrane of a nearby host cell, in this case the C. elegans intestinal cell. The polar tube serves as a bridge between the spore and the intestinal cell and injects the N. parisii sporoplasm into the C. elegans intestinal cell. Sporoplasm becomes a meront, which matures and differentiates so it is able to generate mature N. parisii spores. N. parisii are reported to have a 2.9 to 3.3 hour doubling time inside C. elegans host cells [5]. As more and more spores are synthesized, they cluster together to form separate vesicles in the intestinal cell [5]. Figure 3B outlines the phase-by-phase visual of post-sporoplasm injection in C. elegans and the development of meront to the high concentration of spores in the form of vesicles. N. parisii lack the RB tumor-suppressor gene, a cell cycle inhibitor that stalls cells in the first gap phase (G1) [5]. The lack of the RB is the reason behind why N. parisii has such a rapid intracellular growth. Figure 3C gives a visual of the rapid spore replication during stages of meront development. Notice as the N. parisii (red) take over the C. elegans intestinal cells (blue), but they do not disappear. When intracellular pathogens, such as viruses, produce progeny in a host cell, the cell is forcibly lysed and releases all the progeny viruses out. However, from the stain, there is a reasonable concentration of purple in the area where intestinal cells were located, indicating the cells are still alive [5]. As it turns out, mature N. parisii spores are able to exit the host cell without causing severe damage to the cell. N. parisii is able to manipulate the host cell’s cytoskeleton to commit non-lytic escape [7].
Electron microscope analysis of N. parisii infection in intestinal cells show that the presence of N. parisii in the intestinal cell results in alteration of the terminal web [6,7]. The terminal web is a cytoskeletal structure found in many polarized epithelial cells [6]. N. parisii are able to rearrange ACT-5, a specialized actin isoform that is associated with both the terminal web and microvilli of C. elegans. ACT-5 is relocalized from the apical side to the ecotopical basolacteral side of the cell [7]. The actin relocalization leaves approximately 1 μm-wide gaps in the terminal web [7]. These gaps allow the N. parisii spores formed from the meront to escape the host cell and into the C. elegans lumen without causing cell lysis. Leaving the host cell alive is beneficial to N. parisii because it keeps the host alive, so it can pass off the pathogen to another healthy host [7].
Current Research on N. parisii
Limited information is available on microsporidia, so having a model organism with a natural microsporidian pathogen led to many different experiments. An interesting fact about C. elegans is that they are organisms that lack a professional immune cell, which are cells that have a primary function of consuming foreign units through phagocytosis [10]. C. elegans has to depend solely on its epithelial immunity, which is a non-professional immune system. Non-professional immune cells usually have a primary job outside of pathogen defense and a secondary job of phagocytic responses to a specific pathogen [10].
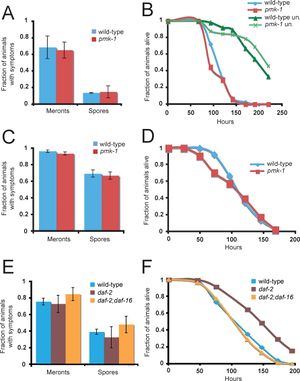
A) At its first larval stage (L1), the C. elegans were infected with N. parisii after 43 hours. Shows the comparison of infection symptoms in wild-type and pmk-1. (p = 0.75 for meronts, p = 0.39 for spores)
B) Comparison of survival rate over time between infected and uninfected (un.) wild-type and pmk-1 after infection at L1. (p = 0.0529)
C) Comparison of infection symptoms between wild-type and pmk-1 in infected fourth stage larvae (L4) after 47 hours. (p = 0.10 for meronts, p = 0.53 for spores)
D) Comparison of survival rate over time between infected and uninfected (un.) wild-type and pmk-1 after infection at L4. (p = 0.65)
E) Infection at L4; comparison of infection symptoms 45 hours later between wild-type, daf-2, and daf-2(e1368);daf-16(mgDf47). Wild type vs daf-2 (p = 0.69 for meronts, p = 0.44 for spores); wild-type vs daf-2(e1368);daf-16(mgDf47) (p = 0.15 for meronts, p = 0.22 for spores); daf-2 vs daf-2(e1368);daf-16(mgDf47) (p = 0.19 for meronts, p = 0.17 for spores).
