Stream biofilm
Introduction

A biofilm is a complex aggregation of microorganisms growing on a solid surface. Biofilms are usually found on solid substrates submerged in or exposed to some aqueous solution. A stream biofilm is the layer on solid surfaces in a stream, such as rocks, plants and other surfaces. This layer is composed of microscopic bacteria, algae, and protozoans in a complex polymer linked assemblage.
Stream biofilms play an important ecological role in their environment. For one thing, they play a major role in the bottom up supply of energy and organic matter to the food web. Organisms within the stream biofilm can use dissolved organic matter, fix nitrogen or recycle organic nitrogen, fix energy and carbon by photosynthesis and chemosynthesis, and they can serve as food resource for organisms of higher trophic levels. For example, it has been found that many invertebrate species gain their nutrients from the microbial biofilms in streams in addition to plant debris. For another, stream biofilms are important in recycling organic matter. Microbial biofilms in aquatic surfaces are actively involved in degradation of plant and animal debris and cycling of nutrients, and thus are beneficial for aquatic ecosystem maintenance. Another function of biofilms in aquatic environment is that they can remove suspended sediment and thus contribute to water quality control.
Physical environment
Characteristics of biofilm matrix

The biofilm matrix is a dynamic environment in which the component microbial cells and are optimally organized to make use of all available nutrients. The major matrix components are microbial cells, polysaccharides and water, together with excreted cellular products. The matrix shows great microheterogeneity, within which numerous microenvironments can exist. Although exopolysaccharides provide the matrix framework, a wide range of enzyme activities can be found within the biofilm, some of which will greatly affect structural integrity and stability. Much of the biofilm matrix – perhaps up to 97% – is actually water. The water can be bound within the capsules of microbial cells or can exist as a solvent whose physical properties such as viscosity are determined by the solutes dissolved in it. Apart from water and microbial cells, the biofilm matrix is a complex of secreted polymers, absorbed nutrients and metabolites, products from cell lysis and even particulate material and detritus from the immediate surrounding environment. Thus, all major classes of macromolecule – proteins, polysaccharides, DNA and RNA – can be present in addition to peptidoglycan, lipids, phospholipids and other cell components. The amount of cellular material within a biofilm can itself vary greatly. Table 1 shows range of composition of biofilm matrices[1].
Factors affecting stream biofilm formation
In freshwater systems, individual species of bacteria can occur as both planktonic (free-floating) and benthic (attached to the substratum) organisms. The equilibrium between these two states depends on environmental factors such as movement of water and substrate availability. Stream biofilms are composed of nutritionally diverse microorganisms, and many environmental factors can affect the biofilm structure and community composition These factors include temperature, depth, width, flow rate, pH, dissolved oxygen, nutrients, light availability, etc. Nutrients in streams are essential for growth, and the concentration of nutrients and the form they are found change continually. The variation of nutrient content can depend upon land use and many other factors of watershed areas. For examples, nutrient input may increase due to fertilization of cropland or lawns and gardens; high rainfall may cause increased wash-off of organic matter such as leaves, twigs, grass, and other debris. Because decomposition of this organic matter releases nutrients, it constitutes an important source of nutrient loading. If the stream is fed by a lake or other water source with naturally high variations in nutrient concentrations, the stream will reflect the same variations too. Also, light can enhance the algal and autotrophic bacterial growth, as well as higher extracellular enzyme activity. One study showed that the presence of light may alter the biofilm composition and function, therefore affecting the processing capacity of organic matter in the stream ecosystem [12]. In all, environmental conditions in streams are characteristic of dynamic temporal and spatial change,which has complex influence on biofilm structures and compositions.
Stream environment
A stream is a body of water with a detectable current, confined within a bed and banks. Streams are inherently heterogeneous and are characterized by a largely unidirectional downstream flow of water that controls the dispersal of suspended microorganisms, biofilm community composition, architecture, and metabolism.
