Transmission of T. Forsythia to humans from a dog bite
Introduction
Tannerella forsythia is an infectious microbe found in the oral cavities of mammals. It is often associated with the periodontal diseases of gingivitis and periodontal disease. Gingivitis is characterized by tissue inflammation, while periodontitis is characterized by alveolar bone resorption as well as bone and tooth loss. The symptoms of these diseases give T. forsythia and other periodontal disease-causing bacteria their namesake. These diseases/infections become present when plaque buildup is great and allow T. forsythia to have a place to live and reproduce. In the case of dogs, it is common for them to have problems with periodontal diseases due to the fact that canines cannot brush their teeth. This poses a problem to humans that are bitten by canines with plaque buildup. If this happens, humans run the risk of being exposed to T. forsythia, and have the potential to get an infection of the epidermis in the form of an abscess.
The old adage: "A dog's mouth is cleaner than a human's" suggests that the microbiome in a dog's mouth is not as infectious or harmful as that of a human's. Dogs tend to use their mouths more than humans due to it being a much stronger and more useful sense than in humans. Canines are constantly licking things and putting things into their mouths because they do not know what it is. Knowing this, and how infectious certain pathogens such as T. forsythia can be, poses the question: Are dogs' mouths really cleaner than humans'?
Tannerella forsythia
T. forsythia is a bacterium found within the mouths and along the gum lines of mammals and primates. More specifically, it is most commonly found in the oral cavities of humans, dogs, cats, and monkeys. When a gram stain is applied, T. forsythia is known to be Gram-negative and survives under anaerobic conditions.[1] T. forsythia only appears in culture from the mouths of mammals with periodontal diseases and has been cultured from abscesses on humans caused by the puncture wounds from infected teeth (caused by humans as well as dogs). T. forsythia is labeled under the species Bacteroidetes, which led to its original name, Bacteroides forsythia. When T. forsythia was initially discovered, researchers had a difficult time classifying it taxonomically. The microbe did not resemble any previously discovered species of oral or enteric gram-negative, anaerobic rods, particularly because of its cell morphology and slow and fastidious growth requirements (Tanner and Izard 2006).[2] T. forsythia has a short, rod-shaped body with tapered ends, and its lack of a flagellum or pili makes it non-motile. Figure 1 shows an image of a T. forsythia cell with both an S-layer and a lack of an S-layer.[3] It can bee seen that it does not have any pilli or flagellum for moevement, and has a circular shape. This lack of mobility or any sort of attachment appendage causes T. forsythia to be reliant on a biofilm in the form of plaque for it to attach to the surface of teeth or other periodontal tissues. These biofilms play an important role in T. forsythia virulence factors and are the ultimate cause of periodontal diseases in mammals. This microbe has an S-layer comprised of glycoprotein that protects both the inner and outer membranes of the cell. This surface/S-Layer comprised of cell surface glycoproteins plays a role in the microbe’s attachment and integration into the oral cavity (Lee et al. 2006).[4] Interestingly, T. forsythia and the biofilms they comprise are resistant to bile. They possess the ability to hydrolyze the components of bile, which is important for a microbe that lives in the oral cavities and have the potential to be exposed to these extreme pH conditions.
Red Complex
T. forsythia is a part of an ensemble of microbes known as the “Red Complex.” This group of bacteria consists of T. forsythia, Treponema denticola, and Porphyromonas gingivalis. All of these bacteria are typically found in the oral cavities of mammals. They all are the cause of periodontal diseases such as periodontitis and gingivitis. One of the symptoms and initial clues that these bacteria are present in the periodontal tissues is swelling, inflammation, and bleeding when probed. This is where the name Red Complex comes from.
Red complex bacteria grow and live in a biofilm attached to the periodontal tissues. This surface attached biofilm is more generally known as plaque and can be found in the subgingival area found around and underneath the teeth (Homma).[5] T. forsythia, other red complex bacteria, and their byproducts are recognized by the host’s immune system and leads to the secretion of cytokines that recruit phagocytic cells such as neutrophils and macrophages. Recognition of any of these bacteria causes the body to release antimicrobial peptides in an attempt to kill off the intrusive bacteria (Homma).[5] In response, the body produces unsuppressed inflammation that causes tissue destruction and increased periodontal pocket depth (Homma).[5] The red, inflamed tissue that results is how the red complex gained its namesake. There is also the possibility for the destruction of the supporting tissues around the teeth as well as the potential for bone/tooth loss. If any of these symptoms are present in your dog, it is recommended that you take them to them a veterinarian because members of the Red Complex are easily transmittable from organism to organism.