F) Survival rate difference between wild-type, daf-2, and daf-2(e1368);daf-16(mgDf47) after L4 infection. Wild type vs daf-2 (p < 0.0001); wild-type vs daf-2(e1368);daf-16(mgDf47) (p = 0.40); daf-2 vs daf-2(e1368);daf-16(mgDf47) (p < 0.0001).
Image Source: http://www.plosbiology.org/article/fetchObject.action?uri=info%3Adoi%2F10.1371%2Fjournal.pbio.0060309&representation=PDF[4]
In Troemel, et al., 2008, they investigated whether or not immunity pathways identified for defense against known pathogens would be able to potentially have the same effect against N. parisii. Preliminary experiments had shown that p38 mitrogen-activated protein kinase (MAPK) is able to help C. elegans with pathogens. p38 MAPK is an insulin signaling pathway that is involved in defending C. elegans from various bacterial and fungal pathogen [6]. In order to determine whether or not p38 MAPK has the same effect on N. parisii, C. elegans were taken and were inserted with a p38 MAPK knockout gene, making a pmk-1 mutant. In Figure 5A to D, they compare infection and survival rates between the wild-type C. elegans with the functional p38 MAPK and the pmk-1 mutant.
Figure 5A showed the percentage of infection symptoms between wild-type and pmk-1 43 hours after they were infected as first stage larvae (L1). The C. elegans were infected with either a meront stage N. parisii or a N. parisii spore to identify which of the two were more infectious. In this figure, they compared the appearance of infection symptoms and found that C. elegans infected with N. parisii spores showed less symptoms of infection than in those infected with meronts. The larger matter of interest was whether or not there was a difference in infection symptoms between the wild-type and pmk-1. However, since the error bars overlap, there were no differences between the percentage of individuals with symptoms for wild-type and pmk-1. Figure 5C had a similar experiment to 5A, the key difference in 5C is that the C. elegans were first infected at fourth stage larvae (L4), instead of L1. The percentage of infection symptoms increased for both meronts and spores. Almost all C. elegans infected with meronts showed symptoms of infection, while spores showed that approximately 60-70% of C. elegans had infection symptoms. In comparison of L1 and L4 infection, L4 infection has a higher fraction of infection symptoms. In meronts, L4 infection symptoms increased by 20% and in spores, L4 infection was over three times the symptoms found in L1. However, once again, there were no significant differences in percentage of infected individuals between wild-type and pmk-1 in both meronts and spores infections. Overall, the graphs showed that the presence or lack of p38 MAPK has no impact on C. elegans’ ability to defend itself against N. parisii as found in other pathogens.
Figure 5B compared the uninfected wild-type (green with triangles) and pmk-1 (light green with X's) with their infected L1 counterparts for their survival rate. Between the uninfected wild-type and pmk-1, the uninfected pmk-1 had a slightly lower initial survival rate than the wild-type as shown beginning after hour 100 of infection. Despite the earlier decline, the uninfected pmk-1 had an overall similar trend in survival rate to the wild-type, indicating that the knockout of p38 MAPK did not cause a significant fitness difference. As expected, the infected strains had a significantly lower survival rate than the uninfected strains, proving N. parisii's presence is a contributing factor of C. elegans death. When comparing the infected wild-type (blue with diamonds) and infected pmk-1 mutant (red with squares), the infected pmk-1 showed a dip at hour 100 post-infection, but realigned with the survival rate of the wild-type at the next reading at hour 125. This result showed that pmk-1 loss did not greatly reduce the survival rate. Overall, for C. elegans infected at the L1 stage, the presence of pmk-1 had no effect on survival rate. Figure 5D is a similar experiment to 5B, but the difference being that the samples were infected at L4, not L1. Once again the pmk-1 mutant has a dip in its survival rate in the beginning from hour 50 to 100, but at hour 100, the pmk-1 survival rate was identical to the rate for the wild-type. For all the infected C. elegans, with or without pmk-1, they died after hour 175 post-infection, when the average lifespan of the C. elegans were upwards of 336 hours [5].
