Roseolovirus
Classification
Viruses; dsDNA viruses, no RNA stage; Herpesvirales; Herpesviridae; Betaherpesvirinae; Roseolovirus [Others may be used. Use NCBI link to find]
Roseolovirus 6A, 6B, 7
Description and Significance
Roseolovirus is a group 1 virus according to the Baltimore classification system. This distinction indicates double stranded viral DNA.
The Roseolovirus genus includes three species: human herpesvirus (HHV)-6A, HHV-6B, and HHV-7.
This virus is capable of both the lytic and lysogenic cycles. The viral genome can integrate into the host's genome and enter the lysogenic cycle indefinitely. Certain drugs have been shown to reactivate the virus (to reenter the lytic cycle). However, natural reactivation rarely occurs in immunocompetent hosts. [4]
Roseoloviruses infect humans. Roseola infantum (aka Exanthema Subitum) is an acute illness that affects young children causing a rash and fever. [3] Roseoloviruses have also been identified in certain autoimmune diseases, like Multiple Sclerosis, though the specific role is often unknown. [2]
Other viruses in Group 1 include the family Poxviridae, the smallpox and cowpox viruses. Within the Herpesviridae family with Roseolovirus are varicella-zoster virus that causes chickenpox and Herpes simplex 1 and 2, the cause of oral and genital herpes. Group 1 viruses replicate using host or viral DNA polymerase. The virus can have an envelope or be nonenveloped and the hosts range from archaea to bacteria to humans. [1]
Structure and Life Cycle
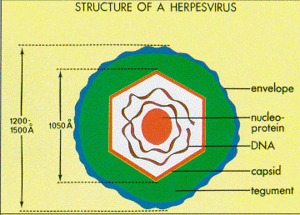
Structure- Herpesviruses have large double-stranded, linear genomes. The capsid forms an icosahedral shape. An envelope, composed of the host cell's plasma membrane, surrounds the capsid. [1]
Life cycle- HHV-6A and HHV-6B both recognize CD46 receptor, an important cell membrane protein found in host cells of the complement system of the immune response. The ubiquity of this receptor indicates the vast infection potential of the virus. However, productive infection is actually limited in vivo. [2] Other cell receptors are also used for binding and infection.
When the virus binds a host cell receptor, the envelope fuses with the plasma membrane and the capsid is released into the cytoplasm, where it is dismantled. The viral DNA is then free to move into the host cell's nucleus. The double-stranded DNA can integrate into the host's genome. [1]
Herpesviruses are capable of both lytic and lysogenic cycles. In the lytic cycle, the host's replication, transcription and translation machinery are used by the virus. The hijacked machinery is used to make many copies of the viral DNA and capsid. The capsid is wrapped in the host's plasma membrane as it leaves the cell. This becomes the envelope. The newly made virus now moves on to infect another cell. [1]
In the lysogenic cycle, the viral genome remains dormant in the host's genome until a signal triggers the initiation of the lytic cycle. This signal is unknown/disputed but certain drugs have been found to induce initiation of the lytic cycle. [4]
Studies on HHV-6A and HHV-6B have shown that the viruses target cells of the immune system, primarily CD4+ T cells. For example, infection of the T cell can cause the downregulation of CD3, which makes the T cell unable to activate by interaction with antigen-producing cells. The viruses can successfully complete lytic cycles in lymphoid cells. B cells are not usually infected. Dormant HHV-6A and HHV-6B viral genomes have been found in dendritic cells. [2]
Pathogenesis
Roseoloviruse HHV-6B can cause roseola infantum (aka Exanthema Subitum) in children under the age of three. Symptoms include a high-grade fever and a rash originating on the trunk. [3] The severity of the fever can induce seizures. Treatment is often limited to fever-reducing medications like acetaminophen and ibuprofen as the rash and fever resolve naturally in a few days. Sedatives may be administered to patients who are susceptible to seizures. [5]
Because herpesviruses can integrate into the host's genome, reactivations can occur in immunocompromised hosts. These symptoms include rash, fever, seizures, encephalopathy, limbic encephalitis, amnesia, cognitive dysfunction, lymphadenopathy, colitis and hepatitis. [4]
Other associations between HHV-6A and HHV-6B and several autoimmune disorders have been investigated. These studies produced results with varying degrees of evidentiary support. [2]
Although there is no evidence of HHV-6A playing a role in the pathogenesis of Multiple Sclerosis, active HHV-6A infection was found in the blood and cerebrospinal fluid of patients with relapsing MS. Other indications of HHV-6A infections were also discovered in MS patients. [2]
References
[1] Slonczewski JL, Foster JW. "Microbiology, An Evolving Science." 2nd Ed. New York: W.W. Norton and Company, Inc., 2011.
[2] Dagna L, Pritchett JC, Lusso P. Immunomodulation and immunosuppression by human herpesvirus 6A and 6B. 2013. Future Viriology. 8:(3) 273. http://go.galegroup.com.ezp1.lib.umn.edu/ps/retrieve.do?sgHitCountType=None&sort=RELEVANCE&inPS=true&prodId=EAIM&userGroupName=umn_wilson&tabID=T002&searchId=R1&resultListType=RESULT_LIST&contentSegment=&searchType=AdvancedSearchForm¤tPosition=1&contentSet=GALE%7CA321112157&&docId=GALE%7CA321112157&docType=GALE&role=&docLevel=FULLTEXT
[3] Exanthem Subitum. Reference.MD. 2012. http://www.reference.md/files/D005/mD005077.html
[4] Pellett PE, Ablashi DV, Ambros PF, et al. "Chromosomally integrated human herpesvirus 6: questions and answers." 2012. Rev. Med. Virol. 22: 144-155. http://onlinelibrary.wiley.com.ezp1.lib.umn.edu/doi/10.1002/rmv.715/pdf
[5] Kohl, Steve. "Human Herpesvirus 6." in Nelson Textbook of Pediatrics, edited by Richard E. Behrman. Philadelphia: W.B. Saunders Co., 1996. http://medical-dictionary.thefreedictionary.com/Roseolovirus+infections
Author
Page authored by Morgan Schwoch, student of Mandy Brosnahan, Instructor at the University of Minnesota-Twin Cities, MICB 3301/3303: Biology of Microorganisms.