Rice tungro bacilliform virus
Baltimore Classification
Higher order taxa
Viruses; Pararetro-transcribing viruses; Caulimoviridae; Tungrovirus ;[NCBI link to find]
Species
Rice tungro bacilliform virus
|
NCBI: Taxonomy |
Description and Significance
In 1975, RTBV was first recognized and then identified as a component member (along with Rice Tungro Spherical Virus (RTSV)) of the virus complex that is responsible for causing rice tungro disease. It is found to occur in Ortyza sativa and Ortyza sp. species of rice.
RTBV is transmitted from plant to plant exclusively by the green leafhopper, Nephotettix virescens. N. virescens can acquire RTBV in 30 minutes during an 8 hour feeding and can retain infectivity for up to 7 days. Nymphs transmit more efficiently than adults and females more efficiently than males. Transmission efficiency is also affected by temperature, increasing as the temperature rises from 10 to 34 degrees C. [Anjaneyulu et al 1995]
Once the plant is infected, the symptoms induced by RTBV is stunting of the plant, red to yellow-orange discoloration, and the reduction of the amounts of grain harvested. Early infections will cause the death of the plant. While the effect of the RTBV is significant on its own, its potential for damage increases when it is combined with RTSV. There has been some genetic resistance seen in plants against RTSV, but to date there is little, if any, genetic resistance being identified for RTBV.
The virus is associated with all rice growing countries of South and South-east Asia. The effect of the virus, while found in most cultivars, will vary in its intensity.
Rice tungro disease, the most important viral disease of rice, is widespread in South and Southeast Asia and is believed to be responsible for annual losses nearing 10 billion US dollars worldwide
.
Genome Structure
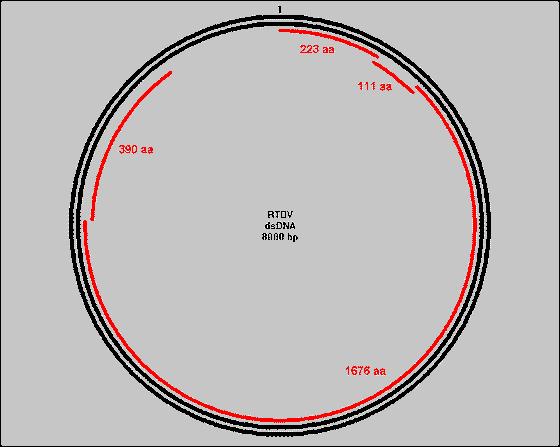
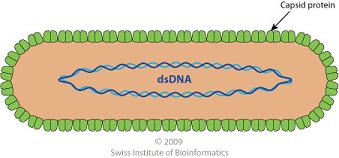
Pararetroviruses have a DNA genome. The DNA of pararetroviruses accumulates within the nucleus as multiple copies of a circular, non-segmented chromosome molecule. The RTBV genome is a double stranded DNA of 8.0 kbp with two site-specific discontinuities resulting from replication by reverse transcriptase. The RTBV genome contains four open reading frames (ORF); the function of ORF 1,2, and 4 are unknown. ORF 3 is thought to be associated with cell-to-cell movement proteins, viral coat proteins, aspartate protease, and, most importantly, the replicase comprising reverse transcriptase and RNaseH activities. However, this has not been confirmed. [Hull 1996]
Virion Structure, Metabolism and Life Cycle
Interesting features of cell structure; how it gains energy; what important molecules it produces.
The RTBV virion consists of an unenveloped capsid in a bacilliform particle shape. It is approximately 30nm in diameter and has a length of 130 nm. The virion contains circular DNA in a single molecule and is a single coat protein species.
Retroviruses have an RNA genome whereas pararetroviruses have a DNA genome; and, a proviral form of retroviruses is integrated into the host chromosome whereas the DNA of pararetroviruses accumulates within the nucleus as multiple copies of a circular chromosome The life cycle of RTBV is not well known but since its DNA structure does not intergrate into the host's DNA, RTBV's life cycle should follow the general viral reproduction life cycle: the viruses attach to the host cell, injects their contents into the cell nucleus, then change the host cell's metabolism to reproduce new viruses. Like other viruses, everything resources have to come from the host.
Rice tungro bacilliform virus (RTBV) replicates only in phloem cells in infected rice plants and its promoter drives strong phloem-specific reporter gene expression in transgenic rice plants.
Ecology and Pathogenesis
The habitat for RTBV is confined to rice cultivars, specifically Oryza sativa and Oryza sp. RTBV is restricted to vascular tissue and is found in sieve tubes of the plants. Many issues contribute to Tungro, specifically on the availability of the virus sources, population and composition of the vector, age and susceptibility of host plants, and synchronization of the three factors mentioned.
