Rabies Lyssavirus
Introduction
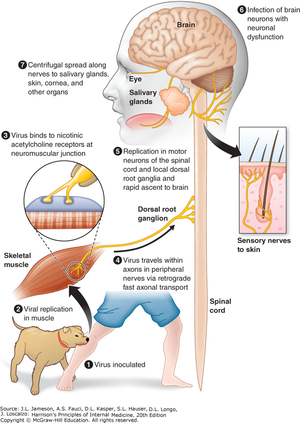

Rabies Lyssavirus, commonly known as the Rabies virus (RABV), is a bullet-shaped neurotropic virus that causes rabies in mammals.[1]
In North America, bats are the leading carrier for the Rabies virus, while dog bites are the most common causes in other parts of the world. Once the Rabies virus is transmitted through bite or saliva of rabid animals, it will replicate in patients’ muscle tissue, then transmit to the central nervous system(CNS)through the axons of the peripheral nerves. Once the Rabies virus arrives at the brain, it will infect the brain neurons, which eventually lead to encephalitis.[2] If bites from rabies-infected animals did not receive immediate care and rabies symptoms (aggressiveness, hallucinations, fever, nausea, foaming at the mouth, fear of water) start to appear in patients, it is usually fatal.[3]
Rabies virus is preventable for humans through vaccination or fast-acting shot that contains rabies immune globulin for those who got bitten recently. Other mammals such as human-pet dogs and cats can also get the treatment for the prevention of rabies virus.[4]
Vaccination for rabies virus is well delivered in developed countries, but it is less accessible in less developed countries around the globe. While preventable through vaccination, there is currently no cure for the Rabies virus, and it carries approximately 55,000 lives away each year.[5]Scientists are actively researching for a possible cure for the rabies virus.
Structure
Rabies Virus (RABV) belongs to the family Rhabdoviruses and the genus Lyssavirus. RABV is a bullet-shaped RNA virus that is about 180 nm in length and 75 nm in width. The "bullet" contains one copy of an enveloped single-stranded non-coding RNA genome that contains about 12,000 nucleotides.[6]The two significant components of the Rabies virus are the ribonucleoprotein complex(RNP) and the surrounding cell envelope. Out of the five proteins, nucleoprotein(N), Phosphoprotein(P), and Polymerase protein(L) are associated with the RNP. The matrix protein is responsible for covering and keeping the RNP in a condensed form. The rabies virus envelope is constructed of a phospholipid bilayer with glycoprotein spikes.[6]
RABV genome encodes five essential structural proteins:
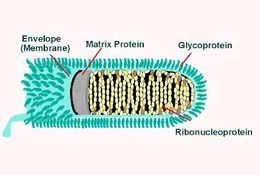
Nucleoprotein(N):
The nucleoprotein encapsidates the non-coding RNA genome to form the nucleocapsid core. The nucleocapsid core, along with polymerase protein and phosphoprotein, constructs the ribonucleoprotein complex(RNP).[2]RNP is responsible for the protection of the rabies virus genome from RNA degradation caused by ribonuclease. Other than genome protection, nucleoprotein is also an antigen for viruses due to their group and strain-specific antigenic determinants.[7]
Phosphoprotein(P):
The phosphoprotein (P) plays a key role in the replication of RABV viral RNA by being the cofactor of an essential enzyme RNA-dependent RNA polymerase (RdRP). The phosphoprotein accomplishes the key RNA synthesis by connecting the nucleoprotein and the polymerase protein in the ribonucleoprotein complex.[8]
Recent research from 2013 revealed that in the strain Nishigahara, P protein is involved in the neuroinvasive of the Rabies virus by mediating the peripheral nerve infection, which caused lethal results in intramuscular inoculated mice. However, the P protein from the derivative strain Ni-CE, while also causing lethal results in mice, was not significant for the stable viral replication process in the host’s muscle cells. It was later investigated that the Nishigahara P protein gene was able to counteract the host interferon(IFN) system, which is responsible for interfering with the viral replication process to protect host cells from infection. [9] The Nishigahara P protein was able to suppress the gene expression of beta interferon and IFN-stimulates genes in the host mice muscle cells. Thus, the infection of the peripheral nerves is enhanced by the Nishigahara P protein. [10] However, it is to be further investigated about the suppression effect with other Rabies Virus strains P protein.
