Quorum Sensing in Methanosaeta harundinacea 6Ac
Overview
The archaeon Methanosaeta harundinacea 6Ac is part of a family of aceticlastic methanogens who exclusively catabolize acetate (Ferry 2008). It is rod-shaped, non-motile, and usually found singly or in pairs, but when grown in acetate with high cell density, the cells form into filaments which are self-contained (Ma et. al 2006), products of quorum sensing.
Quorum Sensing
Quorum sensing is a cell density-dependent process of gene regulation by which unicellular organisms can communicate with others of the same species and co-ordinate their behaviours (Miller and Bassler 2001). Signal molecules called autoinducers are constitutively expressed and secreted out of the cell, which then get taken up by members of the other species present in the area. As such, autoinducer concentration in the immediate environment increases as cell density increases and given that enough autoinducers are uptaken, a threshold concentration is reached inside the cells which triggers a change in gene expression. The cells can then co-ordinate a number of processes together, such as biofilm formation, sporulation (Miller and Bassler 2001, production of virulence factors such as in Pseudomonas aeruginosa (Pesci et al. 1997) , or in the case of Vibrio fischeri, bioluminescence (Hastings and Greenberg 1999). In Gram-positive bacteria, oligopeptides serve as autoinducers, and Gram-negative bacteria use acyl homoserine lactones (AHLs) (Miller and Bassler 2001).
More on quorum sensing
Quorum sensing related virulence in Vibrio cholerae
Autoinducers in M. harundinacea 6Ac
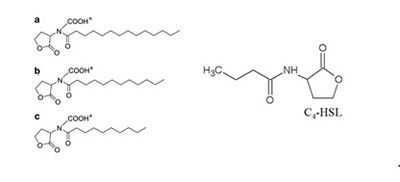
The autoinducers used by M. harundinacea 6Ac are carboxyl-AHLs, which are essentially AHLs with a carboxyl moiety attached to the nitrogen (Zhang et al. 2012). There are 3 slight variants synthesized (see figure 1), which are N-carboxyl-C14-HSL (homoserine lactone), N-carboxyl-C12-HSL, and N-carboxyl-C10-HSL (Zhang et al. 2012). These are made by the enzyme AHL synthase, encoded for by the fliI gene, which is basally transcribed(Zhang et al. 2012). The sensor protein for the carboxyl-AHLs, FilR, is coded for by the filR gene which is also basally transcribed, and the protein binds the autoinducers when they reach a threshold concentration (Zhang et al. 2012), the concentration which is an indication of increased cell-density in the area. The FilR-autoinducer complex binds to certain genes in the DNA resulting in up-regulation of their transcription, and down-regulation of other genes, which results in changes in physiology of the cells as well as a different metabolic pattern (Zhang et al. 2012).
Changes in Physiology
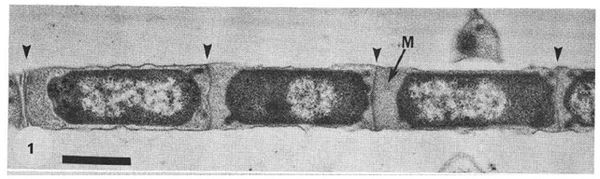
When the cell density is low, M. harundinacea 6Ac exist as short, singular rod-shaped cells (Ma et al. 2006), but when the cell density is high the archaeon’s physiology drastically changes. Chains of cells are stacked end to end forming filaments that can be greater than 200 microns, in contrast to a length of 3-5 microns as single cells (Guo et al. 2009). Beveridge et al. (1986) found that the filaments consist of four major parts: the cells lined up in a chain, spacer plugs which separated each individual cell as well as plugging the two ends of the filament, an amorphous granular matrix, and encasing everything a proteinaceous sheath. The spacer plugs are anchored to the sheath, and appear to have multiple layers. The granular matrix is enclosed by the sheath and the plugs and completely encloses individual cells. The protein sheath is striated and composed of a highly crystalline structure. This lends it tremendous strength against pressure, as well as other physical and chemical disruption (Beveridge et al. 1986). As such, when M. harundinacea 6Ac form into filaments, they can better withstand unfavourable environment conditions and thus have an edge over other micro-organisms in the area. Note that Beveridge et al. (1986) described filaments formed by Methanothrix concilii, which have been renamed to Methanosaeta concilii as elucidated by Patel and Sprott (1990). Given that there is a 92.5% similarity between M. concilii and M. harundinacea 6Ac based on 16S rRNA sequences (Ma et al. 2006), the filament descriptions are likely accurate for M. harundinacea 6Ac as well.