Culture
T. forsythia is a difficult microbe to culture because it is anaerobic. Since these bacteria require environments without Oxygen, these cultures are difficult and expensive to create. It is a relatively slow growing bacterium and requires fastidious growth conditions. If researchers can create these conditions, the fact that T. forsythia is anaerobic poses problems when attempting to perform PCR amplification for further studies. T. forsythia is also known to be able to metabolize a range of substrates, and like other enteric Bacteroidetes species, hydrolyzes aesculin, a major component in bile salts. T. forsythia’s ability to break down aesculin when present, allows it to be bile-resistant. This is a major factor in it being able to build a biofilm on the periodontal tissues. In culture, the colonies of this microbe are very small and are opaque colored. The colonies do not grow on normal agar plates or in planktonic culture.
It has been discovered that from dog bites, bacteria from the Bacteroidetes species could be cultured.[6] Figure 2 to the right shows the relative abundance of various bacterial pathogens that are normally cultured from the mouths of canines with periodontal infections.[7] When cultured from bite wounds inflicted by dogs, the only growth conditions that stimulated T. forsythia was the bacterial cell wall component, N-Acetlymuramic acid. N-Acetylmuramic acid (NAM) is an ether of lactic acid and N-Acetylglucosamine, and can often be found in the peptidoglycan of bacterial cell walls. It is not known why this specific compound is important to T. forsythia growth in vivo. This is especially interesting because when observed in vitro, the only other source of NAM comes from other autolyzed oral bacteria (Roy et al. 2010).[8]
Roy et al. (2010) examined the influence of sialic acid on the stimulation of T. forsythia biofilm growth. On a natural biofilm growth medium, varying amounts of sialic acid were used instead of NAM under the preferred (anaerobic) conditions of T. forsythia.[8] The results showed that sialic acid alone does stimulate the growth of this infectious microbe. However, when sialic acid and NAM are combined as a medium, T. forsythia colonies could grow, but were smaller and not as prevalent (Roy et al. 2010).[8]
Periodontal Diseases in Canines
Periodontal infections are some of the most common in dogs as well as cats today.[9] They first start to form when food buildup occurs on a dog’s teeth, and is not properly wiped away. Canines have no way of brushing their teeth so buildup occurs rapidly and with ease in a dog. Since dogs use taste as a major sense, they are always licking foreign substances and putting things into their mouths. The buildup that ensues acts as a breeding ground for T. forsythia and other Red Complex bacterium. The food and debris buildup then transforms into plaque and accumulates along the dog’s gum line. Plaque can then transform into calculus when combined with saliva and other minerals that are common in a dog’s mouth. Calculus is the hardening of plaque, and because this occurs along the gum line, the result is gum irritation and inflammation.[9] These buildups are breeding grounds for bacteria that live in oral cavities. Microbes from the Bacteroidetes, Actinomyces, and Streptococcus families enjoy these kinds of conditions and have been observed in cultures from canine tooth plaque. These symptoms are triggers for gingivitis and periodontal disease.
In extreme cases, calculus will build up under the gum and cause separation between the gum and the tooth. This leads to bone degeneration, tooth loss, and pus formation, and the canine will officially have an irreversible periodontal disease. In diagnosing periodontal diseases, a veterinarian will probe the infected area of the dog’s mouth, and if more than two millimeters of space are found between the tooth and gum of the affected area, then the canine officially has a periodontal disease.[9] Figure 3 shows a canine with severe plaque and calculus buildup. This dog has many hardened layers or yellowish calculus that must be removed to allow the veterinarian to begin treatment.[10]
Certain canine breeds are more likely to be affected then others. The toy breeds are born with crowded teeth, which make it more likely that they will struggle with plaque buildup. Dogs that groom themselves also run a greater chance of getting an infection due to the nature of some areas of the body these dogs have to clean. Poor nutrition can also be a problem because the dog may not be receiving enough nutrients to ward off disease, or they may be receiving nutrients that are helping the already formed plaque to harden. The best way to prevent your dog from receiving a periodontal infection is to brush their teeth regularly and wipe their mouths after they eat to remove any food buildup. (PetMD)[9]
Attachment and Metabolism
Like all bacteria, T. forsythia has an outer layer known as the S-layer. This S-layer is different from other prokaryotes though. Biochemical and molecular genetics experiments show that the structure of a T-forsythia S-layer is unique (Yoo et al., 2007).[11] According to Lee et al. (2006), it consists of a pair of separate glycoproteins that show almost no homology to other S-layer proteins or any other proteins with sugars attached to them within other bacterium.[12] Interestingly, Sabet et al. proved that the S-layer of T. Forsythia is a presumed virulence factor mediating certain cellular interactions such as a special form of agglutination known as hemagglutination (involving red blood cells), attachment and infiltration of the periodontal tissues, and protective immunity (Yoo, et al. 2007).[11] It is also believed that S-layer proteins play a role in attachment to the tissues and membranes of the oral cavities of infected individuals.
