The Burden of C. difficile and its Link to Nosocomial Infections
Introduction
By Christi Binkley
Clostridium difficile infections are the leading cause of hospital-associated infections (HAIs), or nosocomial infections, in the United States. The infections are becoming increasingly serious, and the presence of hypervirulent strains is causing epidemics estimated to cost the USA healthcare system approximately $3.2 billion annually (22). This breaks down to $3,006-$15,397 per episode (23). These new strains are resistant to fluoroquinolones, and produce more spores and toxins than what has been studied historically (22). It is important to study this bacteria, as it is becoming an increasing risk to public health. The Center of Disease Control is working to prevent C. difficile infections by incorporating its State Antibiotic Resistance Programs across the country, with a goal of reducing the number of incidences of infection by half (26).
Types of HAI
Central line-associated bloodstream infections (CLABSI)
Vascular catheters are used frequently in the United States for a variety of medical purposes, including the administration of medicine, nutritional fluids, or conducting medical tests. They are especially used in intensive care units (ICUs) as central vein catheters are in closer contact to the heart compared to the familiar IV (5). However, these catheters present a risk of bloodstream infection; bacteria or viruses may colonize on the internal surface of the catheter where fluids are being transported, or on the external surface of the catheter. These are serious infections, and symptoms may include a fever, chills, or sore, red skin around the site of infection (3).
Catheter-associated urinary tract infection
Urinary catheters are tubes inserted through the urethra into the bladder to drain urine, and are received by 15-25% of hospital patients (6). When bacteria are present on the catheter, at the time of insertion, or on the skin surrounding the insertion site, a urinary tract infection (UTI) may occur. Signs of infection include lower abdomen pain, fever, or burning sensation while urinating. Risk of infection is increased the longer the catheter is in place, as bacterial biofilms may grow and eventually colonize the patient’s skin, tissue, or organs involved in the urinary tract (8).
Surgical site infections (SSI)
Surgical site infections are infections that develop at the site of recent surgery. It is estimated that 1 to 3 out of every 100 patients undergoing surgery develop an SSI (2). These infections range in intensity and can be caused by a variety of bacteria. SSIs may occur topically on the skin surrounding the surgery site, in the muscle or tissue beneath and surrounding the surgery site, or in organs or implant material. The infections occur due to bacteria such as staphylococcus, streptococcus, and pseudomonas that come in contact to the surgery wound. This may occur via contaminated surgical instruments, contact from the doctor or caregiver, from microbes already present on the patient’s skin, or from airborne microbes. Factors that increase the risk of SSIs include surgeries lasting over two hours, old age, smoking, and any pre-existing diseases or reasons for a compromised immune system (1). Signs of infection include fever, pain and swelling around the area of the surgery, and drainage of cloudy fluid (pus) from the surgery wound. Pus can be cultured to identify the types of bacteria causing infection, and therefore identify the best treatment option. These infections may require patients to remain in the hospital for as long as seven extra days (1).
Ventilator-associated pneumonia
Ventilators are used to deliver oxygen to a patient, using a tube through the patient’s mouth, nose, or hole in the front of the neck. Diseases, most commonly being ventilator-associated pneumonia (VAP), can occur when bacteria enter the lungs and lower respiratory tract. This may be due to biofilms forming on the tube connected to the ventilator, in the patient’s neck and lungs, or via air transmission at the site of tube insertion. Unfortunately, this is extremely common because patients requiring ventilators are often immunocompromised and have underlying medical conditions. Most of the causative bacteria are gram-negative bacteria, however gram-positive cocci also contribute significantly to VAP (10).
Clostridium difficile
C. difficile is a gram-positive firmicute that is found in a wide variety of environments including water, air, feces, and soil (Figure 1). It is anaerobic, can form endospores that may survive for up to two years when conditions are unfavorable, and produces endotoxins (10). It has been found that C. difficile prioritizes growth over toxin production, therefore it is able to grow and colonize, then release toxins in a larger quantity (10). Infection by C. difficile involves a range of physical symptoms, including mild to severe diarrhea, toxic megacolon (infection and inflammation of the colon), and even death in extreme cases. Specifically, about 5-10% of cases end in mortality, averaging to an estimated 14,000-20,000 deaths annually in the US. C difficile is extremely prevalent in the US; however, it is also found worldwide (15).