Biological interactions
Stream biofilm composition and interactions
Stream biofilms are made up of bacteria, archea, fungi, filamentous and single cell algae and diatoms all interspersed among a polymeric matrix produced by the constituent organisms. On and within the biofilm are protozoa, nemotodes and the larvae of macrobentic invertebrates which graze on the autotrophic and saprophytic components. Very efficient cooperations and syntrophic interactions can evolve within a biofilm. In fact, biofilms provide an ideal environment for the establishment of syntrophic relationships. Syntrophism is a special case of symbiosis in which two metabolically distinct types of bacteria depend on each other to utilize certain substrates, typically for energy production. For example, in the degradation of complex organic matter into methane and carbon dioxide, fermentative bacteria initiate the catabolism, producing acids and alcohols that are then readily utilized as substrates by acetogenic bacteria. Finally, the methanogens obtain energy from converting acetate, carbon dioxide, and hydrogen to methane. Also, other microbial interactions such as synergism, competition, predation and commensalism also develops in aquatic biofilms.
Food web interactions
There are also important food web interactions within biofilms or between biofilm organisms and other aquatic organisms. Within stream food webs, biofilms are intensively grazed by protozoa, macrobenthic invertebrates and some fish. Then the biofilm composition will be modified by the predation, as well as physicochemical characteristics of the environment. In turn, the specific microbial composition of the biofilm will modify the nutritional quality of the material for grazing species, which along with habitat factors, will influence the population level and composition of grazing organisms and so on through the food chain.
Benefits of biofilm interactions
Formation of biofilms has been recoganized as the dominant mode of microbial life in most aquatic ecosystems, for biological interactions in biofilms provide microorganism a number of benefits. Bacteria within biofilms live within a matrix of excreted polymeric compounds. This matrix protects the cells within it and facilitates communication among them through chemical and physical signals, as well as enables bacteria to remain within a favorable environmental niche. For example, microorganisms in biofilms may withstand nutrient deprivation, pH changes, oxygen radicals, disinfectants and antibiotics more than planktonic bacteria.
Microbial processes
Nutrient transformation
Many different microbial processes in stream biofilms facilitate nutrient cycling between the biofilm and aquatic environment as well as within the biofilm. Algae and cyanobacteria carry out photosynthesis and capture light energy and produce biomass. Heterotrophic inputs include dissolved, colloidal and fine particulate organic matter. Within the matrix, extracellular release and cell death result in enzymes and other molecular products that are retained due to reduced diffusion rates, and available for utilization by other microorganisms. Nitrogen fixation and conversion is also important processes and contribute to microbial growth and nutrient cycling.
Cell-to-cell communication
Another characteristic process found in a biofilm is that bacteria can communicate using chemical signals. The chemical signals produced by cells and passed through their outer membranes may be sensed by members of the same species and other microbial species that are part of the same biofilm community. The sensing of these chemical signals by neighboring cells in the biofilm can cause the neighboring cells to behave differently different by causing different genetic expression to occur in those cells. Within the biofilm community, bacteria communicate with each other by using chemical signal molecules in response to population density in a process that is called quorum sensing [2]. The cell–cell communication via QS involves the production, release, detection, and response to small hormone-like molecules termed pheromones or autoinducers (AIs). During growth, bacteria produce the AIs, which activate the QS system upon reaching a threshold concentration. Cell communication play an important role in biofilm formation and development.
Gene exchange
Horizontal gene transfer is facilitated by physical interactions called coaggregation and coadhesion in biofilm matrix. Coaggregation refers to interactions occur among suspended cells, while coadhesion refers to interactions between suspended and already attached cells in biofilms. Interactions among bacterial cells of genetically distinct cell types enable genetic exchange in the biofilm and acquisition of new genetic traits.
Key Microorganisms and Examples
Overall microbial community composition
Diverse microorganisms inhabit in stream biofilms, including groups of bacteria, archea, fungi, filamentous and single cell algae and diatoms, and protozoa.
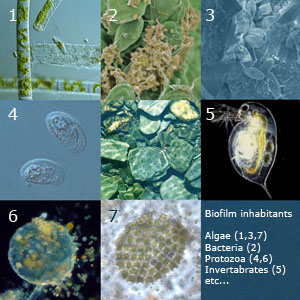
Algae and Cyanobacteria
Algae and Cyanobacteria are the primary producers in stream biofilms. The Chlorophyta or green algae consist of about 7,000 species, most of which occur in fresh water. Some common examples of green algae include the unicellular genera Chlamydomonas and Chlorella. Examples of Cyanobacteria found in a great number in a human impacted stream include members of the genera Oscillatoria, Anabaena and Aphenizomenon[3].
