Psilocybe
Classification
The genus Psilocybe is of the domain Fungi, the phylum Basidiomycota, the class Agaricomycetes, the order Agaricales, and the family Strophariaceae [1].
Description and significance
The genus Psilocybe composes the fungi known as “magic mushrooms.” These are psychedelic mushrooms that produce the metabolite psilocybin [2]. Members of this genus are distributed worldwide and are able to grow in most biomes. The greatest species diversity is found in the neotropic zone, specifically Mexico, while other important members are found in the forests of Japan and China [2,3]. This genus is important because of the increasing use of its hallucinogenic molecule as a recreational psychedelic drug, as well as its potential use in alleviating the symptoms of certain mental illnesses, such as anxiety, depression, and obsessive-compulsive disorder [4,5]. How and why these mushrooms make this molecule is unknown, and current research continues to focus on its effects as opposed to its purpose in the fungi.
Genome structure
The DNA genome of Psilocybe cubensis has been sequenced, and sequencing libraries have been constructed [6]. The genome contains 32 contigs, and has a total length of 46,603,414 base pairs. The GC content of the total genome is 46.09%, and the AT content is 53.91% [7]. The noncoding sequence of the genome is 26,985,414 base pairs in length. It has a GC content of 43.24% and an AT content of 56.76% (7). After being annotated using FunAnnotate, the genome was found to contain 13,478 genes [6].
The genera Psilocybe and Panaelous have many similarities in their rRNA sequences (8,9). The ITS-1 region of Psilocybe varies from roughly 295 to 350 base pairs. In comparison, the genus Panaeolus, shows little variation with a range of about 20 base pairs [8].
A common feature among species in the Psilocybe genus is that all the necessary anabolic, transport, and regulatory genes used in metabolite biosynthesis are organized into genes that may have been acquired via horizontal gene transfer. The psilocybin pathway in particular may have been acquired because of the horizontal gene transfer of a gene cluster [10]. Horizontal gene transfer of gene clusters also further supports that the Psilocybe genus is closely related to Panaeolus, another fungi genus that has species that also contain the hallucinogen psilocybin [10].
Cell structure
Psilocybe mushrooms are gilled mushrooms that are typically small with brown caps [11, 12]. They produce basidiospores that have a non-angular shape with two layers detectable through light microscopy [13]. Dormant basidiospores have clearly defined pores on their nuclei, a well defined plasma membrane, mitochondria, lipid bodies, and ribosomes. One of the defining features of the genus Psilocybe is the thickness of the basidiospores they produce. However, Psilocybe spores do not have an endoplasmic reticulum (14]. When these spores are germinated with a hot water extract of horse dung, the only differences seen are an increase in small vacuoles and electron density in the germ tubes [14].
Metabolic processes
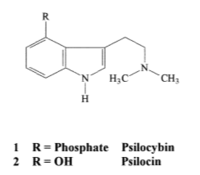
Psilocybe acquire energy by breaking down organic molecules, and therefore, it is a chemoorganoheterotroph. Psilocybe, like other fungi, excrete the enzymes onto the material that is to be metabolized and ingest the material once it is hydrolyzed [15]. Members of the genus Psilocybe synthesize the hallucinogenic indole alkaloid molecule (Figure 2) [8]. The difference in the structure of this molecule (either psilocybin or psilocin) between some members of the genus is due to phosphorylation (psilocybin) or hydroxylation (psilocin) at the R position [8]. Psilocybin contains an indole-amine ring in its structure (Figure 2) and is derived from tryptophan, the only amino acid with an indole-amine ring. Tryptophan is then decarboxylated to tryptamine [16]. After tryptamine is produced, it is methylated two times to become DMT, which is hydroxylated to psilocin. Psilocin is then phosphorylated to synthesize psilocybin [16].
Oral consumption of psilocybin results in dephosphorylation of the molecule, primarily by alkaline phosphatase, in the intestine and liver. The dephosphorylated form of psilocybin (psilocin) is responsible for psychotropic effects in humans [17].
Psilocybe have a unique blue-staining reaction that happens after the fruit body of the Psilocybe is damaged. This reaction occurs when psilocybin is oxidized after the fruit body becomes exposed to oxygen and water. The extent to how much bluing occurs depends on the concentration of psilocin in the organism [18].