There are many reasons why C. difficile is the most common bacteria in nosocomial infections. Inherently, its optimal growth temperature is that of the human body, therefore allowing it to thrive in a human host. It is unaffected by many antibiotics, therefore when a patient takes an antibiotic, the healthy gut flora die and C. difficile no longer has competition for nutrients or space. Also, the genome of C. difficile has been sequenced, and researchers found that a large region of the genome codes for mobile genetic machinery such as transposons used in conjugation. This increases the rate of successful gene incorporation into the host cell’s genetic material (10).
Furthermore, much of C. difficile’s virulence is regulated by its environmental conditions. For example, surface protein expression is upregulated in times of stress, allowing it to stick to its host cells more strongly. It is also able to upregulate toxin production when temperature increases; this adaptation allows the bacteria to reduce energy expenditure by only producing toxins when in contact with host cells (10).
In hospitals
In a study looking at C. difficile’s presence as a hospital-acquired infection, it was found that the most commonly contaminated surfaces in the hospital included the bathroom handrails, toilet seats, and tray tables (11). This is expected, as C. difficile are found in human feces and therefore in bathrooms. They are transmitted often in spore form on the hands of doctors and other medical personnel (12). The most common way to diagnose a patient with a C. difficile infection (CDI) is to take a stool sample and test for the presence of the bacteria or its toxins. Hospitals and labs that test for C. difficile must report to Infection Prevention and Control daily (23).
C. difficile is similar to other prevalent hospital pathogens, such as MRSA and vancomycin-resistant enterococci. They all inhabit the skin and are transmitted between healthcare providers and patients. However, C. difficile is distinct in that it forms spores, which are resistant to alcohols and common hospital cleaning agents. Also, C. difficile has not been shown to be affected significantly by hand washing or use of alcohol hygiene products, whereas these alcohol products are very effective against MRSA and other organisms that do not form spores (23).
In nursing homes
Especially vulnerable to C. difficile infections are those in nursing homes. Many people living in nursing homes are attempting to live a healthy lifestyle and not require hospitalization; however, pathogens such as C. difficile can cause severe complications in elderly people, many of whom are immunocompromised due to old age or a preexisting medical condition. Infections can cause malnutrition, physical weakness and frailty, or possible hospitalization for elderly individuals. Over 80% of C. difficile associated deaths are in Americans older than 65 years, and more than 100,000 infections occur annually in this age bracket of nursing home residents (26).
Some regions may experience high transmission rate between hospitals and the nursing home. Holding nursing homes to similar standard as hospitals, in regards to sanitation measures and reporting infection rates, can help decrease the presence of C. difficile. There are currently efforts to create prevention programs, with goals such as allocating more money to education and prevention of C. difficile infections, reducing hospital transmission of infections, and designing reports and standards that nursing homes must follow when diseases and infections are diagnosed. Proposals have also been made to penalize hospitals for excess C. difficile infections, as incentive to reduce the infection prevalence (25).
Toxin Production
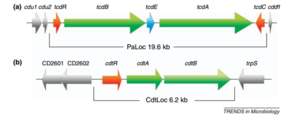
C. difficile produces two different toxins, toxins A and B, which are coded by the genes tcdA and tcdB. These genes, in addition to accessory genes tcdC, tcdR, and tcdE, comprise the pathogenicity locus, or PaLoc, on the chromosome (Figure 2) (21). It is thought that a nonsense mutation of tcdC is a main cause of the increased toxicity in current C. difficile strains (22). There are currently 31 variations of the PaLoc known to exist in nature; these are known as toxinotypes. Recently, a third toxin has been isolated, called binary toxin or CDT. CDT is encoded by the genes cdtA and cdtB which are not in the PaLoc, but rather at its own CDT locus. The role of this third toxin is a current area of research, however recent studies are suggesting that CDT aids C. difficile in adhering to host cells by inducing formation of protrusions that are microtubule-based (21, 23). This implies that CDT is important for colonization of hosts and eventual onset of infection (23).