Changes in Metabolism
The metabolic patterns between the 2 distinct morphologies are significantly different. Both methane production and the reaction rate of methanogenesis are higher in the filamentous cells than in the short rods (Zhang et al. 2012). It was also shown by Zhang et al. (2012) that the filamentous cells produce lower levels of cellular proteins compared to the short cells, and had less biomass. The significance of this is that M. harundinacea 6Ac can better compete with other micro-organisms for resources in the environment, such as for acetate with acetoclastic microbes, and as well they can divide quicker due to faster metabolism and the lower biomass required, and outgrow any competitors.
Similarities to Bacterial Quorum Sensing
There are quite a few similarities between the quorum sensing mechanism of M. harundinacea 6Ac and that of bacteria. The filI-filR system is a homolog of a universal quorum sensing system in bacteria, the luxI-luxR system, which essentially works the same way. The luxI gene encodes a protein which synthesizes the autoinducer, and the autoinducer is bound by the sensor protein encoded by luxR, and the LuxR-autoinducer complex facilitates the changes in gene expression (Miller and Bassler 2001). FilI synthesizes the autoinducer carboxyl-AHL, which as mentioned above is only one carboxyl moiety away from being an AHL, which is the autoinducer utilized by Gram-negative bacteria. This suggests that possible horizontal gene transfer, the flow of genetic information between prokaryotes of different species (Syvanen M. 2012), might have occurred between bacterial species and the archaeon. In addition, several other Methanosaeta spp.(such as Methanosaeta thermophila, and other methanogenic archaea (such as Methanosarcina mazei have been found to respond to carboxyl-AHLs and undergo morphological changes in a cell density-dependent manner (Zhang et al. 2012), and so AHL-based quorum sensing may be a universal mechanism in prokaroytes.
Summary
Quorum sensing is a demonstrated process in the archaeon M. harundinacea 6Ac, via the usage of carboxyl-AHLs and the FilR protein, and the changes in gene expression result in the formation of filaments composed of 4 major parts, the cells, spacer plugs, extracellular matrix, and a sheath. As well the filamentous cells have a higher and faster methane output rate, and have decreased protein production, and as a result a lower biomass. These changes enable the archaea to work together as a group and give them an edge over other microbes in the environment, namely being more resistant to unfavourable environment conditions, and being able replicate faster.
References
Beveridge T.J., Patel G.B., Harris B.J. and Dennis Sprott G. “The Ultrastructure of Methanothrix concilii, a Mesophilic Aceticlastic Methanogen.” Canadian Journal of Microbiology, 1986, DOI: 10.1139/m86-128
Davis B.M., Jensen R., Williams P., and O’Shea P. “The Interaction of N-Acylhomoserine Lactone Quorum Sensing Signaling Molecules with Biological Membranes: Implications for Inter-Kingdom Signaling.” PLoS ONE, 2010, 5(10):e13522.doi:10.1371/journal.pone.0013522
Ferry, J.G. “Acetate-Based Methane Production.” Bioenergy, 2008, pp. 155-170
Guo X., Zhang G., Liu X. and Dong X. “Detection of the Quorum Sensing Signals in Methanogenic Archaea.” Acta Microbiologica Sinica, 2011, 51(9):1200-1204
Hastings J.W. and Greenberg E.P. “Quorum Sensing: the Explanation of a Curious Phenomenon Reveals a Common Characteristic of Bacteria”. Journal of Bacteriology, 1999, 181(9):2667-2668
Ma K., Liu X., and Dong X. “Methanosaeta harundinacea sp nov., a novel acetate-scavenging methanogen isolated from a UASB reactor.” International Journal of Systematic and Evolutionary Microbiology, 2006, DOI: 10.1099/ijs.0.63887-0
Miller M.B., and Bassler B.L. “Quorum Sensing in Bacteria.” Annual Review of Microbiology, 2001, DOI: 10.1146/annurev.micro.55.1.165
Patel, B.G. and Dennis Sprott G. “Methanosaeta concilii gen. nov., sp. nov. (“Methanothrix concilii”) and Methanosaeta thermoacetophila nom. rev., comb. nov.” International Journal of Systematic and Evolutionary Microbiology, 1990, DOI: 10.1099/00207713-40-1-79
Pesci E.C., Pearson J.P., Seed P.C., and Iglewski B.H. “Regulation of las and rhl quorum sensing in Pseudomonas aeruginosa”. Journal of Bacteriology, 1997, 179(10):3127-3132
Syvanen M. “Evolutionary Implications of Horizontal Gene Transfer”. Annual Review of Genetics, 2012, DOI: 10.1146/annurev-genet-110711-155529
Zhang G., Zhang F., Ding G., Li J., Guo X., Zhu J., Zhou L., Cai S., Liu X., Luo Y., Zhang G., Shi W. and Dong X. “Acyl Homoserine Lactone-Based Quorum Sensing in a Methanogenic Archaeon.” The ISME Journal, 2012, DOI:10.1038/ismej.2011.203s