Polymerase protein(L):
Polymerase protein is responsible for the transcription of the RABV RNA. During transcription, the L protein will transcribe the viral RNA into leader RNA and five mRNAs that are each capped and polyadenylated. The RNA-dependent RNA polymerase protein is a potential target for rabies treatment because it produces all viral proteins for RABV and requires interaction with other viral proteins for the primary transcription.[11]
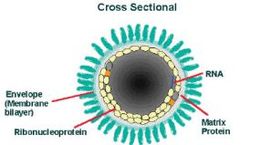
Matrix Protein(M):
Matrix protein locates on RABV’s inner envelope. It has been known for two decades that matrix protein is responsible for interaction with glycoprotein. M protein recruits RNPs, whose condensation forms the budding of the bullet-shaped particles.[12] The membrane matrix protein is later known for being a factor for the balance of RABV transcription and replication through inhibiting transcription and stimulating the replication process.[13]
Glycoprotein(G):
The translation of G protein is taken place on hosts’ endoplasmic reticulum bound ribosomes.[14] The glycosylated G-protein Forms glycoprotein spikes on the virus’s outer envelope, which allows the virus to be recognized and aid in its binding to host cells.[15]Glycoprotein has the ability to activate the immune system following the production of antibodies, which has been used in a beneficial way for the production of recombinant vaccines. [14]
History

Rabies is an ancient disease. It was called "lyssa" by the ancient Greeks, which means violence. Rabies was first mentioned in text coming from 2300 B.C. when people in Babylon found the death of humans and dogs were related to getting bid by a dog. After the discovery, if a Babylonians dog transmitted another person’s rabies, the dog owner had to pay a fine for it.
Back in the first century A.D., the transmission method of rabies was correctly suggested by Roman scholar Celsus. A cure, however, is yet to be found even with the well-developed scientific field in the 21st century. Back in ancient times, various remedies are being suggested: Celsus suggested holding the patient underwater. Others have suggested laying the rabid dog hair on the patient or letting the patient ingest rabid dog hair. Going into the 18th century, Madstones, which are hairballs found in the ruminants stomachs, was thought to be a valuable rabies treatment. It was reported that in 1849, Abraham Lincoln's son Robert was bitten by a rabid dog and survived with the Madstone treatment. However, the reliability of the report is to be considered.
The first treatment was introduced by Louis Pasteur(Figure 5), a french chemistry teacher. After dabbling with chicken cholera, which is a rapid spreading commonly found in chicken yards, he noticed one of the old virulent cultures no longer caused disease when it was inoculated into a chicken. Pasteur later inoculates a group of chicken with the old virulent culture and left another group of chicken without the inoculation. With some fresh chicken cholera injected to all chickens, those with inoculation survived while the other did not. At that time, Pasteur concluded that the inoculated chicken had used the weaker strains of virulent culture to improve their immunity against the fresh germs. With his chicken cholera experiment, Pasteur found a way to help others become immune without a risk of getting the disease. [16] he then continues with anthrax vaccine for cattle and vaccine for rabies.