Toxin A and toxin B cause host cell death by catalyzing the glucosylation of Rho-GTPases. These are small regulatory proteins that aid in the organization of the cell’s cytoskeleton (21). In infected humans, the results in often severe colonic inflammation and tissue damage. Fluids flow rapidly into the intestinal lumen, which is then flushed out as diarrhea (22). To understand the effects of toxin A and toxin B independently, knockout lines were grown with three different toxin profiles: A+B- only producing toxin A, A-B+ only producing toxin B, and A-B- the double knockout (the wild type is A+B+). Knockouts were made by placing an intron in the DNA sequence coding for the toxin, rendering the cell unable to use that respective gene (21).
The specific functions of toxin A and toxin B independently seem to vary across studies, likely due to subtle differences in strains of C. difficile used, and what cell lines were used to test virulence. Some studies have found that toxin A alone is able to produce symptoms of C. difficile infection and toxin B requires the presence of toxin A to be virulent, whereas other studies that found the opposite to be true (21). It was originally believed that Toxin A was the main cause of virulence, when testing the purified toxin on hamsters and studying the infection symptoms. Administration of toxin B alone only lead to virulence when small concentrations of toxin A were present, suggesting the toxins work synergistically (22). Furthermore, previous studies showed that treatment using vaccines with toxin A toxoids protected animals fully from C. difficile infections. Later, in vivo studies began showing that toxin A was not necessary for toxin B to have virulence. Hospitalized patients showing full signs of infection were found to be infected with strains of C. difficile that were A-B+. How these strains became prevalent and why they contrast past studies is currently unknown (22).
Strains and Identification
Techniques used to identify strains of C. difficile include restriction endonuclease analysis (REA), PCR-ribotyping, and pulsed-field gel electrophoresis (PFGE). Identifying the strain of C. difficile present is important for proper treatment of disease outbreak or sanitation measures, however culturing and identification takes time. Currently, the bacteria are isolated from stool samples then sent to a reference lab to identify the strain. Not many public health labs offer this service, presenting another issue for fast identification. It is important that as many strain-identification techniques are used as possible, as each technique groups the strains slightly differently, and therefore can be further identified via confirmation from another method. The strains within the 027/NAP1/BI group are the most epidemic in the United States (15).
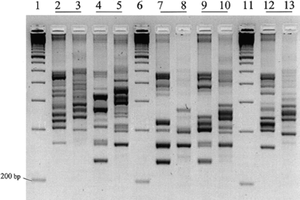
REA is a technique that uses point mutations to differentiate strains. The mutations are identified on a gel after completing PCR for the target sequence (16). This method works well at discriminating on a gel, however interpretation of the results is often subjective (19). PCR-ribotyping uses PCR to amplify the rRNA operon encoded in C. difficile’s DNA. The operon includes the space between the 16S and 23S rRNA genes on the chromosome, which may differ in sizes as small as 10 nucleotides across C. difficile strains. Currently, the PCR products are often large and difficult to distinguish on a gel. Research is being done to discover primers that more closely surround the rRNA operon, hence producing smaller PCR products that will distinguish themselves more clearly on a gel (Figure 3) (17). It is a quick and easy method, however due to lack of standardization across labs regarding the primers used, the results are difficult to compare (19). Finally, PFGE requires the digestion of restriction enzymes (smaI, KspI, SacII or NruI), then separates the DNA fragments to analyze differences in fragment length. PFGE is very time consuming, taking from 4-5 days, and the equipment is expensive. New methods currently being explored include multi-locus sequence typing, and surface-layer protein A sequence typing (19).
Treatment
Infections due to C. difficile usually cause the victim to have a fever, loss of appetite, abdominal pain, and diarrhea. The most common treatment is taking antibiotics orally for approximately ten days. Appropriate antibiotics include metronidazole, vancomycin, and fidaxomicin (12). Metronidazole does not have FDA approval for the treatment of C. difficile infections, however it has been shown to be effective in less severe infections (24). Other antibiotics should not be taken, as they may eliminate healthy microbe communities and give C. difficile an advantage. Unfortunately, in about 20% of patients who are treated for a C. difficile infection using antibiotics, the infection returns. Antibiotics can be used again to treat the infection, however recent practice has found that fecal transplants are successful alternatives (12). Many patients who are cured of a C. difficile infection will continue to have small amounts of the bacteria in their stools for multiple weeks after treatment (23).