Yoo et al. 2007 has proven that BspA, a known outer cell-surface protein, acts as a virulence factor in the pathogenesis of T. forsythia.[11] Honma et al. (2001) showed with their research that BspA is involved with the binding to fibronectin and other extracellular matrix proteins of a host epithelial cell.[13] It has also been ascertained that BspA intercedes epithelial cell attachment and invasion (Inagaki et al., 2006)[14], alveolar bone loss in mice (Sharma et al., 2005a)[15], and coaggregation with another oral cavity dwelling bacterium, Fusobacterium nucleatum (Sharma et al., 2005b) (Yoo et al. 2007).[16][11] F. nucleatum functions as an important microbe in dental plaque biofilm formation. Oral cavity bacteria do not have the necessary attachment machinery to latch onto a oral tissue epithelium on their own, so they rely on proteins such as those in the S-layer and BspA to help with attachment as well as biofilms to help keep them in place.
Other studies have shown that protein phosphorylation and cytoskeletal remodeling are necessary for the incursion of host epithelium and periodontal tissues (Inagaki et al., 2006).[14] This allows the cell to avoid being initially exposed to the host’s immune defenses. Terminal sugars known as Sialic acids are normally found on the external surface of glycoproteins, as well as the cell surface of T. forsythia (Angata & Varki 2002; Varki 1997).[17][18] Sialic acid assists in a variety of biological functions such as extracellular interactions, the stabilization of glycoproteins and other cellular proteins to the exterior of the cell, and hiding ligand-binding receptors from the body’ immune system (Angata & Varki, 2002; Varki, 1997; Bradshaw et al., 1998).[17] These sialic acids are important to biofilm formation and adhesion to periodontal tissues. The enzyme, NanH sialidase, synthesizes sialic acid and is an important molecular structure when trying to develop a targeted antibiotic for T. forsythia.[18] By turning on the salidase inhibitor, there is the potential to prevent biofilms from forming on the surface of periodontal tissues due to sialic acid not being syntehsized. This targeted approach increases the potential of limiting periodontal diseases from occurring and prevents transmission from canine to humans.
T. forsythia is classified as an asacchrolytic bacterium due to its inability to break down carbohydrates for energy. Instead, it utilizes peptides that it breaks down from the proteins of its host. This is done via a trypsin-like protease and the PrtH protease. These enzymes are also considered to be a part of T. forsythia virulence because of their ability to degrade host periodontal tissues and cleave components of the host’s immune system (MicrobeWiki, Tannerella Forsythia).[1] The trypsin-like protease is involved in the degradation of host proteins into smaller peptides and does not appear to have much of an affect on virulence. However, the PrtH protease cleaves larger protein substrates while also functioning as a detachment factor. This attachment function helps T. forsythia adhere to the host epithelium and causes the immune response of the release of chemokine IL-8 (Sharma 2010).[19] When this protein is found in large quantities, it is known as forsythia detachment factor (FDF) (Nakajima et al. 2006).[20]
Abscesses
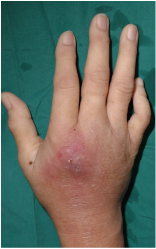
Normally found in oral cavities, bacteria such as T. forsythia have been cultured in other locations on the human body. T forsythia is found in abscesses that form from a cut or puncture wound caused by an infected tooth. An instance where this could possibly occur would be if a dog bites a human and the canine is suffering from a periodontal disease. The dog is most likely playing host to T. forsythia and other Red Complex bacteria. These microbes are hiding in the plaque biofilm that has built up along the periodontal tissues. When an infected dog bites a human, the canine’s teeth puncture the epidermis of that person, and pass along the bacteria to a new host. This transmission of T. forsythia allows for the infection of host cell epithelium and the underlying sebaceous glands. Members of the Red Complex can be detected in and are often the cause of what are known as acute periradicular abscesses.