The first human being treated successfully with this vaccine was a 9-year-old boy named Joseph Meister, who was scratched by a rabid dog. Joseph recovered after receiving 13 inoculations in 11 days from Pasteur. After the success in 1885, the post-exposure vaccine was being actively researched by scientists and widely used by over 20,000 people.[17]The first available inactivated Rabies vaccine was developed in 1908, and it is estimated by the World Health Organization that the vaccine saves more than 270,000 lives per year. [18]Today, the post-exposure prophylaxis (PEP), which consists of human rabies immune globulin and rabies vaccine is being used as the standard post-treatment for rabies. It is given on the exposure day to the rabies virus. Three doses of the rabies virus would be given to the patients on days 3, 7, and 14. [19]
Vaccination

The rabies vaccine is universal today and is divided into preventive vaccination and post-exposure vaccination. Between 15,000 and 40,000 people are vaccinated each year after a possibly rabid animal bite. Bites from unvaccinated rabid dogs that carry dog RABV cause most of the rabies-related death cases. The Rabies vaccine is universal today and is divided into preventive vaccination and post-exposure vaccination. Between 15,000 and 40,000 people are vaccinated each year after a possibly rabid animal bite. Globally speaking, Bites from unvaccinated dogs cause most of the rabies-related death cases.[4]It is required for mammal pet owners (examples are dogs and cats owners) in the United States to have their pets vaccinated for rabies. Animals coming from high-risk countries for rabies virus need rabies vaccine certificates to enter the State. [20]
Preventive Vaccination
Rabies virus’s preventive vaccine is given to humans and animals as prevention from the rabies virus. Before the 1970s, the Rabies vaccine contained weakened live viruses. However, a pair of kittens felt extreme illness after receiving the vaccination. Thus the updated version of the Rabies vaccine itself cannot cause rabies because it is now made from the inactivated rabies virus. [21] A preventive dose of rabies vaccine take longer to have any effect on animals, and it is commonly given to pets. The majority of humans are not given the preventive vaccine routinely unless they work in an environment where there is a higher chance of exposure to rabid animals.[4]
When the vaccine is being injected, the recipient's immune system responds to the killed virus as if it is a viral infection. After preventive vaccination, there are rabies antibodies present in the blood of the vaccine recipient, and their active immune system protects them from further attacks from the Rabies virus by responding rapidly to future encounters. Pet dogs (Figure 6) usually receive an annual intramuscular dose of preventive vaccine, when they are traveling outside of the United States, extra doses are required for boosting the immunity against rabies.[21]
Post-exposure vaccination

The rabies post-exposure prophylaxis (PEP) is the treatment given to patients after a suspicious bite, scratch, or another form of contact with a rabid animal. Depending on the level of exposure, the patients will receive different treatment. For the most severe exposure, such as direct contact with bats, or deep scratches/bites from rabid animals would require immediate vaccination, rabies immunoglobulin, and treatment for the wound sites. There is a high chance of preventing the rabies virus from infecting the central nervous system if the patient receives the above PEP immediately after exposure. One of the leading causes of the death case related to the rabies virus is hard-to-access prevention, post-treatment, along with lack of knowledge of the rabies virus in less developed countries. WHO is working on reducing the cost of rabies so that it can be more accessible for patients with fewer resources. [22]
Evolution
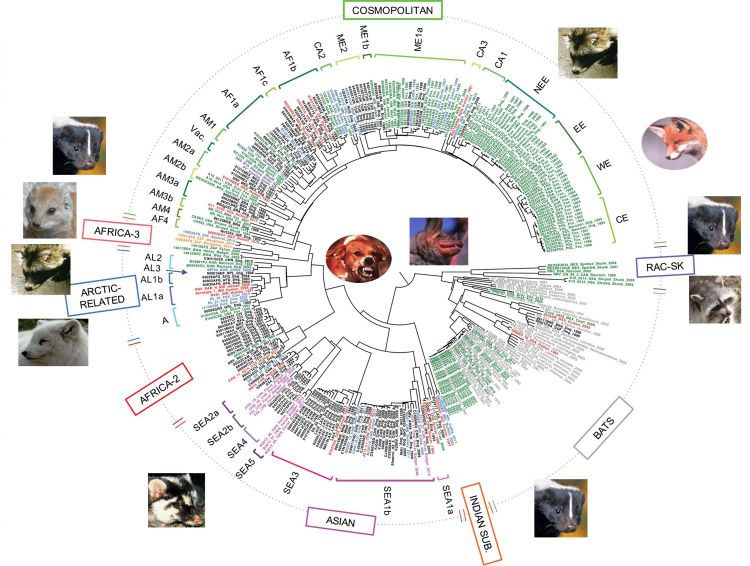
Cécile Troupin and his colleagues in Paris have compared 321 viral genome sequences of the rabies virus that was collected from 66 countries over 65 years. Their resulting circular phylogenetic tree (Figure 8) shows the distinct evolutionary patterns of bat-related rabies and dog-related rabies. bat-related rabies virus is commonly found in bats and some carnivores and does not evolve at a uniform rate, while dog-related rabies is found in dogs and wild carnivores, and evolve at a steady pace. Dog-related rabies is responsible for most of the human rabies cases and deaths.