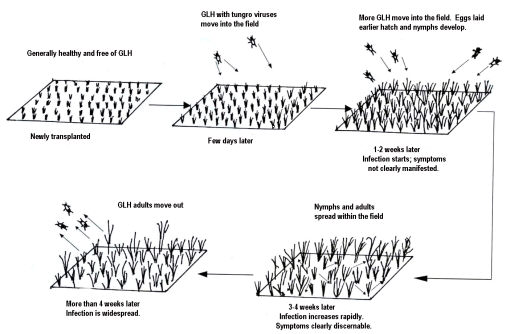
The insect acquires the virus by feeding on the plant for a short time in an 8-hr acquisition access period (minimum of 30 minutes). It can transmit the virus immediately after feeding. Either or both viruses can be transmitted during a 1 hour inoculation access period (minimum of 7 minutes).
The virus does not remain in the vector’s body. After feeding on a diseased plant, the insect can transmit the virus for about 5 days and the longest is about a week. The insect becomes reinfective after re-acquisition feeding.
Once a plant is infected there is nothing tha can be done to save it. The virus will be passed along to the other plants i n the field via the vector.
.
References
Anjaneyulu, Satapathy, Shukla. 1995 Rice Tungro. Science Publishers, Inc. Lebanon, New Hampshire. 1995. 228 pages.
Cereal Knowledge Bank. Rice Knowledge Bank. Tungro Accessed via http://images.google.com/imgres?imgurl=http://www.knowledgebank.irri.org/RiceDoctor/Seed_and_Grain_Symptoms/image194.jpg&imgrefurl=http://www.knowledgebank.irri.org/RiceDoctor/Fact_Sheets/Diseases/Tungro.htm&usg=__3wJYVX8IEv-LTEBoNdPKRXLHZzc=&h=334&w=506&sz=36&hl=en&start=13&um=1&tbnid=ntUe17eJNOoCYM:&tbnh=86&tbnw=131&prev=/images%3Fq%3Dplant%2Bi%2Bnfestation%2Bcycle%2Bof%2Brice%2Band%2Bgreen%2Bleaf%2Bhopper%26hl%3Den%26safe%3Doff%26rlz%3D1G1ACGW_ENUS321%26um%3D1 on 4/19/2009.
Herdt,RW, Khush, E, Toenniessen, G. 1991. Research priorities for rice biotechnology. Rice Biotechnology. pp. 19-54, CABI, Wallingford, UK, 1991.
Hibino, Roechan & Sudarisman, Phytopathology 68: 1412, 1978.
ICTV. ICTVdB Decriptions. 00.015.0.04 Tungrovirus Accessed via http://www.ncbi.nlm.nih.gov/ICTVdB/ICTVdB/00.015.0.04.htm on April 14, 2009.
Kunii, M, Kandra, M, Nagano, H, Ulyeda, I, Kishima, Y, Sano, Y. 2004. "Reconstruction of putative DNA virus from endogenous rice tungro bacilliform virus-like sequences in the rice genome: implications for integration and evolution." BMC Genomics 2004 5:80
Marmey, P, Rjas-Mendoza,A, de Kochko, A, Beachy, RN, Fauquet, CM. 2005. "Characterization of the protease domain of Rice Tngro Bacilliform virus responsible for the processing of the capsid protein from polyprotein" Virology Journal 2005 2:33.
Molecular-biology.suite101.com/article.cfm/viruses. Accessed via http://Molecular-biology.suite101.com/article.cfm/viruses on April 16, 2009.
NCBI Taxonomy Database. Accessed via http://www.ncbi.nlm.nih.gov/Taxonomy/ on April 18, 2009.
Pooggin, MM, Ryabbova, LA, He, X. 2006. "Mechanism of ribsome shunting in Rice tungro bacilliform praretrovirus" RNA 2006 12: 841-850.
Saito, Y., Roechan, M., Tantera, DM and Iwaki, M. (1975). Phytopathology 65: 793
Staginnus, C, Richert-Poggeler, KR. 2006. "Endogenous pararetroviruses: two-faced travelers in the plant genome." Trends in Plant Science 2006 11(10): 485-491.
Tiongco ER, Sebastian LS, Bajet NB. 1998. Manual on virus and virus-like diseases of rice in the Philippines. Department of Agriculture Philippine Rice Research Institute, Maligaya, Muñoz, Nueva Ecija. 18 p.
Tyagi, Y, Rajasubramaniam, H, Rajam, MV, Dasgupta, I. 2008. "RNA-interference in rice against Rice tungro bacilliform virus results in its decreased accumulation in inoculatedrice plants" Transgenc Research 2008, 17:897-204.
Yin, Y, Zhu, Q, Dai, C, Lamb, C, and Beachy, RN. 1997. "RF2a, a bZIP transcriptional activator of the phloem-specific rice tungro bacilliform virus promoter, functions in vascular development" EMBO J. 1997 September 1; 16(17): 5247–5259.
Author
Page authored by Trung Lam and Mark Lebednick, students of Prof. Jay Lennon at Michigan State University. A Viral Biorealm page on the family Rice tungro bacilliform virus