Tetrahymena thermophila
Classification
Higher order taxa
Domain: Eukaryota; Kingdom: Chromalveolata; Phylum: Ciliophora; Class: Oligohymenophorea; Order: Hymenostomatida; Family: Tetrahymenidae
Species
|
NCBI: Taxonomy |
- Tetrahymena thermophila
Description and Significance
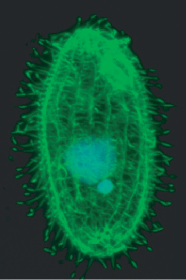
Tetrahymena thermophila is a large, motile, phagocytic, unicellular eukaryote. The organism is about 20 x 50 µm. T. thermophila live in temperate freshwater environments. With a doubling time of less than 2 hours, it is considered one of the fastest growing eukaryotes. It can readily grow to a high density on a wide range of media, with a temperature range of 12 °C to 41 °C.
T. thermophila is an essential model organism that has been used to study biological phenomena which includes the following: cilia biogenesis, telomerase structure and function, small RNA mediated self versus non-self discrimination, and epigenetic inheritance.
Genome Structure
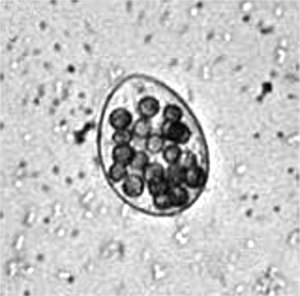
T. thermophila contain two nuclei, a macronucleus and a micronucleus. Each has been sequenced[1]. The macronucleus is ultimately derived from the micronucleus. The micronucleus is diploid and contains 5 pairs of chromosomes. The macronucleus contains 45 copies of 275 chromosomes formed by fragmentation, as well as 9000 copies of the rRNA gene. The macronucleus contains 15% less genetic information than the micronucleus, which represents noncoding DNA and transposable elements. The macronucleus is kept intact through asexual reproduction by telomerase activity. Additionally the mitochondrial genome has been sequenced [2].
Cell Structure
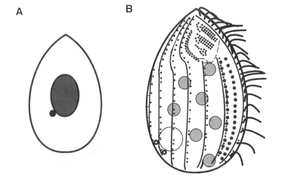
Tetrahymena thermophila has all the basic structures of animal cells (ER, golgi, mitochondria, actin, tubulin etc.) except intermediate filaments. Additionally T. thermophila contain enlarged food vacuoles and a contractile vacuole which aids in regulating osmotic pressure. The cell has 18-21 rows of cilia lining its cell membrane. Over 50% of the membrane is replaced by another class of lipids called phosphonolipids. Instead of the phosophorus-oxygen-carbon bond characteristic of phospholipids, phosphonolipids have a direct phosphorus-carbon bond and are resistant to the enzyme, phospholipases. Since phospholipases are secreted by T. thermophila, phosphonolipids prevent the degradation of its own membrane. Food enters the organism through the oral apparatus and exits through the cytoproct. The cytoproct has microtubules that reach and drape over food vacuoles or electron dense material that approach it [3].
Metabolism
T. thermophila is a chemoorganoheterotroph which obtains energy through aerobic respiration. In lab T. thermophila's generation time is between 2-3 hours. It requires 11 essential amino acids, six B-complex vitamins, Fe3+ and trace metals. A culture of bacteria is also sufficient to support T. thermophila. Additionally T. thermophila is unable to synthesize purines and pyrimidines. The lack of rudimentary synthetic pathways reflects the obligate predatory lifestyle of T. thermophila in nature.
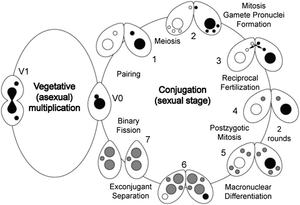
Life Cycle
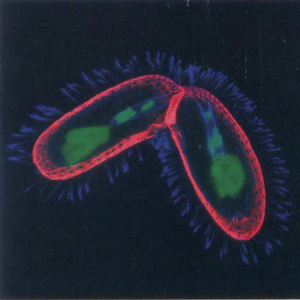
T. thermophila has two different types of nucleus that perform distinct functions. The presence of these two nuclei reflects T. thermophila’s ability to reproduce both sexually and asexually. The diploid micronucleus is the vegetative germ-line during which the macronucleus is transcribed, translated and transmitted to the next asexual generation by mitosis. It is predicted that the 120 Mb genome has about 27,500 predicted open reading frames, but is transcriptionally silent in the vegetative state. Food stress induces T. thermophila to reproduce sexually, producing pronuclei by meiosis of the micronucleus and exchange with a cell of a different mating type. The highly evolved macronucleus has the ability to differentiate from a mitotic sibling. Without the process of cell division, sexually mating cells produce a zygotic genome with the macronucleus ultimately derived from the micronucleus [4].
T. thermophila has four distinct phases in its life cycle: conjugation, immaturity, maturity, and senility. Conjugation is the sexual stage of the life cycle, in which two cells pair to form a temporary junction to exchange gamete nuclei. The mating cells further generate and differentiate the necessary nuclear structures for their sexual progeny. During conjugation the essential nuclear events include meiosis, gamete nucleus formation, fertilization and nuclear differentiation [5].
Ecology
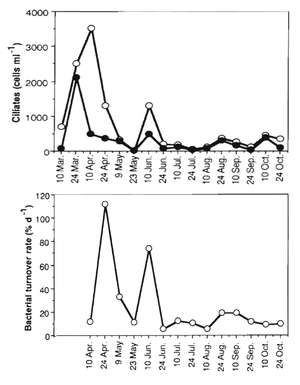
Tetrahymena thermophila is common in fresh water ponds throughout the eastern United States, particularly those that do not completely ice over during the winter months. T. thermophila distribution is very localized; this contrasts with the idea that "everything is everywhere". The organism is most abundant June through September. It is often found in proximity to decaying vegetation.[6]
Ciliates such as T. thermophila are important consumers of bacteria in lakes. They also constitute an important link between bacteria and metazoan zooplankton. The microbial loop in lakes is not well understood. Ciliates may cause significant turnover of bacteria in lakes, but this has yet to be elucidated.
References
http://www.jbc.org/content/255/9/4198.full.pdf
Author
Page authored by Hannah Pak and Luke Pryke, students of Prof. Jay Lennon at Indiana University.