Dog-related rabies spread rapidly worldwide in the 15th century as trade began happening between the continents, this is also the period when the evolutionary diversification of RABV most likely occurred.[23] It is possible that today's dog-related rabies-infected species arose as an outcome of the combination of the historical spread of rabid animals and host jumping. Rabies virus is long known for its ability to jump between different host species. Host jumping is related to multiple evolutionary patterns; the parallel changes in different host species' amino acid sequences are an example of numerous evolutionary patterns of dog-related rabies. It is also revealed that dog-related rabies might require subtle adaptive evolution for its establishment in a new carnivore host. [24]
Case Study
Although most of the untreated patients of rabies died, there were a few cases that stood out where the patient has not treated pre or post-exposure to the rabies virus and still survived the disease. We are going to focus on the story of Jeanna Giese, who is the first known rabies survivor that did not receive preventive treatments before symptom onset.
Jeanna Giese

Jeanna Giese was infected with the Rabies virus when she picked up a rabid bat outside her church in Wisconsin when she was 15. She and her parents did not seek medical support but cleaned the superficial wound. Rabies symptoms start to appear on Giese three weeks after her encounter with the rabid bat-vomiting, fatigue, double vision. When the symptoms appeared, it was too late for her to receive the post-exposure treatment. Giese's doctor Rodney Willoughby, who was treating her at the Children's Hospital of Wisconsin in Milwaukee, decided to induce a coma on Giese to provide her immune system time to build up antibodies against the rabies virus. The treatment done by Giese's doctor is soon to be known as the Milwaukee protocol. Since the rabies virus infects patients' brains and affects the patient's ability to regulate breathing, heartbeat, and salivation, by suppressing the brain function, the doctors believed it could save Giese from death. While seducing Giese, the doctor also injected antivirals ribavirin (commonly used for treating chronic hepatitis C, a type of liver infection) and amantadine (used for treating Influenza A). Six months after awakening from the coma, Giese was given a compound similar to B-complex vitamin folic acid to improve her speech. After being in isolation for 31 days and receiving a total of 76 days of treatment and hospitalization,[25] Giese survived the rabies virus and recovered most of her cognitive functions. In March 2016, Giese became a mother of two.[26][27]
Milwaukee Protocol

Milwaukee protocol (MP) seems promising, according to the case of Jeanna Giese. However, there is a lack of scientific evidence supporting the protocols of therapeutic measures. A decades after Giese’s successful treatment, there have been at least 29 documented Milwaukee protocol failures in treating rabies patients.[28]
In the 29 cases, 41% received the major components of the Milwaukee protocol (coma and ketamine), which failed in all. MP mentions the use of therapeutic coma in rabies, which has also been controversial, there has not been any scientific evidence, but numeral documented failures. A therapeutically induced coma through intravenous anesthetic agents would impair the neurological examination, which increases the risk of infections and should be avoided. The impact of sedation mentioned in MP still has an unknown effect on rabies patients, and caution should be taken. [29]
Research
New research from November 2019 has raised new hopes for lowering the transmission rate of rabies virus from dogs to humans. The researchers were able to find a way to stop rabies by shutting down the critical responses in the immune system. The currently existing rabies vaccine works by activating the B cells in the immune system. However, in James McGettigan of Thomas Jefferson University, USA, researchers were able to design a rabies vaccine that includes a particle containing a weakened rabies virus and B cell-activating factor (BAFF). It was hypothesized that the new rabies vaccine would be faster and stronger, which was supported by testing on mice. Mice that received the new vaccine had a more intense immune response than mice that received the typical vaccine. This new strategy for rabies vaccine enhanced the efficiency of the antibody response and has the potential to replace the currently used vaccine, which contains inactivated RABV. [30]
Conclusion
Being an ancient disease, the Rabies Lyssavirus has been researched and studied intensively. Although there is currently no cure for the Rabies virus, new research has shed light on possible vaccines that could be more efficient at preventing the RABV in animals and humans. The Rabies virus has also been a research tool for viral neuronal tracing. While the failure cases increased in the past decades for the Milwaukee protocol, we are still hopeful to see what new research on Rabies Lyssavirus come up and discover more applications that are beneficial for human society.