Abscesses are an accumulation of exudate (fluid emitted through a wound) consisting of bacteria, bacterial by-products, inflammatory cells, lysed inflammatory cells, and the content of those cells (Ozebek, et. al, 2010).[21] When these bacteria enter the surrounding epithelial cells, the immune system is unable to suppress the invasion. The breaking or puncturing of the upper layers of skin causes abscesses. Bacteria are able to get under the skin and infect the sebaceous and sweat glands that lie underneath the epidermis.[22] This causes an inflammatory response as the body attempts to rid itself of these pathogens. The inflammatory response causes a number of symptoms that are important in properly diagnosing abscesses. In the middle of an abscess, there is liquefaction and an aggregate of dead cells. Along with the liquid and dead cells present are bacteria and other debris that are difficult to remove. As time goes on the infected area begins to grow, which creates tension under the skin and greater inflammation characterized by redness, swelling, warmth of the affected area, and pain to the touch. Pressure from the tension causes the pain. Meanwhile, the bacteria are infecting the sebaceous glands and helping to generate pus. This pus is a buildup of dead white blood cells that were attempting to stop the spread of infection, along with other dead epithelial cells and debris.[22] Figure 4 gives a good image of an infected abscess. This wound is very red and beginning to pus, suggesting that it has been infected for a while.[23]
In abscesses, knowing the specific microbe that has caused the infection is important to treating it. Knowing specifically what the microbe is and where it came from allows for the proper antibiotic to be prescribed.
Infections caused by T. forsythia have been known to be most susceptible to the following antibiotics: amoxicillin, clavulanate, ampicillin, doxycycline, tetracycline, and clindamycin (Tanner & Izard, 2006), with amoxicillin and clavulanate being the most effective.[2] Abscesses are often mistaken for cellulitis, so this is another reason why proper detection and identification of the correct bacterium is so crucial. Often with an abscess infection, antibiotics are not enough to completely cure these infections. They tend to need to be incised by a medical professional so they can drain, because drainage does not occur naturally. In extreme cases, abscesses will need to be removed surgically if they have damaged the surrounding tissues badly enough.[22] It is necessary to have an abscess treated as soon as possible because there is the possibility that they can begin to infect deeper tissues and possibly allow the infection enter the bloodstream. Patients with compromised immune systems are especially at risk due to the fact that they do not have the proper defense systems to ward off infections.
Conclusion
There is an old adage that goes like so: “A dog’s mouth is cleaner than a human’s mouth.” With bacteria such as T. forsythia making plaque biofilms in the mouths of canines, it is safe to say that this is not always the case. These plaque biofilms are the cause of periodontal diseases and are the main threat to humans if and when a dog bites them. With a resistance to bile and multiple systems of attachment to periodontal tissues, T. forsythia is a dangerous bacterium to both dogs and humans. The oral cavity of mammals acts a perfect place for these anaerobic microbes to live. Plenty of epithelial proteins provide enough nutrients for the trypsin-like proteases to create peptides for the microbe to consume. The mouths of dogs are excellent places to reproduce given that dogs do not brush their teeth on a normal basis. This allows T. forsythia to live and replicate without the pressure of having the biofilm it is generating to be wiped away. In a dog’s mouth, T. forsythia has a constant food source and does not have to worry about the selection pressure of brushing one’s teeth that is a problem for microbes that inhabit human oral cavities.
T. forsythia is not as prevalent in culture from humans. This is due to humans brushing their teeth regularly and seeing a dentist twice a year. However, in countries with poor economic and health care systems, certain periodontal diseases are seen more often. Without the infrastructure to provide dental care, periodontal diseases are more of a problem in these countries than in wealthier ones.[24] It is the same situation for dogs. In wealthier countries that can provide proper veterinary treatment, dogs are less likely to have infections within their oral cavities. Certain bad habits that humans have such as smoking and consumption of other tobacco products also play a role in plaque biofilm formation. Even with a first-rate economic system, members of the CDC predict that approximately 1 in 2 Americans are dealing with a periodontal infection.[24] In both dogs and humans, the severity of periodontal infections gets worse with age. As age increases, the complexity and effectiveness of the immune system decreases. Bacteria such as T. forsythia capitalize on this by infecting an older host without needing to worry about being quickly detected or the immune system sending out a defense against the microbe. This increases the rate of infection as well as the severity of the resulting immune response. With a weakened immune system comes the possibility of a severely infected abscess. In elderly patients, it is more common that bacterial infections need to be surgically removed due to their increased prevalence.
References
- ↑ 1.0 1.1 "Tannerella Forsythia." MicrobeWiki. N.p., n.d. Web. 24 Apr. 2016.
- ↑ 2.0 2.1 "Tannerella Forsythia, a Periodontal Pathogen Entering the Genomic Era." Periodontology 2000 Periodontol 2000 42.1 (2006): 88-113. Web.