Ecology
Psilocybe play a part in early decay of wood [8]. Via horizontal and vertical gene transfer, the genus also developed dung-decaying properties [8]. Psilocybin modulates insect behavior by acting as a neurotransmitter agonist, affecting the feeding behavior of social insects and invertebrates like termites. Several wood-decaying fungi, like Psilocybe, have repellents and toxins that prevent termites from feeding on wood, which provides a competitive fitness advantage [8].
The Psilocybe genus is found all over the world, but the neotropic ecozone contains the most diversity (2). One member of the genus, P. semilanceata, exists in both cool climates like the coasts of Northern America and Europe and warm areas like India and Chile. However, P. semilanceata’s preferred habitat is grassy fields and meadows that have been fertilized by animal feces [2]. P. semilanceata acts as a saprobe by gaining nutrients from the decaying grass roots, unlike P. cubensis which feeds and lives on top of the dung [2].
Pathology
The genus Psilocybe consists of fungi that produce the hallucinogenic molecule, psilocybin, the dephosphorylated form of which creates psychotropic effects in humans [2, 17]. Psilocin undergoes further metabolism after ingestion, similar to that of serotonin because of their structural similarity [14]. However, psilocin also acts as a competitive inhibitor of monoamine oxidase (MAO) which metabolizes serotonin. Therefore, the metabolism, and dephosphorylation, of psilocybin to psilocin may result in elevated levels of serotonin [17].
Psilocybin is part of a structurally diverse group of molecules. These molecules are antagonists of the 5-HT2A receptor and can induce hallucinogenic effects, such as changes in thoughts, perceptions, and emotions [4]. Recent discoveries suggest that these molecules might be useful in treating mental health disorders. When given a high-dose of psilocybin, terminal cancer patients experienced a significant decrease in their depression and anxiety, and it allowed for improvements in their attitude and general outlook on life [2]. These changes were sustained in over 80% of patients [2]. A specific member of this genus, Psilocybe argentipes, is able to reduce marble-burying behavior in mice. Marble-burying behavior is considered an animal model for obsessive-compulsive disorder in humans, indicating that psilocybin might be effective in the clinical treatment of OCD [19].
Current Research
The study of the Psilocybe genus has become increasingly popular in the last few decades, specifically in terms of its use as an antibacterial agent as well as in the treatment of mental health disorders.
In June 2020, a strain of Psilocybe cubensis called Psilocybe cubensis DPT1 was isolated and analyzed for its phytoconstituents. The strain was found to produce alkaloids, flavonoids, tannins, and saponins [20]. This allowed the mushroom to act as a good antibacterial agent as well as a larvicidal agent for mosquitos. The mushroom extracts proved to be antibacterial against both Gram-positive and Gram-negative bacteria [20].
While historically, psilocybin has been used recreationally, the possibility of using the molecule to treat certain psychological disorders has become more likely, as it has been discovered that psilocin is structurally similar to serotonin. This allows it to react with human serotonin receptors [21]. In July 2020, it was discovered that by expressing a heterologous biosynthesis pathway from Psilocybe cubensis and supplementing it with a novel cytochrome P450 reductase from P. cubensis in Saccharomyces cerevisiae, it is possible to produce psilocybin in a controlled environment. This is a critical step in potentially using psilocybin as a treatment in a pharmaceutical setting [21].
References
[1] Schoch CL, Ciufo S, Domrachev M, Hotton CL, Kannan S, Khovanskaya R, Leipe D, Mcveigh R, O'Neill K, Robbertse B, Sharma S, Soussov V, Sullivan JP, Sun L, Turner S, Karsch-Mizrachi I. NCBI Taxonomy: a comprehensive update on curation, resources and tools. Database (Oxford). 2020 Jan 1;2020:baaa062. doi: 10.1093/database/baaa062.
[2] de Mattos-Shipley KM, Ford KL, Alberti F, Banks AM, Bailey AM, Foster GD. The good, the bad and the tasty: The many roles of mushrooms. Stud Mycol. 2016;85:125-157. doi:10.1016/j.simyco.2016.11.002
[3] He, P., He, X. & Zhang, C. Interactions between Psilocybe fasciata and its companion fungus Acremonium strictum. Ecol Res 21, 387–395 (2006). https://doi.org/10.1007/s11284-005-0123-0
[4] Griffiths RR, Johnson MW, Carducci MA, et al. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: A randomized double-blind trial. J Psychopharmacol. 2016;30(12):1181-1197. doi:10.1177/0269881116675513
[5] Matsushima Y, Shirota O, Kikura-Hanajiri R, Goda Y, Eguchi F. 2009. Effects of Psilocybe argentipes on marble-burying behavior in mice. Biosci Biotechnol Biochem. 73(8):1866-8.