Prevention
CLABSI Prevention
Extensive sterilization measures and precautions are being used to reduce the instances of CLABSIs. Patients are often covered in a sterile sheet, and their skin is cleaned with antiseptic at the site of catheter insertion (4). The healthcare providers should wash their hands and arms, in addition to wearing gloves. This reduces the risk of transmission via person-person contact. Minimizing the amount of time that the patient is using the catheter also helps reduce the risk of infection (4).
Catheter-associated UTI
Sterilization is again the most important precaution to take in avoiding a UTI from the use of a urinary catheter. Cleaning the skin area surrounding the insertion site is crucial, along with making sure the catheter is inserted correctly and not bumped or moved significantly throughout the day. As soon as the patient no longer needs the catheter, it should be removed to minimize opportunities for contact with pathogens (13).
SSI prevention
Preventing SSI’s also requires complete sterilization of all tools used in surgery, including the hands and arms of the operating doctor/surgeon. Medical personnel must take extra precautions, such as washing their hands after every interaction with an infected patient, in order to minimize the spread of C. difficile (2). Hospital guests are also at risk of contracting C. difficile and spreading the bacteria, therefore visitors and guests must take the same precautions.
Ventilator-associated pneumonia
Cleaning the ventilator equipment, tubes, and other accessories is necessary to reduce the presence of C. difficile and other pathogens that may be present. Using mouthwash and keeping the inside of the patient’s mouth clean may also aid in prevention (14). Because a ventilator allows for airborne pathogens to have easier access to a patient’s lungs, it is also important to have quality air ventilation systems in the room or building. This will reduce the risk of disease in both patients and the healthcare providers.
General prevention measures
There are many simple sanitation precautions that can be observed, however they are often overlooked. To prevent patients from exposure to C. difficile, handles of common medical equipment such as thermometers should be sanitized immediately after use. When possible, disposable instruments are ideal because they are often sterile in their packaging, and eliminate patient to patient transmission via shared medical tools. Gowns and gloves should be worn to reduce skin to skin contact between any individuals. Bleach and other strong cleaners should be used on the entire medical examination room, all equipment, doorknobs, restrooms, and any place an infected individual or exposed healthcare provider was present. No-touch approaches for disinfection are also available. This usually involves hydrogen peroxide vapor or UV-light, which have both been shown to kill C. difficile spores. Alert systems can also be put into place to notify healthcare providers in an area or department that there has been a new diagnosis of C. difficile infection. Awareness will allow more people to take precautionary measures and hence decrease transmission (23).
Current Research
A large area of current research regarding C. difficile infections (CDIs) is tracking and modeling the prevalence of infections across the globe. By understanding how CDI prevalence changes across locations and with respect to environmental factors, we can better understand the exact factors that drive CDIs. This will allow for appropriate prevention and control strategies to be employed.
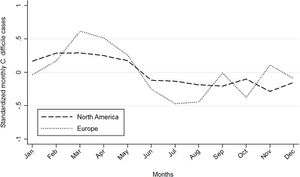
A recent study looked at seasonal variation, on a global scale, as an environmental factor that may be a factor in increased risk of contracting a CDI. The researchers complied data from 20 different studies on human CDI incidence around the world for analysis.They found that CDI prevalence was similar in the northern and southern hemispheres, where CDI prevalence peaks in the spring (March through April) and decreases over the summer and into autumn. This pattern was also found across North America and Europe, where there was no significant difference in CDI prevalence found between these two regions (Figure 4). A likely explanation as to why CDIs peak in the spring is tied to antibiotic consumption. In the winter, the use of antibiotics is higher than at any other time of the year, due to an increased number of respiratory infections and other illnesses (27). The antibiotics consumed wipe out a large number of bacteria in the guts and colon, however C. difficile spores are resistant to antibiotics, and therefore can proliferate and colonize when competing bacteria are eliminated (28). There is a lag time between the consumption of antibiotics and the onset of CDI, hence why springtime is when the prevalence peaks rather than winter. This study also noted that regions farther from the equator have more prominent seasonal patterns in antibiotic consumption, due to more seasonal differentiation in these regions. This means that predicting CDI onset in these areas is more easily accomplished (27).