- ↑ [http://jdr.sagepub.com/content/90/1/109/F1.expansion.htm Sketot, G. "Potential of the Tannerella Forsythia S-layer to Delay the Immune Response." Journal of Dental Reseearch (2010): n. pag. Web. 18 Apr. 2016.
- ↑ "Identification and characterization of the genes encoding a unique surface (S-) layer of Tannerella forsythia." (2006) Gene 371:102–111.
- ↑ 5.0 5.1 5.2 [[1] “Tannerella Forsythia- A new intruder of the Human Periodontal Pocket.” (n.d.): 1-2. Spotlight on a Lab. University of Buffalo.]
- ↑ "Microbiology of Animal Bite Wound Infections." Clinical Microbiology Reviews. American Society for Microbiology, 24 Apr. 2011. Web. 11 May 2016.
- ↑ [http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1517-83822007000100006 "Detection of Putative Periodontal Pathogens in Subgingival Specimens of Dogs." Detection of Putative Periodontal Pathogens in Subgingival Specimens of Dogs. Brazilian Journal of Microbiology, Jan. 2007. Web. 11 May 2016
- ↑ 8.0 8.1 8.2 "A Novel Sialic Acid Utilization and Uptake System in the Periodontal Pathogen Tannerella Forsythia." Journal of Bacteriology. American Society for Microbiology (ASM), 26 Feb. 2010. Web. 24 Apr. 2016.
- ↑ 9.0 9.1 9.2 9.3 "PetMD." Dog Gum Disease. PetMD, n.d. Web. 21 Apr. 2016.
- ↑ [http://www.kingwestvets.com/brush-dogs-teeth/ "How to Brush Dogs' Teeth | Step by Step Guide | King West Vets." King West Vets. King West Vest Veterinary Clinic, 15 Jan. 2013. Web. 11 May 2016
- ↑ 11.0 11.1 11.2 11.3 "Identification of Tannerella Forsythia Antigens Specifically Expressed in Patients with Periodontal Disease." FEMS Microbiology Letters 275.2 (2007): 344-52. Web. 23 Apr. 2016.
- ↑ Identification and characterization of the genes encoding a unique surface (S-) layer of Tannerella forsythia. Gene 371:102–111(2006).
- ↑ "Development of a Gene Inactivation System for Bacteroides Forsythus: Construction and Characterization of a BspA Mutant." Infection and Immunity. American Society for Microbiology, July 2001. Web. 11 May 2016.
- ↑ 14.0 14.1 Porphyromonas gingivalis vesicles enhance attachment, and the leucine-rich repeat BspA protein is required for invasion of epithelial cells by ‘‘Tannerella forsythia’’. Infect Immun 74, 5023–5028.
- ↑ Tannerella forsythia-induced alveolar bone loss in mice involves leucine-rich-repeat BspA protein. J Dent Res 84:462–467 (2005a).
- ↑ Synergy between Tannerella forsythia and Fusobacterium nucleatum in biofilm formation. Oral Microbiol Immunol 20:39–42. (2005b)
- ↑ 17.0 17.1 Chemical diversity in the sialic acids and related a-keto acids: an evolutionary perspective. Chem Rev 102, 439–470 (2002).
- ↑ 18.0 18.1 Sialic acids as ligands in recognition phenomena. FASEB J 11, 248–255 (1997).
- ↑ "Virulence Mechanisms of Tannerella Forsythia." Periodontology 2000 54.1 (2010): 106-16. Web. 24 Apr. 2016
- ↑ "Isolation and Identification of a Cytopathic Activity in Tannerella Forsythia." Biochemical and Biophysical Research Communications 351.1 (2006): 133-39. Web. 24 Apr. 2016.
- ↑ "Real-time Polymerase Chain Reaction of “red Complex” (Porphyromonas Gingivalis, Tannerella Forsythia, and Treponema Denticola) in Periradicular Abscesses." Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology 110.5 (2010): 670-74. Web. 18 Apr. 2016.
- ↑ 22.0 22.1 22.2 "Abscess: Causes, Symptoms, Tests, and Treatment." WebMD. WebMD, n.d. Web. 11 May 2016.
- ↑ [http://www.handsurgery.sg/hand-infection.html "Hand Infection." Hand & Reconstructive Microsurgery Center. National University Hospital, n.d. Web. 11 May 2016.
- ↑ 24.0 24.1 "CDC: Half of American Adults Have Periodontal Disease | Perio.org." CDC: Half of American Adults Have Periodontal Disease | Perio.org. American Academy of Periodontology, n.d. Web. 24 Apr. 2016.