References
- ↑ Rabies - Symptoms and causes [Internet]. Mayo Clinic. 2019. Available from: https://www.mayoclinic.org/diseases-conditions/rabies/symptoms-causes/syc-20351821
- ↑ 2.0 2.1 Long, S., Prober, C. and Fischer, M. (2012). Principles and practice of pediatric infectious disease. 4th ed. Saunders.
- ↑ Kidshealth.org. (2019). Rabies (for Parents) - Nemours KidsHealth. [online] Available at: https://kidshealth.org/en/parents/rabies.html.
- ↑ 4.0 4.1 4.2 Rabies Vaccine [Internet]. Cdc.gov. 2009. Available from: https://www.cdc.gov/vaccines/hcp/vis/vis-statements/rabies.pdf
- ↑ Is Rabies Really 100% Fatal? | Viruses101 | Learn Science at Scitable [Internet]. Nature.com. 2013. Available from: https://www.nature.com/scitable/blog/viruses101/is_rabies_really_100_fatal/
- ↑ 6.0 6.1 What is Rabies? | Rabies | CDC [Internet]. Cdc.gov. 2019. Available from: https://www.cdc.gov/rabies/about.html.
- ↑ Koser M, McGettigan J, Tan G, Smith M, Koprowski H, Dietzschold B et al. Rabies virus nucleoprotein as a carrier for foreign antigens. Proceedings of the National Academy of Sciences [Internet]. 2004;101(25):9405-9410. Available from: https://www.pnas.org/content/101/25/9405
- ↑ Okada K, Ito N, Yamaoka S, Masatani T, Ebihara H, Goto H et al. Roles of the Rabies Virus Phosphoprotein Isoforms in Pathogenesis. Journal of Virology [Internet]. 2016;90(18):8226-8237. Available from: https://jvi.asm.org/content/90/18/8226
- ↑ DE ANDREA, M., RAVERA, R., GIOIA, D., GARIGLIO, M. and LANDOLFO, S. (2002). The interferon system: an overview. European Journal of Paediatric Neurology, [online] 6, pp.A41-A46. Available at: https://www.sciencedirect.com/science/article/pii/S1090379802905738?via%3Dihub
- ↑ Yamaoka, S., Ito, N., Ohka, S., Kaneda, S., Nakamura, H., Agari, T., Masatani, T., Nakagawa, K., Okada, K., Okadera, K., Mitake, H., Fujii, T. and Sugiyama, M. (2013). Involvement of the Rabies Virus Phosphoprotein Gene in Neuroinvasiveness. Journal of Virology, [online] 87(22), pp.12327-12338. Available at: https://www.ncbi.nlm.nih.gov/pubmed/24027304.
- ↑ Nakagawa K, Kobayashi Y, Ito N, Suzuki Y, Okada K, Makino M et al. Molecular Function Analysis of Rabies Virus RNA Polymerase L Protein by Using an L Gene-Deficient Virus. Journal of Virology [Internet]. 2017;91(20). Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5625484/
- ↑ Mebatsion T, Weiland F, Conzelmann K. Matrix Protein of Rabies Virus Is Responsible for the Assembly and Budding of Bullet-Shaped Particles and Interacts with the Transmembrane Spike Glycoprotein G [Internet]. Journal of Virology. 1999. Available from: https://jvi.asm.org/content/73/1/242.long
- ↑ Finke S, Mueller-Waldeck R, Conzelmann K. Rabies virus matrix protein regulates the balance of virus transcription and replication. Journal of General Virology [Internet]. 2003;84(6):1613-1621. Available from: https://www.microbiologyresearch.org/docserver/fulltext/jgv/84/6/1613.pdf?expires=1575256460&id=id&accname=guest&checksum=8E3AA286D649C7B0CAF0AAF05D0D07DC
- ↑ 14.0 14.1 Cherian S, Singh R, KP S. Rabies Glycoprotein: A Benefit to the Virus, us or both? [Internet]. Rroij.com. 2015. Available from: http://www.rroij.com/open-access/rabies-glycoprotein-a-benefit-to-the-virus-us-or-both.php?aid=58725
- ↑ Dietzschold B e. Structure and function of rabies virus glycoprotein. - PubMed - NCBI [Internet]. Ncbi.nlm.nih.gov. 1978. Available from: https://www.ncbi.nlm.nih.gov/pubmed/680401
- ↑ Chicken cholera | ari.info | ari.info [Internet]. ari.info. 2015. Available from: http://www.animalresearch.info/en/medical-advances/veterinary-medicine/chicken-cholera