[6] McKernan K, Kane LT, Crawford S, Chin CS, Trippe A, McLaughlin S. A draft reference assembly of the Psilocybe cubensis genome. F1000Res. 2021;10:281. Published 2021 Jun 15. doi:10.12688/f1000research.51613.2
[7] CoGe genome browser: Psilocybe cubensis (Psilocybe cubensis P.envy), https://genomevolution.org/coge/GenomeInfo.pl?gid=60487
[8] Lee, James Chun-I., Michael Cole, and Adrian Linacre. "Identification of members of the genera Panaeolus and Psilocybe by a DNA test: a preliminary test for hallucinogenic fungi." Forensic science international 112.2-3 (2000): 123-133. https://doi.org/10.1016/S0379-0738(00)00181-X
[9] Maruyama, Takuro, et al. "Phylogenetic relationship of psychoactive fungi based on rRNA gene for a large subunit and their identification using the TaqMan assay (II)." Forensic science international 163.1-2 (2006): 51-58. https://doi.org/10.1016/j.forsciint.2004.10.028
[10] Reynolds H, Vijayakumar V, Gluck-Thaler E, et al. Horizontal gene cluster transfer increased hallucinogenic mushroom diversity. Evolution Letters. 2(2): 88-101.
[11] Encyclopedia of forensic sciences (2nd ed.). (2012). Academic Press.
[12] Dalefield, R. (2017). Veterinary toxicology for Australia and New Zealand. Elsevier Science Publishing.
[13] Ramírez-Cruz V, Guzmán G, Villalobos-Arámbula AR, et al. Phylogenetic inference and trait evolution of the psychedelic mushroom genus Psilocybe sensu lato (Agaricales). Botany. 91(9): 573-591.
[14] Stocks DL, Hess WM. Ultrastructure of Dormant and Germinated Basidiospores of a Species of Psilocybe. Mycologia. 62(1): 176-191.
[15] Segner, Suzanne, and Karen-Beth G Scholthof. “Environmental Microbiology: Bacteria & Fungi on the Foods We Eat.” The American Biology Teacher, vol. 69, no. 3, Mar. 2007.
[16] Fricke, Janis, Felix Blei, and Dirk Hoffmeister. "Enzymatic synthesis of psilocybin." Angewandte Chemie International Edition 56.40 (2017): 12352-12355.
[17] Dinis-Oliveira, Ricardo Jorge. “Metabolism of Psilocybin and Psilocin: Clinical and Forensic Toxicological Relevance.” Drug Metabolism Reviews, vol. 49, no. 1, Jan. 2017, pp. 84–91.
[18] Austin, Emily et al. “Acute renal injury cause by confirmed Psilocybe cubensis mushroom ingestion.” Medical mycology case reports vol. 23 55-57. 22 Dec. 2018, doi:10.1016/j.mmcr.2018.12.007
[19] Matsushima Y, Shirota O, Kikura-Hanajiri R, Goda Y, Eguchi F. 2009. Effects of Psilocybe argentipes on marble-burying behavior in mice. Biosci Biotechnol Biochem. 73(8):1866-8.
[20] Dhanasekaran D, Latha S, Suganya P, et al. Taxonomic identification and bioactive compounds characterization of Psilocybe cubensis DPT1 to probe its antibacterial and mosquito larvicidal competency. Microb Pathog. 2020;143:104138. doi:10.1016/j.micpath.2020.104138
[21] N. Milne, P. Thomsen, N. Mølgaard Knudsen, P. Rubaszka, M. Kristensen, I. Borodina. Metabolic engineering of Saccharomyces cerevisiae for the de novo production of psilocybin and related tryptamine derivatives, Metabolic Engineering, Volume 60, 2020. doi.org/10.1016/j.ymben.2019.12.007.
[22] Rockefeller, Alan. “Fruit Bodies of the Psilocybe Semilanceata Species.” Mushroom Observer, 23 Sept. 2007, https://mushroomobserver.org/image/show_image/6514.
Edited by Emily Budhram, Julia Kern, Alison Leung, and Tommy Pak, students of Jennifer Bhatnagar for BI 311 General Microbiology, 2021, Boston University.