Other areas of current research include exploring treatments for CDIs, such as fecal bacteriotherapy (28), and modeling the locations and demographics of people affected by CDIs in order to assess risk factors. Understanding the driving factors of CDI is essential to developing prevention and control strategies.
References
(1) The Johns Hopkins University. "Surgical Site Infections."Web. <http://www.hopkinsmedicine.org/healthlibrary/conditions/surgical_care/surgical_site_infections_134,144/>.
(2) The Center for Disease Control. "FAQ's about Surgical Site Infections."Web. <http://www.cdc.gov/HAI/pdfs/ssi/SSI_largertext.pdf>.
(3) Kusek, Linda. "Preventing central line-associated bloodstream infections."Journal of nursing care quality 27.4 (2012): 283-287.
(4) The Center for Disease Control. "FAQ's about Catheter-Associated Bloodstream Infections."Web. <http://www.cdc.gov/hai/pdfs/bsi/BSI_tagged.pdf>.
(5) "Central Line-associated Bloodstream Infections: Resources for Patients and Healthcare Providers." 2010.Web. <http://www.cdc.gov/HAI/bsi/CLABSI-resources.html>.
(6) The Centers for Disease Control and Prevention. "Catheter-associated Urinary Tract Infections (CAUTI)." 2015.Web. <http://www.cdc.gov/HAI/ca_uti/uti.html>.
(7) Nicolle LE. Catheter associated urinary tract infections. Antimicrob Resist Infect Control. 2014;3:23. doi: 10.1186/2047-2994-3-23. <http://www.aricjournal.com/content/3/1/23/about#citations>.
(8) The Centers for Disease Control and Prevention. "Catheter-associated Urinary Tract Infections." 2010.Web. <http://www.cdc.gov/HAI/ca_uti/cauti_faqs.html>.
(9) Gordon, Steven, Tedja, Rudy, Cleveland Clinic. "Hospital–Acquired, Health Care–Associated, and Ventilator–Associated Pneumonia."Web. <http://www.clevelandclinicmeded.com/medicalpubs/diseasemanagement/infectious-disease/health-care-associated-pneumonia/Default.htm>.
(10) Najafi, Shaheen. "Clostridium difficile." 2011.Web. <https://microbewiki.kenyon.edu/index.php/Clostridium_difficile>.
(11) Blakney, Rebekah, et al. "The relationship between patient functional status and environmental contamination by Clostridium difficile: a pilot study."Infection (2015): 1-5. http://link.springer.com/article/10.1007%2Fs15010-015-0770-1
(12) The Centers for Disease Control and Prevention. "Clostridium difficile Infection Information for Patients." 2015.Web. <http://www.cdc.gov/hai/organisms/cdiff/Cdiff-patient.html>.
(13) The Center for Disease Control. "FAQ's about Catheter-Associated Urinary Tract Infections."Web. <http://www.cdc.gov/hai/pdfs/uti/CA-UTI_tagged.pdf>.
(14) The Center for Disease Control. "FAQ's about Ventilator-Associated Pneumonia."Web. <http://www.cdc.gov/HAI/pdfs/vap/VAP_tagged.pdf>.
(15) Tenover FC, Åkerlund T, Gerding DN, et al. Comparison of Strain Typing Results for Clostridium difficile Isolates from North America. Journal of Clinical Microbiology. 2011;49(5):1831-1837. doi:10.1128/JCM.02446-10. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3122689/>.
(16) I. Marsh, R. Whittington, D. Cousins, PCR-restriction endonuclease analysis for identification and strain typing ofMycobacterium aviumsubsp.paratuberculosisandMycobacterium aviumsubsp.aviumbased on polymorphisms in IS1311, Molecular and Cellular Probes, Volume 13, Issue 2, April 1999, Pages 115-126, ISSN 0890-8508, http://dx.doi.org/10.1006/mcpr.1999.0227.