Since pmk-1 loss did not show any significant results, the laboratory members tested the parallel pathway to pmk-1, the daf-2/daf-16 insulin/insulin-like growth factor (IGF) signaling pathway, which is a pathway found in C. elegans that helps regulate resistance to a variety of pathogens [6]. Two mutants were made, a daf-2 mutant that knocked out the daf-2 insulin receptor that induces the resistance to pathogens in C. elegans and a daf-2;daf-16 mutant that knocked out both daf-2 and daf-16, its downstream transcription factor. In Figure 4E, the C. elegans were infected at L4 because daf-2 is necessary for larval development and infection at L1 is not an option [6]. The percentage of C. elegans with infection symptoms for each infection type (meronts and spores) were calculated for each type: the wild-type, daf-2, and daf-2;daf-16. Those that were infected with meronts had a higher percentage of infection symptoms than spores as shown earlier with the pmk-1 vs wild-type experiments. The daf-2;daf-16 mutants had a slightly higher susceptibility to infection symptoms than the wild-type and daf-2, while daf-2 showed an average infection rate slightly lower than wild-type. However, due to the variation in the the replications, the error bars overlapped among wild-type, daf-2, and daf-2;daf-16 indicating that any slight differences in infection rate were not significant. In Figure 5F, there was a comparison of the survival rate of wild-type, daf-2, and daf-2;daf-16. Wild-type and daf-2;daf-16 rates overlapped almost completely, indicating that the lack of both daf-2 and daf-16 has no impact on the survival rate. However, daf-2 was able to survive longer than wild-type and daf-2;daf-16. From hour 75 to 200, daf-2 has a higher fraction of alive C. elegans than the other two. Nonetheless, daf-2 still declined, most likely from the N. parisii infection. From Figure 5F, we can take away that the presence of daf-16 was able to provide C. elegans with some sort of minor infection resistance to prolong its lifespan for several days longer.
Although PMK-1 p38 MAPK and the DAF-2/DAF-16 pathways are essential to helping C. elegans defend against pathogens such as P. aeruginosa and S. aureus, the data shown indicated that they are not entirely beneficial in C. elegans’ defense against N. parisii or other microsporidia. The potentially interesting lead would be investigating how what other immunity pathways daf-16 is involved in. In further research conducted by the Ausubel laboratory, they found that the example pathogens for the Troemel experiments, P. aeruginosa and S. aureus, which were the pathogens are defeated by the expression of the p38 MAPK and DAF-2/DAF-16 pathways, actually induced genes that N. parisii did not induce [6]. The takeaway from this elaborate experiment is that N. parisii is a distinct pathogen from previously studied C. elegans pathogens and further research would be necessary to determine how to better defend the C. elegans intestinal cells from this pathogen.
Conclusion
Microsporosis cases have been found in every continent excluding Antarctica with no treatment available for its infection. As cases of microsporidiosis are becoming more and more well known, we anticipate allocation of funding to conduct research in order to better understand this unique phylum. As shown by the Troemel, et al., 2008, experiments, microsporidia are unique organisms that are resistant to many of the defense systems utilized to treat infection by other pathogens we are more familiar with. Since C. elegans and humans both have intestines with epithelial cells covered with apical microvilli anchored to a terminal web, C. elegans would be a good possible research organism to experiment with. In addition to this commonality, C. elegans are inexpensive to obtain and maintain in the laboratory, so it is very convenient to conduct further studies on N. parisii in C. elegans to better understand the microsporidian infections, test and develop possible anti-microsporidian drugs to enhance immunity to the microsporidia for humans [8].
Aside from being used for developing a treatment for gastrointestinal microsporidiosis, the opposite could be conducted. It is possible that with more research, N. parisii could be utilized in a controlled manner in agriculture to reduce the population of nematodes that are considered pests. A similar microsporidium, Antonospora locustae, from the same clade as N. parisii has already been used successfully to control the grasshopper populations [5].
References
[9] Slonczewski, J.L. and Foster, J.W. Microbiology: An Evolving Science. "W.W. Norton & Company, Inc." 2013. Third Ed.
[10] Rabinovitch, M. "Professional and non-professional phagocytes: an introduction". "Cell Biology". 1995. Volume 5 Issue 3. p.85-87.
Edited by Amanda He of Joan Slonczewski for BIOL 238 Microbiology, 2014, Kenyon College.