- ↑ O'Niell F. A History of Rabies [Internet]. Tuckahoevet.com. Available from: https://www.tuckahoevet.com/post/a-history-of-rabies
- ↑ Society M. Rabies virus: can we treat the untreatable? [Internet]. Microbiologysociety.org. 2019. Available from: https://microbiologysociety.org/blog/rabies-virus-can-we-treat-the-untreatable.html
- ↑ Rabies Postexposure Prophylaxis (PEP) | Medical Care | Rabies | CDC [Internet]. Cdc.gov. 2019. Available from: https://www.cdc.gov/rabies/medical_care/index.html
- ↑ Rabies Vaccine Certificate Required When Coming From These High-Risk Countries | Bringing an Animal into U.S. | Importation | CDC [Internet]. Cdc.gov. 2019. Available from: https://www.cdc.gov/importation/bringing-an-animal-into-the-united-states/rabies-vaccine.html
- ↑ 21.0 21.1 The Rabies Vaccine: Facts, Controversy, and Benefits | CertaPet [Internet]. CertaPet. 2019. Available from: https://www.certapet.com/rabies-vaccine/
- ↑ Rabies [Internet]. Who.int. 2019. Available from: https://www.who.int/news-room/fact-sheets/detail/rabies
- ↑ Bourhy H, Reynes J, Dunham E, Dacheux L, Larrous F, Huong V et al. The origin and phylogeography of dog rabies virus. Journal of General Virology [Internet]. 2008;89(11):2673-2681. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3326349/#R25
- ↑ Troupin C, Dacheux L, Tanguy M, Sabeta C, Blanc H, Bouchier C et al. Large-Scale Phylogenomic Analysis Reveals the Complex Evolutionary History of Rabies Virus in Multiple Carnivore Hosts. PLOS Pathogens [Internet]. 2016;12(12):e1006041. Available from: http://10.1371/journal.ppat.1006041
- ↑ Willoughby R, Tieves K, Hoffman G, Ghanayem N, Amlie-Lefond C, Schwabe M et al. Survival after Treatment of Rabies with Induction of Coma. New England Journal of Medicine [Internet]. 2005;352(24):2508-2514. Available from: https://www.nejm.org/doi/full/10.1056/NEJMoa050382
- ↑ Lite J. Medical Mystery: Only One Person Has Survived Rabies without Vaccine--But How? [Internet]. Scientific American. 2008. Available from: https://www.scientificamerican.com/article/jeanna-giese-rabies-survivor/
- ↑ Wisconsin rabies survivor gives birth to twins [Internet]. TMJ4. 2016. Available from: https://www.tmj4.com/news/local-news/wisconsin-rabies-survivor-jeanna-giese-frassetto-gives-birth-to-twins
- ↑ Zeiler F, Jackson A. Critical Appraisal of the Milwaukee Protocol for Rabies: This Failed Approach Should Be Abandoned. Canadian Journal of Neurological Sciences / Journal Canadien des Sciences Neurologiques [Internet]. 2015;43(1):44-51. Available from: https://www.cambridge.org/core/journals/canadian-journal-of-neurological-sciences/article/critical-appraisal-of-the-milwaukee-protocol-for-rabies-this-failed-approach-should-be-abandoned/8A47C583B24B2B2E43248770F78CC35A/core-reader
- ↑ Shehabi Y, Chan L, Kadiman S, Alias A, Ismail W, Tan M et al. Sedation depth and long-term mortality in mechanically ventilated critically ill adults: a prospective longitudinal multicentre cohort study. Intensive Care Medicine [Internet]. 2013;39(5):910-918. Available from: https://www.ncbi.nlm.nih.gov/pubmed/23344834
- ↑ Plummer J, McGettigan J. Incorporating B cell-activating factor (BAFF) into the membrane of rabies virus (RABV) particles improve the speed and magnitude of vaccine-induced antibody responses. PLOS Neglected Tropical Diseases [Internet]. 2019;13(11):e0007800. Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0007800
Edited by Yangyang Liu, student of Joan Slonczewski for BIOL 116 Information in Living Systems, 2019, Kenyon College.