<http://www.sciencedirect.com/science/article/pii/S089085089990227X>.
(17)Bidet, P., Barbut, F., Lalande, V., Burghoffer, B. and Petit, J.-C. (1999), Development of a new PCR-ribotyping method for Clostridium difficile based on ribosomal RNA gene sequencing. FEMS Microbiology Letters, 175: 261–266. doi: 10.1111/j.1574-6968.1999.tb13629.x
(18) Bidet, Philippe, et al. "Development of a new PCR-ribotyping method for Clostridium difficile based on ribosomal RNA gene sequencing." FEMS Microbiology Letters 175.2 (1999): 261-266.
<http://femsle.oxfordjournals.org/content/175/2/261.figures-only>.
(19) Janezic, Sandra, and Maja Rupnik. "Molecular typing methods for Clostridium difficile: pulsed-field gel electrophoresis and PCR ribotyping." Clostridium difficile. Humana Press, 2010. 55-65. <http://link.springer.com/protocol/10.1007%2F978-1-60327-365-7_4#page-1>
(20) Eckert, Catherine, et al. "Prevalence and pathogenicity of binary toxin–positive Clostridium difficile strains that do not produce toxins A and B." New Microbes and New Infections 3 (2015): 12-17.
<http://ac.els-cdn.com/S2052297514000055/1-s2.0-S2052297514000055-main.pdf?_tid=adfb4866-e7a7-11e4-a7ec-00000aab0f01&acdnat=1429566989_20598ad3aeca28918c6a8061cc63e8a3>.
(21)Kuehne, Sarah A., et al. "The role of toxin A and toxin B in Clostridium difficile infection." Nature 467.7316 (2010): 711-713. <http://www.nature.com/nature/journal/v467/n7316/pdf/nature09397.pdf>.
(22) Carter, Glen P., Julian I. Rood, and Dena Lyras. "The role of toxin A and toxin B in the virulence of Clostridium difficile." Trends in microbiology 20.1 (2012): 21-29.
<http://ac.els-cdn.com/S0966842X1100196X/1-s2.0-S0966842X1100196X-main.pdf?_tid=77e5bb16-e7ad-11e4-8275-00000aab0f27&acdnat=1429569475_da85a2645d2046d4eb2278f4c889fe9a>
(23) Erik R. Dubberke, Philip Carling, Ruth Carrico, Curtis J. Donskey, Vivian G. Loo, L. Clifford McDonald, Lisa L. Maragakis, Thomas J. Sandora, David J. Weber, Deborah S. Yokoe and Dale N. Gerding (2014). Strategies to Prevent Clostridium difcile Infections in Acute Care Hospitals: 2014 Update. Infection Control & Hospital Epidemiology, 35, pp 628-645 doi:10.1086/522262 <http://journals.cambridge.org/action/displayAbstract?fromPage=online&aid=9496695&fileId=S0195941700093279>.
(24) Mayo Clinic Staff. "C. difficile Infection- Treatments and Drugs." 2013.Web. <http://www.mayoclinic.org/diseases-conditions/c-difficile/basics/treatment/con-20029664>.
(25) Advancing Excellence in America's Nursing Homes. Prevent and Manage Infections Safely: C. Difficile., 2013. Web.
<https://www.nhqualitycampaign.org/files/AE_Factsheet_for_Leadership_Infections.pdf>.
(26) The Centers for Disease Control and Prevention. "Nearly half a million Americans suffered from Clostridium difficile infections in a single year." February 25 2015.Web. <http://www.cdc.gov/media/releases/2015/p0225-clostridium-difficile.html>.
(27) Furuya-Kanamori, Luis, et al. "Clostridium difficile Infection Seasonality: Patterns across Hemispheres and Continents–A Systematic Review." PloS one10.3 (2015). <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4361656/pdf/pone.0120730.pdf>.
(28) Varnell, Rebecca. "Clostridium difficile infection and fecal bacteriotherapy." 2015.Web. <https://microbewiki.kenyon.edu/index.php/Clostridium_difficile_infection_and_fecal_bacteriotherapy>.
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2009, Kenyon College.