Actinomycetes
There are varieties of bacteria and fungi in streams biofilms and the community composition greatly vary depending on the living environment. Actinomycetes contribute significantly to organic matter processing. Actinomycetes are a group of gram-positive bacteria (Class Actinobacteria) characterized by a high G_C content that perform a wide array of important functions in various habitats. In many streams, leaf litter is an important source of energy and carbon. Actinomycetes secrete enzymes that can break down large molecules, such as cellulose, chitin, and lignin, into smaller compounds that can be taken up by the biota. Actinomycetes examples include Actinokineospora, Streptomyces, Nocardiodes, Pseudonocardia, Nocardia, and Micromonospora. They have been found on decomposing plant litter.
Protozoa
Protozoa such as amoebae, ciliates and flagellates are found virtually everywhere that liquid water is present, including stream biofilms, and are important components of aquatic ecosystems.They are major consumers of bacteria, and are an important food source for other protozoa and animals (such as rotifers, insects and fish larvae). They have important roles in carbon cycling and mineralization of nutrients in microbial food webs and contribute to decomposition of leaf litter in streams. Ciliates are commonly found protozoa in stream biofilms, and they live on eating bacteria and algae, as well as other protozoans. Respiration of Ciliates can be anaerobic (occurring without oxygen) or aerobic (requiring oxygen). Ciliates commonly found in stream biofilm are diverse, and examples include Oligohymenophorea, Phyllopharyngea, Spirotrichea, Colpodea, Litostomatea. Paramecuim (Paramecium caudatum), Stentor (Stentor coeruleus), etc.
Macroinvertebrate
Different macroinvertebrate can also be found within and on stream biofilms, primarily functioning as predators, and they impact the microbial community structure, composition and diversity. Examples of macroinvertebrate in or near stream biofilms include mayfly (e.g. Deleatidium), stonefly (e.g. Zelandobius and Acroperla), Caddisfly, and midge larvae.
Current Research
Stream biofilm microbial communities and affecting factors
Streams are inherently heterogeneous and have continually changing conditions. So microorganism abundance and distribution in stream biofilms are characteristic of temporal and spatial variations in their community composition [5], architecture [6], and metabolism [8]. Therefore, many recent research studies focus on diversity of microbial community composition and change in response to environmental factors. As an example, Bacteria are generally influenced by DOM availability and composition in streams, many studies have examined the influence of DOM and nutrients on microbial compositions in biofilms. While most study focused primarily on algae, Olapade et al studied the influence of DOM and inorganic nutrients on bacteria [10]. The study investigated the response of stream biofilm bacteria to DOM (glucose, leaf leachate, and algal exudates) and inorganic nutrients (phosphate and nitrate alone and in combination), under controlled laboratory conditions, also how individual phylogenetic groups in biofilm are affected was analyzed by molecular biology techniques. The results, together with other studies from the research group [9, 11] suggest that there are interactions among the different bacterial groups in biofilms that are impacted by the associated nutrient dynamics among seasons in stream ecosystems.
Stream biofilm formation and dispersal
Dispersal and colonization are important for the assembly and biodiversity of microbial communities. Though dispersal mechanisms has become increasingly understood in model bacterial biofilms, the colonization and dispersal processes in complex biofilms and the relevant controlling factors remain elusive. In a recent study[4], complex biofilms in stream microcosms were grown and investigated. Specifically, dispersal and colonization patterns of fluorescently labeled cells and microbeads in nascent and mature biofilms were studied, under conditions of laminar and turbulent flow. The research showed that the transport of disperser cells onto and into biofilms were primarily influenced by hydrodynamics and resident biofilm topography, but attachment processes were influenced by the biological properties of the dispersers. This original study suggested an experimental approach to perform mechanistic studies of complex biofilms relevant in natural aquatic environment.
Theories for biofilm research
While ever-improving biology approaches and techniques are greatly facilitating research in biofilms and provide insights into the microbial community composition, structure and function, there is a great need for ecological theories compile various concepts and models in biofilm research. Battin and colleagues proposed a perspective and research approach from the view of landscape ecology theory[7]. As they suggested, the concept can contribute to understanding pathways of biofilm development and thus help manipulate the growth of biofilms so as to address critical ecological and environmental question.
References
Edited by student of Angela Kent at the University of Illinois at Urbana-Champaign.